* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download as a PDF
Convolutional neural network wikipedia , lookup
Haemodynamic response wikipedia , lookup
Donald O. Hebb wikipedia , lookup
Adult neurogenesis wikipedia , lookup
Neuroregeneration wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
Environmental enrichment wikipedia , lookup
Brain Rules wikipedia , lookup
Apical dendrite wikipedia , lookup
Neuroeconomics wikipedia , lookup
Aging brain wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Biochemistry of Alzheimer's disease wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Synaptogenesis wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Single-unit recording wikipedia , lookup
Neuroplasticity wikipedia , lookup
Multielectrode array wikipedia , lookup
Neural oscillation wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Artificial general intelligence wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neural coding wikipedia , lookup
Anatomy of the cerebellum wikipedia , lookup
Sexually dimorphic nucleus wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Axon guidance wikipedia , lookup
Mirror neuron wikipedia , lookup
Development of the nervous system wikipedia , lookup
Central pattern generator wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Nervous system network models wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Hypothalamus wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Circumventricular organs wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Optogenetics wikipedia , lookup
Neuroanatomy wikipedia , lookup
Brain (1996), 119, 1145-1152
Human brainstem preganglionic parasympathetic
neurons localized by markers for nitric oxide
synthesis
W. P. Gai and W. W. Blessing
Departments of Medicine and Physiology and Centre for
Neuroscience, Flinders University, Australia
Correspondence to: W. P. Gai, Department of Physiology,
Flinders Medical Centre, Bedford Park, SA, Australia 5042
Identification of human parasympathetic preganglionic
neurons in pontomedullary regions has been largely based
on studies using cholinesterase histochemical procedures,
and so far there is no adequate account of the location of these
cells. Nitric oxide synthase (NOS) is present in brainstem
parasympathetic preganglionic salivatory neurons in the
rabbit (Zhu et al., 1996). In the present study we have used
histochemical and immunohistochemical staining for NOS to
examine possible preganglionic parasympathetic neurons in
the human brainstem. We examined, in five human brains,
the distribution, through the caudal pons and rostral medulla,
of NOS-positive neurons in serial sections stained with
NADPH diaphorase for histochemistry, and with antibodies
against neuronal NOS peptide for immunohistochemistry. In
scattered pontomedullary regions (rostral to the dorsal motor
nucleus and the nucleus ambiguus) known to contain
parasympathetic preganglionic neurons in animals, we
observed groups of NOS-positive neurons which correspond
in morphology and distribution with NOS-positive parasympathetic preganglionic neurons in rabbits. These neurons
are probably parasympathetic preganglionic neurons in the
human brainstem, involved in the control of lacrimation,
salivation, oral and nasopharyngeal secretion, as well as the
control of the dilation of extra- and intracranial blood vessels.
Keywords: nitric oxide synthase; parasympathetic preganglionic; human brainstem; lacrimation; salivation
Abbreviation: NOS = nitric oxide synthase
Introduction
The most rostral cranial preganglionic parasympathetic
neurons are located in the midbrain, in the Edinger-Westphal
nucleus (axons in cranial nerve III). The most caudal cranial
parasympathetic neurons are located in the medulla, in the
dorsal motor nucleus of the vagus and in the nucleus ambiguus
(axons in cranial nerve X). Other brainstem parasympathetic
preganglionic neurons project via cranial nerves VII and IX
to sphenopalatine, submandibular, sublingual, otic and other
small unnamed ganglia in various extracranial locations.
Postganglionic neurons project to lacrimal and salivary
glands, to mucosal glands in the mouth and nasopharynx, as
well as to arteries supplying the cranial vasculature, including
major cerebral arteries.
In spite of the diverse functions served by the various target
tissues, the parent preganglionic parasympathetic neurons are
usually lumped together and assigned to the 'salivatory
nuclei', either to the superior salivatory nucleus (axons in
cranial nerve VII) or the inferior salivatory nucleus (axons
© Oxford University Press 1996
in cranial nerve IX). Accounts of the location of these
salivatory nuclei in the human brainstem are sketchy.
Textbooks of human neuroanatomy usually refer to studies
utilizing the cholinesterase histochemical procedure as a
marker of acetylcholine-synthesizing neurons (Lewis and
Shute, 1959). The more critical accounts acknowledge the
lack of evidence available for specifying the location of
these parasympathetic neurons in the human (Ranson and
Clark, 1959).
In the human brainstem Feiling (1913), on the basis of
evidence from retrograde neuronal degeneration in a patient
who died with injuries to the IX, X, XI and XII nerves,
identified salivatory neurons just dorsal to the inferior olivary
nucleus. Braak (1972), in a Nissl study, identified the human
inferior salivatory nucleus dorsomedial to the rostral portion
of the nucleus tractus solitarius. A similar region was also
designated as the inferior salivatory nucleus by Paxinos et al.
(1990), and by McRitchie and Tork (1993) on the basis of
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
Summary
1146
W. P. Gai and W. W. Blessing
In the rabbit, the parasympathetic preganglionic neurons
described by Hanamori and Smith (1989) on the basis of
retrograde transport studies from appropriate branches of
cranial nerves VII and IX, are very similar, in both distribution
and morphology, to neurons containing NOS in the same
general region of the brainstem (Gai et al., 1995). We recently
confirmed in the rabbit that NOS is present in nearly all
neurons retrograde labelled after application of tracer to the
chorda tympani nerve or to preganglionic axons travelling
along the submandibular duct (Zhu et al., 1996). This close
correspondence between identified preganglionic salivatory
neurons and NOS-positive neurons in the rabbit suggested
that NOS might prove useful for identifying parasympathetic
preganglionic neurons in the human. The distribution of
NOS-positive neurons in the human brainstem, including the
medulla and caudal pons, has been mapped (Kowall and
Mueller, 1988), but not in detail sufficient to determine
whether the distribution of NOS-positive neurons corresponds
to that of the salivatory nuclei defined in animals. We now
present, for the human, a detailed map of NOS-positive
neurons in those pontomedullary regions which, in the rabbit
and in other experimental animals, have been demonstrated
to contain parasympathetic preganglionic neurons.
Table 1 List of cases
Case
Sex
Age
(years)
Post-mortem
delay (h)
Cause of death
N20
N21
N22
N23
N24
F
M
M
F
F
84
88
86
86
71
15
19
25
17
7
Peritonitis
Renal failure
Cancer
Cancer
Cardiac infarct
microtome, and serial sections collected into 15 or 30
consecutive containers. One set of sections was
immunostained using a rabbit polyclonal antiserum, raised
against the C-terminal sequence (amino acid residue 14091429) of rat cerebellar NOS peptide by Dr Colin Anderson
of the Department of Anatomy and Cell Biology, University
of Melbourne (available from Auspep Pty Ltd, Parkville,
Victoria, Australia). Staining of neural tissue is abolished by
preincubation of diluted antiserum with the peptide antigen.
The antiserum recognizes a single band corresponding to
neural NOS in blots from rat brain. Our previous study in
rabbit (Gai et al., 1995) and the present study in humans,
demonstrates that the antiserum stains only neurons. Blood
vessels known to contain endothelial NOS were not stained.
The NOS antiserum was used at 1:2000 dilution. Control
sections were incubated with non-immune rabbit serum, and
there was no neuronal staining in these sections. Another set of
sections was stained with the NADPH diaphorase procedure.
Free-floating sections were incubated for 2-3 h at 37°C, in
a solution containing 1.25 mg nitroblue tetrazolium, 5.0 mg
beta-NADPH (Sigma Chemical, Sydney, Australia), and 0.1 %
Triton X-100 in 10 ml of 0.1 M Tris buffer (pH 8.0). Control
sections were processed identically except NOS antibody or
beta-NADPH were omitted from the incubation medium.
Sections were mounted, dehydrated and coverslipped. Other
section series were stained for Nissl substance (cresyl violet)
or myelin (Weil method).
The microscopic image of a section was superimposed
onto a Macintosh Ilfx computer screen using a camera lucida
device. Boundaries of nuclei or fibre bundles were outlined
under dark-field illumination, and positive cells marked under
bright-field illumination. The information was recorded in
the computer using Canvas software (Deneba, Fla., USA).
The rostrocaudal distance of a section was calculated in
reference to the obex.
Results
Material and methods
We examined five human brains, obtained at autopsy from
patients dying from non-neurological causes {see Table 1).
The brains were perfuse-fixed with 4% formaldehyde and
2% picric acid in 0.1 M phosphate buffer at pH 7.4, as
previously described in detail (Halliday et al., 1988). The
brainstem was sectioned transversely (50 nm) on a freezing
Nitric oxide synthetase positive neurons were observed in all
brains studied. The NADPH diaphorase reaction and the
NOS antibody procedure stained similar sets of neurons
throughout the brainstem. No positive staining was observed
in control sections. The overall distribution of NOS-positive
neurons was similar, but there was a gradual decrease in the
number and staining intensity of positive neurons with longer
post-mortem delays, for both diaphorase reaction and NOS
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
cholinesterase histochemistry. Neither of the latter studies
defines a superior salivatory nucleus, and no inferior
salivatory nucleus was identified in the human brainstem in
a detailed study using both cholinesterase histochemistry and
choline acetyltransferase immunohistochemistry (Mizukawa
et al., 1986).
In experimental animals, appropriate retrograde intraxonal
tracing studies demonstrate that parasympathetic preganglionic neurons are located in the pons at the level of
the facial nucleus, and in rostral portions of the nucleus
tractus solitarius. This has been demonstrated in rat (Hiura,
1977; Contreras et al., 1980; Nicholson and Severin, 1981;
Mitchell and Templeton, 1981), rabbit (Matsuo et al., 1980;
Hanamori and Smith, 1989), hamster (Whitehead and Frank,
1983), cat (Satomi et al., 1979; Nomura and Mizuno, 1981;
Tramonte and Bauer, 1986), dog (Chibuzo and Cummings,
1980) and monkey (Perwaiz and Karim, 1982). The
preganglionic neurons do not form discrete cell groups, so
that it is difficult to identify them on Nissl appearance alone;
nor is there a clear rostrocaudal distinction between neurons
with axons exiting the brainstem in VII and those exiting in
IX, so that Contreras et al. (1980) question the wisdom of
the traditional division of the neurons into superior and
inferior salivatory nuclei.
NOS in preganglionic parasympathetic neurons
Discussion
The human pontomedullary region examined in the present
study contains many NOS-positive neurons, distributed in a
manner quite similar to neurons demonstrated to be
parasympathetic preganglionic neurons by appropriate
retrograde transport studies in experimental animals {see
Introduction). The distribution is also similar to that of NOSpositive neurons in the rabbit, cells which have been shown
by double-labelling procedures to include salivatory
preganglionic neurons (Zhu et al., 1996). Most of them are
medium sized (Zhu et al., 1996). We consider it reasonable to
suppose that, in humans, NOS also occurs in parasympathetic
preganglionic neurons with axons exiting in VII and IX
cranial nerves. Other brainstem parasympathetic preganglionic neurons (with axons exiting in III and X) do not
appear to contain markers for NOS (Kowall and Mueller,
1988; W. P. Gai and W. W. Blessing, unpublished
observations). The rostrally located NOS-positive neurons
(at levels of rostral half of the facial nucleus) are dispersed
and not easily distinguished from other neurons in Nisslstained sections. The caudally located neurons can be
identified in Nissl sections, but the neurons are continuous
with other neurons of the dorsal motor nucleus of the vagus
which are NOS-negative.
In the human oral spinal trigeminal nucleus, Olszewski
and Baxter (1982) described a cell group termed the oval
nucleus. They state that 'the nucleus lies along the dorsal or
dorsolateral border of the nucleus tractus spinalis trigemini
oralis, and extends from the level of the oral pole of the
dorsal motor nucleus to the caudal pole of the main sensory
trigeminal nucleus, a distance of 5 mm' (p. 167). In plate
XX of Olszewski and Baxter (1982) the oval nucleus is
situated in a region corresponding to the more rostral extent
of the interstitial nucleus of the nucleus tractus solitarius.
However, in plate XVIII of the atlas, the oval nucleus is
consigned to the oral spinal trigeminal nucleus, ventrolateral
to the true position of the nucleus tractus solitarius at this
rostrocaudal level. The NOS-positive cell group we observed
in the oral spinal trigeminal nucleus is just ventromedial to
the region designated as the oval nucleus in plate XVIII of
Olszewski and Baxter (1982).
The inferior salivatory nuclei, described dorsomedial to
the rostral portion of the nucleus tractus solitarius by Paxinos
et al. (1990), McRitchie and Tork (1993) and Braak (1972),
presumably correspond to the NOS-positive cells shown in
our Fig. ID and E. No superior salivatory cells are identified.
The cholinesterase procedure may have demonstrated these
neurons without being able to distinguish them from the
neurons of the accessory facial nucleus and other
cholinesterase-positive (but NOS-negative) neurons in the
region. Satoh and Fibiger (1985a, b), in a careful histochemical (acetylcholinesterase reaction) and immunohistochemical (choline acetyltransferase antibodies) study of
the baboon brainstem, described parasympathetic preganglionic neurons in regions very similar to those identified
in the present study. Our study is the first to identify these
more rostral parasympathetic neurons in the human brainstem.
The supposed existence of superior and inferior subnuclei
arose from the knowledge that the axons were distributed in
either the facial or the glossopharyngeal nerves. The excellent
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
antibody. In the present study, sections from case N24 (7 h
post-mortem delay) were used for mapping the distribution of
NOS-positive cells. Nitric oxide synthetase-positive perikarya
have been divided into medium sized (long axis >25 (im)
and small neurons (long axis <25 (im).
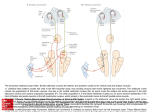
Many NOS-positive neurons were found in pontomedullary
regions (Fig. 1) known to contain preganglionic parasympathetic neurons in experimental animals. Medium-sized
neurons were located in a region bordered by the spinal
trigeminal nucleus laterally, the facial nucleus medially and
the medial vestibular nucleus dorsally. Rostrally, these
neurons were scattered in a narrow region between the oral
spinal trigeminal nucleus and the facial nucleus, medial to
the descending intramedullary fibres of the facial nerve
(Fig. IB and C). At the level of the midportion of the facial
nucleus, ~12 mm rostral to the obex, a few NOS-positive
neurons were found ventrolateral to the facial nucleus
(Fig. ID). At the level ~11 mm rostral to the obex, these
ventrally positioned neurons maintained a similar relation to
the facial nucleus, whereas dorsally located neurons became
more concentrated in a region dorsomedial to the oral spinal
trigeminal nucleus (Fig. IE). Further caudally, at the level
~10 mm rostral to the obex, the ventral cell group was no
longer present, whereas the dorsal group was still quite
well developed, now situated medial to the nucleus tractus
solitarius (Fig. 2F and G). At the level 9 mm rostral to the
obex, only a few large NOS-positive neurons were found
medial to the nucleus tractus solitarius (Fig. 1H).
Most medium-sized NOS-positive neurons had spindleshaped or ovoid perikarya with two prominent dendrites
emanating from the long ends of the cell body (Fig. 2A-D,
G and H). Those NOS-positive neurons located ventral to
the facial nucleus were usually round or ovoid (Fig. 2E
and F). The dendrites of most medium-sized NOS-positive
neurons located rostrally (corresponding to level 11-13 mm
rostral to the obex; see Fig. 1) were arranged in a ventrolateral
to dorsomedial direction, roughly parallel with the trajectory
of descending facial nerve tract (Fig. 2A-F), but many
dendrites of caudally situated neurons were horizontally
arranged (Fig. 2G and H).
As shown in Fig. I, small NOS-positive neurons were
scattered widely in the pontomedullary region including the
region containing medium-sized NOS-positive neurons. The
oral spinal trigeminal nucleus contained a discrete NOSpositive neuron group that was apparent with the naked eye.
This cell group was located in the centre of the oral spinal
trigeminal nucleus, and extended ~5 mm rostrocaudally (Figs
1B-H and 3A and B). Numerous NOS-positive fibres (large
and small calibres) were seen in the intramedullary tracts of
the facial and glossopharyngeal nerves (Fig. 3C-E).
1147
1148
W. P. Gai and W. W. Blessing
A
/
14 mm /
.
NTS / O)
IC
P
so-
• ••
/ ,
/ /
•'
vn /
! •
-•
^
\
2 mm
\
• •
••
Fig. 1 Computer camera lucida
drawings showing the distribution of
NOS-positive neurons in the caudal
pons and rostral medulla. Sections
are arranged from rostral (A) to
caudal (H). The numbers in the
upper left corner of each drawing
indicate the approximate distance to
the obex. The diagrams on the left
show the distribution of all NOSpositive neurons in a 50 Jim thick
section at the given level. The
enlarged views on the right show the
distribution of medium-sized NOSpositive neurons in more detail. Each
marker represents one NOS-positive
neuron. Large squares represent
perikarya with long axis >25 |im.
Small squares represent perikarya
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
NTS
NOS in pregangUonic parasympathetic neurons
1149
PrP
with the long axis <25 |im. Vo =
oral spinal trigeminal nucleus; Vp =
principle sensory trigeminal nucleu:
VI = abducent nucleus; VII = facis
nucleus or facial fibre tract; VIIII =
lateral vestibular nucleus; Vlllm =
medial vestibular nucleus; VIIIs =
superior vestibular nucleus; VIIIsp
spinal vestibular nucleus; icp =
inferior cerebellar peduncle; 10 =
inferior olivary nucleus; mcp =
middle cerebellar peduncle; ml =
medial lemniscus; mlf = medial
longitudinal fasciculus; NTS =
nucleus tractus solitarius; p =
pyramidal tract; PrP = prepositus
hypoglossal nucleus; SO = superior
olivary nucleus; A = nucleus
ambiguus.
.•v!*.
H
9 mm
2 mm
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
NTS
1150
W. P. Gai and W. W. Blessing
5 0pm
B
2 5 u m
5 0 u m
2 5 u m
4
50um
2 5um
'rmseakt
If
H
2 5um
Fig. 2 Photomicrographs showing the morphology of medium-sized NOS-positive neurons in lateral pontomeduliary regions. The
approximate areas from which the photographs were taken are indicated by the horizontal pairs of arrows in Fig. 1. All photomicrographs
are presented as the left side of corresponding brainstem region, with medial to the right. (A) Nitric oxide synthase positive neurons
medial to the oral trigeminal nucleus -12 mm rostral to the obex (see Fig. 1C). (B) Enlarged view of A. (C and D) Nitric oxide synthase
positive neurons in the region between the facial nucleus and descending facial nerve fibres (see Fig. ID). (E and F) Nitric oxide
synthase positive neurons ventrolateral to the facial nucleus (sec Fig. IE). (G) Nitric oxide synthase positive neurons medial to the oral
spinal trigeminal nucleus and the NTS ~i() mm rostral to the obex (refer to Fig. !G). (H) Er.lu.rged view of G.
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
4
NOS in preganglionic parasympathetic neurons
1151
B
20/ym
Fig. 3 (A) Nitric oxide synthase positive neurons and their dendrites in the oral spinal trigeminal nucleus. (B) Enlarged view of A. (C)
Nitric oxide synthase positive fibres in the intramedullary tract of the glossopharyngeal nerve. (D) Nitric oxide synthase positive fibres in
the genu of the facial nerve. (E) Nitric oxide synthase positive fibres in the descending intramedullary tract of the facial nerve.
discussion section in a detailed study of the rat (Contreras
et al., 1980) questions the utility of the superior/inferior
dichotomy, as well as emphasizing the complexity of function
of these parasympathetic preganglionic cells. In the species
studied, neurons with axons exiting in the glossopharyngeal
nerve have a brainstem distribution generally similar to those
with axons exiting in the facial nerve.
In rats, neurons in the more dorsal parasympathetic neurons
project to otic ganglia, to ganglia associated with the
sublingual and submandibular salivary glands, as well as to
mucosal glands in the oral cavity and vessels supplying
all these structures (Contreras et al., 1980). In addition,
postganglionic neurons supply the vertebrobasilar arterial
system (Suzuki, 1989; Haiaetai, 1993; Suzuki and Hardebo,
1993; Shimizu, 1994). By analogy, the more dorsal NOSpositive neurons identified in the present human study also
presumably project to ganglia supplying parasympathetic
innervation to the salivary glands, mucosal glands in the oral
cavity and to the vertebrobasilar arterial system.
Neurons on the lateral apect of the facial nucleus can be
retrograde labelled from the sphenopalatine ganglion and the
greater superficial petrosal nerve, indicating that they are
likely to include lacrimai preganglionic cells, neurons
innervating the vasculature and mucous glands of the nasal
and palatal mucosa, and possibly neurons with postganglionic
connections innervating cerebral blood vessels in forebrain
regions (Contreras et al., 1980; Hara et al., 1993; Shimizu,
1994). These more ventrally located parasympathetic
preganglionic neurons are also observed in retrograde
transport studies from the sphenopalatine ganglion in rabbits,
and the same more ventral region also contains a dense group
of NOS-positive neurons (Zhu et al., 1996). The more
ventrally located parasympathetic preganglionic neurons in
the human may therefore be parasympathetic preganglionic
neurons controlling, via appropriate postganglionic neurons,
the functions of lacrimai glands, as well as mucus-secreting
cells in the nasopharynx. In addition, blood supply to these
structures, as well as to other extracranial vessels and cerebral
blood vessels supplying the forebrain may also be regulated
by these brainstem parasympathetic neurons.
Acknowledgements
We wish to thank Ms R. A. Flook for excellent technical
assistance. This study was supported by the Australian Brain
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
D
1152
W. P. Gai and W. W. Blessing
Foundation (NSW), the Julia Fair Foundation (SA), the
National Heart Foundation of Australia and the NH and MRC.
acetyltransferase immunohistochemistry and acetylcholinesterase
histochemistry. Brain Res 1986; 379: 39-55.
Nicholson JE, Severin CM. The superior and inferior salivatory
nuclei in the rat. Neurosci Lett 1981; 21: 149-54.
References
Braak H. Nuclei of the human brain stem. V. The dorsal
glossopharyngeus-vagus-complex. [German]. Z Zellforsch Mikrosk
Anat 1972; 135: 415-38.
Chibuzo GA, Cummings JF. Motor and sensory centers for the
innervation of mandibular and sublingual salivary glands: a
horseradish peroxidase study in the dog. Brain Res 1980; 189:
301-13.
Feiling A. On the bulbar nuclei, with special reference to the
existence of a salivary centre in man. Brain 1913; 36: 255-65.
Gai WP, Messenger JP, Yu Y-H, Gieroba ZJ, Blessing WW. Nitric
oxide-synthesising neurons in the central subnucleus of the nucleus
tractus solitarius provide a major innervation of the rostral nucleus
ambiguus in the rabbit. J Comp Neurol 1995; 357: 348-61.
Halliday GM, Li YW, Joh TH, Cotton RGH, Howe PRC, Geffen
LB, et al. Distribution of monoamine-synthesizing neurons in the
human medulla oblongata. J Comp Neurol 1988; 273: 301-17.
Hanamori T, Smith DV. Gustatory innervation in the rabbit: central
distribution of sensory and motor components of the chorda tympani,
glossopharyngeal, and superior laryngeal nerves. J Comp Neurol
1989; 282: 1-14.
Hara H, Zhang Q-J, Kuroyanagi T, Kobayashi S. Parasympathetic
cerebrovascular innervation: an anterograde tracing from the
sphenopalatine ganglion in the rat. Neurosurgery 1993; 32: 822-7.
Hiura T. Salivatory neurons innervate the submandibular and
sublingual glands in the rat: horseradish peroxidase study. Brain
Res 1977; 137: 145-9.
Kowall NW, Mueller MP. Morphology and distribution of
nicotinamide adenine dinucleotide phosphate (reduced form)
diaphorase reactive neurons in human brainstem. Neuroscience
1988; 26: 645-54.
Olszewski J, Baxter D. Cytoarchitecture of the human brain stem.
2nd ed. Basel: Karger, 1982.
Paxinos G, Tork 1,
to brainstem nuclei
acetylcholinesterase
nervous system. San
Halliday G, Mehler WR. Human homologs
identified in other animals as revealed by
activity. In: Paxinos G, editor. The human
Diego: Academic Press, 1990: 149-202.
Perwaiz SA, Karim MA. Localization of parasympathetic
preganglionic neurons innervating submandibular gland in the
monkey: an HRP study. Brain Res 1982; 251: 349-52.
Ranson SW, Clark SL. The anatomy of the nervous system. 10th
ed. Philadelphia: W. B. Saunders, 1959.
Satoh K, Fibiger HC. Distribution of central cholinergic neurones
in the baboon (Papio papio). I. General morphology. J Comp Neurol
1985a; 236: 197-214.
Satoh K, Fibiger HC. Distribution of central cholinergic neurons in
the baboon (Papio papio). II. A topographic atlas correlated with
catecholamine neurons. J Comp Neurol 1985b; 236: 215-33.
Satomi H, Takahashi K, Ise H, Yamamoto T. Identification of the
superior salivatory nucleus in the cat as studied by the HRP method.
Neurosci Lett 1979; 14: 135-9.
Shimizu T. Distribution and pathway of the cerebrovascular nerve
fibers from the otic ganglion in the rat: anterograde tracing study.
J Auton Nerv Syst 1994; 49: 47-54.
Suzuki N. Origins, pathways and effects of electrical stimulation of
dilatory peptidergic vascular nerves in rat brain. Lund: Grahn's
Boktryckeri, 1989.
Suzuki N. Hardebo JE. The cerebrovascular parasympathetic
innervation. [Review]. Cerebrovasc Brain Metab Rev 1993; 5:
33-46.
Lewis PR. Shute CCD. Selective staining of visceral efferents in
the rat brain stem by a modified Koelle technique. Nature 1959;
183: 1743^1.
Tramonte R, Bauer JA. The location of the preganglionic neurons
that innervate the submandibular gland of the cat: a horseradish
peroxidase study. Brain Res 1986; 375: 381^1.
Matsuo R, Yamamoto T, Kawamura Y. Morphological and functional
evaluation of the superior salivatory nucleus in rabbits. Exp Neurol
1980: 68: 147-57.
Whitehead MC, Frank ME. Anatomy of the gustatory system in the
hamster: central projections of the chorda tympani and the lingual
nerve. J Comp Neurol 1983: 220: 378-95.
McRitchie DA, Tork I. The internal organization of the human
solitary nucleus. Brain Res Bull 1993; 31: 171-93.
Zhu B-S, Gai W-R Yu Y-H. Gibbins IL, Blessing WW. Preganglionic
parasympathetic salivatory neurones in the brainstem contain
markers for nitric oxide synthesis in the rabbit. Neurosci Lett 1996.
In press.
Mitchell J, Templeton D. The origin of the preganglionic
parasympathetic fibres to the mandibular and sublingual salivary
glands in the rat: a horseradish peroxidase study. J Anal 1981; 132:
513-18.
Mizukawa K, McGeer PL, Tago H, Peng JH, McGeer EG, Kimura
H. The cholinergic system of the human hindbrain studied by choline
Received November 15, 1995. Revised February 3, 1996.
Accepted February 16, 1996
Downloaded from http://brain.oxfordjournals.org/ at Pennsylvania State University on March 4, 2014
Contreras RJ, Gomez MM, Norgren R. Central origins of cranial
nerve parasympathetic neurons in the rat. J Comp Neurol 1980;
190: 373-94.
Nomura S, Mizuno N. Central distribution of afferent and efferent
components of the chorda tympani in the cat as revealed by the
horseradish peroxidase method. Brain Res 1981; 214: 229-37.