* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download The Study of Brain Activity in Sleep
Human brain wikipedia , lookup
Optogenetics wikipedia , lookup
Emotion and memory wikipedia , lookup
Cognitive neuroscience of music wikipedia , lookup
Time perception wikipedia , lookup
Brain–computer interface wikipedia , lookup
Synaptic gating wikipedia , lookup
Holonomic brain theory wikipedia , lookup
Aging brain wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Functional magnetic resonance imaging wikipedia , lookup
Neuroesthetics wikipedia , lookup
Neuroeconomics wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Memory consolidation wikipedia , lookup
Brain Rules wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Circadian rhythm wikipedia , lookup
Lunar effect wikipedia , lookup
Electroencephalography wikipedia , lookup
Neuroplasticity wikipedia , lookup
Neural oscillation wikipedia , lookup
Effects of blue light technology wikipedia , lookup
Philosophy of experience wikipedia , lookup
Biology of depression wikipedia , lookup
Spike-and-wave wikipedia , lookup
Neuroscience in space wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Delayed sleep phase disorder wikipedia , lookup
Sleep apnea wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Rapid eye movement sleep wikipedia , lookup
Neuroscience of sleep wikipedia , lookup
Sleep paralysis wikipedia , lookup
Sleep and memory wikipedia , lookup
Sleep deprivation wikipedia , lookup
Sleep medicine wikipedia , lookup
Effects of sleep deprivation on cognitive performance wikipedia , lookup
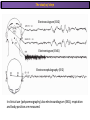
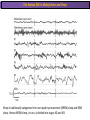
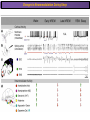
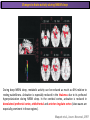
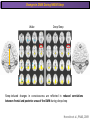
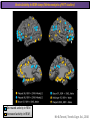
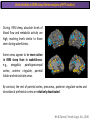
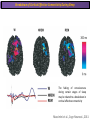
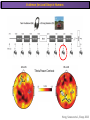
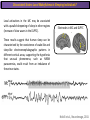
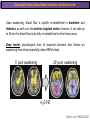
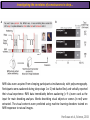
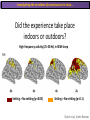
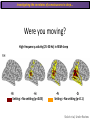
The Study of Brain Activity in Sleep The hours of our life other activities (19%) Sleep 36% work and work-related (16%) sport (1%) television (11%) phone/e-mail (1%) household activities (8%) eat/drink (5%) socializing (3%) By the age of 80, a person will have spent more than 25 years sleeping. American Time Use Survey, Bureau of Labor Statistics. Sleep: A Definition Behavioral Definition: Behavioral state with characteristic immobile posture and diminished but readily reversible sensitivity to external stimuli. Electrophisiological Definition: Electrophyiological Characteristics of Human Sleep The study of sleep Electrooculogram (EOG) Electromiogram (EMG) Electroencephalography (EEG) In clinical use (polysomnography) also electrocardiogram (EKG), respiration and body position are measured. The Human EEG in Wakefulness and Sleep Sleep is traditionally categorized into non-rapid eye movement (NREM) sleep and REM sleep. Human NREM sleep, in turn, is divided into stages N2 and N3. EEG Sleep Stages: Wakefulness Dominated by waves of low amplitude and high frequency (low-voltage fast activity), including beta (16-25 Hz) and gamma (>25 Hz) oscillations. When eyes close in preparation for sleep, EEG alpha activity (8–13 Hz) becomes prominent, particularly in occipital regions (‘ idling’ rhythm in visual areas?). The waking EOG reveals frequent voluntary eye movements and eye blinks. The EMG reveals tonic muscle activity with additional phasic activity related to voluntary movements. EEG Sleep Stages: Stage N1 Characterized by loss of alpha activity and appearance of a low-voltage mixed frequency EEG pattern with prominent theta activity (3–7 Hz). Eye movements become slow and rolling, and muscle tone relaxes. Although there is decreased awareness of sensory stimuli, a subject in stage N1 may deny that he was asleep. Motor activity may persist for a number of seconds during stage N1. Occasionally individuals experience sudden muscle contractions (hypnic jerks), sometimes accompanied by a sense of falling and dream-like imagery. EEG Sleep Stages: Stage N2 Stage N2 is heralded in the EEG by the appearance of K-complexes and sleep spindles, which are especially evident over central regions. K-complexes are made up of a high-amplitude negative sharp wave followed by a positive slow wave, and are often triggered by external stimuli. Sleep spindles are waxing and waning oscillations at around 12–15 Hz that last about 1 second and occur 5–10 times a minute. Eye movements and muscle tone are much reduced. Individuals are partially disconnected from the environment (increased arousal threshold) EEG Sleep Stages: Stage N3 The EEG shows prominent Slow Waves in the delta range (1-4 Hz, 75 μV amplitude in humans). Eye movements cease during stage N3 and EMG activity decreases further. The threshold for arousal is higher than in stage N2. The process of awakening from slow wave sleep is drawn out, and subjects often remain confused for some time (sleep inertia). Sleep Hallmarks: Slow Waves (and K-Complexes) Changes in neuromodulators during NREM sleep (in particular reduced levels of arousal-related neurotransmitters) lead cortical and thalamic neurons to enter in a “bistable” state. In this condition, a spontaneous or induced opening of leakage K+ channels triggers a series of membrane currents that produce the slow oscillation. The slow oscillation is characterized by a hyperpolarization phase or down-state (neuronal silence, 1), which lasts a few hundreds of milliseconds, and a slightly longer depolarization phase or up-state (neuronal spikes of activity, 2). The slow oscillation is synchronized across the cortical mantle by corticocortical and thalamo-cortical connections. Sleep Hallmarks: Slow Waves (and K-Complexes) Topographically, slow waves are especially prominent over medial and lateral prefrontal cortex. K-complexes are high-amplitude slow oscillations usually (but not always) triggered by external stimuli. They are most likely the EEG correlate of global slow oscillations due to the near-synchronous activation of the cortical mantle by the RAS (as opposed to a single cortical source). Sleep Hallmarks: Spindles Spindles are generated in thalamic circuits as a consequence of cortical firing. When the cortex enters an up-state, strong cortical firing excites GABAergic neurons in the reticular nucleus of the thalamus. These in turn strongly inhibit thalamocortical neurons, triggering intrinsic currents that produce a rebound burst of action potentials. These bursts percolate within local thalamoreticular circuits and produce oscillatory firing at around 12–15 Hz. Thalamic spindle sequences reach back to the cortex and are globally synchronized by corticothalamic circuits, where they appear in the EEG as sleep spindles. EEG Sleep Stages: REM After deepening through stages N2 to N3, NREM sleep lightens and returns to stage N2, after which the sleeper enters REM sleep (“paradoxical sleep”: the EEG trace is “similar” to the activated EEG of waking or of stage N1). The EEG is characterized by low-voltage fast-activity, often with increased power in the theta band (3–7 Hz). So called sawtooth waves (2-5 Hz) tipically occur in bursts, especially before the appearance of Rapid Eye Movements (REMs) EEG Sleep Stages: Tonic and Phasic components of REM Tonic aspects of REM sleep include the activated EEG and a generalized loss of muscle tone, except for the extraocular muscles and the diaphragm. REM sleep is also accompanied by penile erections. Phasic features of REM include irregular bursts of REMs and muscle twitches. Physiological Sleep Regulation Changes in Neuromodulation During Sleep Wakefulness promoting structures • Reticular formation: glutamatergic neurons • Locus coeruleus: noradrenaline • Pedunculopontine nucleus and laterodorsal tegmental nucleus: acetylcholine • Raphe neurons: serotonine • Ventral periacqueductal grey: dopamine • Nucleus basalis: acetylcholine • Tuberomammillary neurons: histamine • Posterior hypothalamus: orexin (hypocretin) Ascending reticular activating system • Wakefulness maintained by multiple neuronal systems with different neurotransmitters • Systems are partially redundant, no one system appears to be necessary or sufficient for wakefulness • Widespread projections to cortex, subcortical relays and brainstem or spinal cord, mutually excitatory influences on each other Sleep Cycles Hypnogram The succession of NREM sleep stages followed by an episode of REM sleep is called a sleep cycle, and lasts approximately 90–110 minutes in humans. There are a total of 4– 5 cycles per night. Slow wave sleep (N3) is prominent early in the night, especially during the first sleep cycle, and diminishes as the night progresses. As slow wave sleep wanes, periods of REM sleep lengthen and show greater phasic activity. A healthy young adult typically spends about 5% of the sleep period in stage N1, about 50% in stage N2, 20–25% in stage N3, and 20–25% in REM sleep. A Two Process Model of Sleep Regulaton The two-process model of sleep regulation posits that the interaction of its two constituent processes a homeostatic Process S and a circadian Process C determines the timing of sleep and waking. Sleep Homeostasis (Process S) Wake Circadian Alerting (Process C) Sleep Wake Sleep The time course of Process S is derived from a physiological variable: EEG slowwave activity. Borbély et al., Hum. Neurobiol., 1982 Slow Wave Activity as an index of Sleep Pressure Slow-Wave Activity (SWA) is classically calculated as the mean power spectral density in the 0.54.0 Hz frequency range. The figure shows the SWA profile in NREM sleep during the night for an individual subject (average 1-min values, % of the mean of 4 NREM episodes). Rapid eye movement (REM) episodes are indicated by hatched areas. Early Late Riedner et al., Sleep, 2007 Slow Wave Activity as an index of Sleep Pressure During late NREM sleep, when compared with early NREM sleep, are commonly observed: (1) reduced SWA, (2) fewer large-amplitude slow waves, (3) decreased wave slopes, (4) more frequent multipeak waves Riedner et al., Sleep, 2007 Possible Significance of Slow Wave parameters half-period amplitude - peak-to-peak amplitude + real slow waves are nothing like «theoretical» waves Amplitude: Extent of the neuronal population recruited in the down-state (i.e., small SWs are based on the recruitment of fewer neurons as compared to large SWs). Slope: This parameter may reflect synaptic strength (i.e., steeper slow waves are generated when neuronal populations are rapidly and efficiently synchronized). Negative Peaks: The presence of multiple peaks may indicate a sub-optimal synchronization of distinct neuronal populations (partially asynchronous local waves). Riedner et al., Sleep, 2007 DAY Sleep Intensity is Locally (Homeostatically) Regulated Motor Learning Arm Immobilization NIGHT (SLEEP) High SWA Low SWA Huber et al., 2002, Nature Huber et al., 2006, Nature Neuroscience Sleep intensity is locally regulated on a use-dependent basis Possible Relationship between Sleep and Plasticity Given the association between wake-dependent activities and networkspecific changes in sleep features (in particular, SWA), several authors suggested that sleep may be involved in the regulation of learning and brain plasticity. In particular… The synaptic homeostasis hypothesis suggests that wake activities are associated with a potentiation of synaptic connections, and that sleep may have a role in re-normalizing synapses (down-scaling), allowing for a new learning cycle. The hypothesis of synaptic potentiations suggests that locally regulated sleep-related events may help potentiating (up-scaling) specific connections in order to favor memory consolidation. Changes in Brain Functioning during deep sleep and REM Changes in brain activity during NREM sleep During deep NREM sleep, metabolic activity can be reduced as much as 40% relative to resting wakefulness. Activation is especially reduced in the thalamus due to its profound hyperpolarization during NREM sleep. In the cerebral cortex, activation is reduced in dorsolateral prefrontal cortex, orbitofrontal and anterior cingulate cortex (slow waves are especially prominent in these regions). Maquet et al., Journ. Neurosci, 1997 Changes in DMN During NREM Sleep Wake Deep Sleep Sleep-induced changes in consciousness are reflected in reduced correlations between frontal and posterior areas of the DMN during deep sleep Horovitz et al., PNAS, 2009 Changes in «Resting State» Connectivity During NREM Sleep Wake VS N1 Wake VS N2 Wake < Sleep Wake VS N3 Wake > Sleep The net reduction in connectivity observed during deep sleep reflect a reduction in brain interregional cross talk, i.e., a loss of global integration well in line with a phenotypically concomitant reduction in consciousness. Modularity, a spatial measure of functional segregation, generally increased across the brain with deepening sleep. Tagliazucchi & Laufs, Nueron, 2014 Brain Activity in REM sleep (Meta-analysis of PET studies) Decreased activity in REM Increased activity in REM Nir & Tononi, Trends Cogn. Sci., 2010 Brain Activity in REM sleep (Meta-analysis of PET studies) During REM sleep, absolute levels of blood flow and metabolic activity are high, reaching levels similar to those seen during wakefulness. Some areas appear to be more active in REM sleep than in wakefulness: e.g., amygdala, parahippocampal cortex, anterior cingulate, parietal lobule and extrastriate areas. By contrast, the rest of parietal cortex, precuneus, posterior cingulate cortex and dorsolateral prefrontal cortex are relatively deactivated. Nir & Tononi, Trends Cogn. Sci., 2010 Breakdown of Cortical Effective Connectivity During Sleep The fading of consciousness during certain stages of sleep may be related to a breakdown in cortical effective connectivity Massimini et al., Cogn Neurosci., 2011 Dissociated States Local Sleep and Local Wakefulness Local Sleep during Wakefulness After a long period in an awake state, cortical neurons can go briefly ‘offline’ as in sleep, accompanied by slow waves in the local EEG. Neurons often go offline in one cortical area but not in another [Local Sleep] When local OFF periods happen in areas relevant for behavior (i.e. motor cortex during a reaching task) they lead to performance errors! 8 Vyazovskiy et al., J. Nature., 2011 Evidences for Local Sleep in Humans Theta Power Contrast Hung, Sarasso et al., Sleep, 2013 Local Sleep vs Micro Sleep Local Sleep Spatially circumscribed intrusion of sleep during wakefulness Involves only relatively small areas while most of the brain shows activity patterns typical of wakefulness Micro-Sleep Temporally circumscribed intrusion of (global) sleep during wakefulness Involves most brain regions and may last from a fraction of a second to several seconds Dissociated States: Local Wakefulness in Sleeping Individuals? Local activations in the MC may be associated with a parallel deepening of sleep in other regions (increase of slow waves in the DLPFC). Electrodes in MC and DLPFC These results suggest that human sleep can be characterized by the coexistence of wake-like and sleep-like electroencephalographic patterns in different cortical areas, supporting the hypothesis that unusual phenomena, such as NREM parasomnias, could result from an imbalance of these two states. Nobili et al., NeuroImage, 2011 Dissociated States: Wake-Sleep Transition wake Falling Asleep deep sleep Slow wave density during the falling asleep process Slow wave origin distribution during the falling asleep process Siclari, Bernardi et al., SLEEP, 2014 Dissociated States: Wake-Sleep Transition In the early phase of the falling-asleep period, slow waves typically involve some regions more than others: Slow Wave Cortical Involvement • The primary visual cortex and the medial and lateral parietotemporal areas are less affected by slow waves, and thus “more awake” Conscious experiences are highly visual and frequently characterized by vestibular sensations • The prefrontal regions are more involved by slow waves and thus “more asleep” The dreamer generally lacks insight into the hallucinatory character of the experience RED areas show more slow wave activity at the beginning of the falling asleep period. BLUE areas show more activity when the sleeper enters in deep sleep. Islands of sleep and wakefulness can coexist across distinct brain areas Siclari, Bernardi et al., SLEEP, 2014 Dissociated States: Sleep-Wake Transition and Sleep Inertia Upon awakening, blood flow is rapidly re-established in brainstem and thalamus, as well as in the anterior cingulate cortex. However, it can take up to 20 min for blood flow to be fully re-established in other brain areas. Sleep Inertia: physiological state of impaired alertness that follows an awakening from sleep (especially, deep NREM sleep). 5’ post awakening 20’ post awakening H2O PET Balkin et al. BRAIN 2002 Dissociated States: Arousal Disorders Sleepwalking Confusional Arousal Bassetti et al. Lancet 2005; Terzaghi et al. Sleep 2009, Mahowald and Schenck Nature 2005 Dreams, or Conscious Experiences during different stages of sleep 1950s: Dreaming = REM sleep ‘Tell me whether you had a dream’ • Periods of ocular motility: yes 74% • Periods of ocular inactivity: yes 17% 1960s onwards: NREM dreaming ‘Tell me wheter you had a dream’ ‘What was going through your mind before you woke up ?’ How are conscious experiences distributed in sleep? What was the last thing going through your mind prior to the alarm sound? CE= Conscious Experience CEWR = Conscious Experience Without Recall NCE = No Conscious Experience Siclari et al, Frontiers Psy., 2013 Combined TMS-EEG Studies of Sleep Cognitive State Wakefulness Wake-Sleep Transition NREM sleep REM sleep Type of Experience Daydreaming Hypnagogic hallucination Dreaming Dreaming Freq. Content Typical Features 80% Mainly thoughts. Dreamlike in up to 25%. Independent of external stimuli (by definition). Compared to REM: more abrupt topic changes. 80-90% Short static images (snapshots) or brief sequences of disconnected frames. Sensation of falling. Sometimes influenced by activities performed prior to sleep. Compared to other sleep stages: fewer emotions, fewer characters, less selfrepresentation, less bizarre, closer to reality. Early in the night: thoughtlike and conceptual. Later in the night vivid and hallucinatory experiences. Compared to REM: shorter, less dreamlike, more thought-like, less vivid, more conceptual, under greater volitional control, more plausible, more related to current concerns, less emotional. Late in the night sometimes indistinguishable from REM reports. Vivid, hallucinatory experiences. Compared to wakefulness: singlemindedness, reduced self-awareness, reduced executive control, high degree of emotionality, altered mnemonic processes 23-75% 71-93% Siclari et al, SAoNaP, 2012 Variability in Conscious Experiences during Sleep NREM sleep N2, 6:09 AM I was thinking about perfume and fragrance. The very last word was ‘fragrance’. Siclari et al, Frontiers Psy., 2013 Variability in Conscious Experiences during Sleep NREM sleep N3, 6:09 AM The last thing was raspberries, a pint of raspberries. Siclari et al, Frontiers Psy., 2013 Variability in Conscious Experiences during Sleep REM sleep, 3:28 AM I was doing this experiment with another girl. I asked her what time it was and she said 7:07. No, she actually said 6:55. Her boyfriend was in the room, too. The last scene was just her face. It was quite a long dream before that. Siclari et al, Frontiers Psy., 2013 Investigating the correlates of consciousness in sleep… fMRI data were acquired from sleeping participants simultaneously with polysomnography. Participants were awakened during sleep stage 1 or 2 (red dashed line) and verbally reported their visual experience. fMRI data immediately before awakening (= 9 s) were used as the input for main decoding analyses. Words describing visual objects or scenes (in red) were extracted. The visual contents were predicted using machine learning decoders trained on fMRI responses to natural images. Horikawa et al, Science, 2013 Investigating the correlates of consciousness in sleep… “What I was just looking at was some kind of characters. There was something like a writing paper for composing an essay, and I was looking at the characters from the essay or whatever it was. It was in black and white and the writing paper was the only thing that was there.” Horikawa et al, Science, 2013 Investigating the correlates of consciousness in sleep… Did the experience take place indoors or outdoors? High frequency activity (25-50 Hz) in REM sleep RH Setting > No settting (p<0.05) Setting > No settting (p<0.1) Siclari et al, Under Review Investigating the correlates of consciousness in sleep… Were you moving? High frequency activity (25-50 Hz) in REM sleep RH Setting > No settting (p<0.05) Setting > No settting (p<0.1) Siclari et al, Under Review Investigating the correlates of consciousness in sleep… Was anyone speaking? High frequency activity (25-50 Hz) in REM sleep LH -8s -6s Setting > No settting (p<0.05) -4s -2s Setting > No settting (p<0.1) Siclari et al, Under Review Conclusions Key-Points • Sleep is typically evaluated and classified using EEG (preferably also with EMG and EOG) • Sleep is accompanied by changes in regional brain metabolism detectable using PET and fMRI • Sleep deepening is associated with relevant changes in brain interregional connectivity (reduction) • Several sleep parameters are homeostatically regulated, potentially reflecting/influencing wake-dependent plasticity • Islands of wake and sleep may potentially coexist during either behavioral states (potential explanation for dreams?) Suggested Reading Tononi, G., & Siclari, F. (2015). Sleep and Dreaming. The Neurology of Consciousness 2nd Edition. Academic Press. Scientific American, 89-105. Tononi, G., & Cirelli, C. (2014). Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron, 81(1), 1234. Nir, Y., & Tononi, G. (2010). Dreaming and the brain: from phenomenology to neurophysiology. Trends in Cognitive Sciences, 14(2), 88-100 Siclari, F., Bernardi, G., Riedner, B. A., LaRocque, J. J., Benca, R. M., & Tononi, G. (2014). Two distinct synchronization processes in the transition to sleep: a highdensity electroencephalographic study. SLEEP, 37(10), 1621-1637. Sarasso, S., Pigorini, A., Proserpio, P., Gibbs, S. A., Massimini, M., & Nobili, L. (2014). Fluid boundaries between wake and sleep: experimental evidence from Stereo-EEG recordings. Archives italiennes de biologie, 152(2/3), 169-177.