* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Attention induces synchronization-based response gain in steady
Artificial neural network wikipedia , lookup
Emotional lateralization wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Synaptic gating wikipedia , lookup
Affective neuroscience wikipedia , lookup
Central pattern generator wikipedia , lookup
Neuroeconomics wikipedia , lookup
Convolutional neural network wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Cortical cooling wikipedia , lookup
Recurrent neural network wikipedia , lookup
Emotion perception wikipedia , lookup
Multielectrode array wikipedia , lookup
Microneurography wikipedia , lookup
Functional magnetic resonance imaging wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Types of artificial neural networks wikipedia , lookup
Visual search wikipedia , lookup
Emotion and memory wikipedia , lookup
Neuroethology wikipedia , lookup
Optogenetics wikipedia , lookup
Nervous system network models wikipedia , lookup
Time perception wikipedia , lookup
Perception of infrasound wikipedia , lookup
Neural engineering wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Neural oscillation wikipedia , lookup
Visual extinction wikipedia , lookup
Executive functions wikipedia , lookup
Development of the nervous system wikipedia , lookup
Neural coding wikipedia , lookup
Neuroesthetics wikipedia , lookup
Response priming wikipedia , lookup
Evoked potential wikipedia , lookup
Psychophysics wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Visual spatial attention wikipedia , lookup
Visual selective attention in dementia wikipedia , lookup
C1 and P1 (neuroscience) wikipedia , lookup
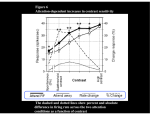
© 2007 Nature Publishing Group http://www.nature.com/natureneuroscience ARTICLES Attention induces synchronization-based response gain in steady-state visual evoked potentials Yee Joon Kim, Marcia Grabowecky, Ken A Paller, Krishnakumar Muthu & Satoru Suzuki When attention is voluntarily directed to a spatial location, visual sensitivity increases at that location. What causes this improved sensitivity? Studies of single neuron spike rates in monkeys have provided mixed results in regard to whether attending to a stimulus increases its effective contrast (contrast gain) or multiplicatively boosts stimulus-driven neural responses (response or activity gain). We monitored frequency-tagged steady-state visual evoked potentials (SSVEPs) in humans and found that voluntary sustained attention multiplicatively increased stimulus-driven population electrophysiological activity. Analyses of intertrial phase coherence showed that this attentional response gain was at least partially due to the increased synchronization of SSVEPs to stimulus flicker. These results suggest that attention operates in a complementary manner at different levels; attention seems to increase single-neuron spike rates in a variety of ways, including contrast, response and activity gains, while also inducing a multiplicative boost on neural population activity via enhanced response synchronization. Perceptual abilities vary immensely as a function of where an individual voluntarily allocates attention. How does attention increase sensitivity to visual stimuli that are presented at an attended location? Three competing hypotheses have been proposed to explain how attention modulates the activation of visual cortical networks. According to the contrast gain hypothesis, the effects of attention are equivalent to increasing stimulus contrast. Thus, this hypothesis predicts that attention should cause a leftward shift in the neural contrast-response function. Neural responses grow with increasing stimulus contrast, following a nonlinear sigmoidal contrast-response function. Therefore, attention should boost neural responses when stimulus contrast is within or below the dynamic range of the neural contrast-response function, but not when stimulus contrast is above the point of response saturation (Fig. 1a). According to two other hypotheses, the response gain and activity gain models, attention multiplicatively increases the responses of the visual neurons that selectively respond to the attended stimulus. Both of these hypotheses predict that attention should boost neural responses most strongly for stimuli with high contrast (Fig. 1b). The response and activity gain hypotheses differ in terms of the effects of attention on spontaneous neural activity. The response gain hypothesis postulates that attention multiplicatively increases only the stimulus-driven component of neural responses, predicting that attention should have no effect on spontaneous neural activity, whereas the activity gain hypothesis postulates that attention multiplicatively boosts the net neural activity, including spontaneous activity (Fig. 1b). To test these hypotheses, stimulus contrast must be varied from a sub-threshold level to a response-saturation level to obtain full contrast-response functions for attended and ignored stimuli. The contrast gain hypothesis predicts that the effects of attention should be largest for moderate-contrast stimuli (in the middle of the dynamic range), whereas the response and activity gain hypotheses predict that attention effects should be largest for high-contrast stimuli, either with (activity gain) or without (response gain) the boosting of spontaneous activity. Numerous electrophysiological and neuroimaging studies have demonstrated that attention increases neural activity for attended, relative to ignored, stimuli. Fixed stimulus contrasts, however, were typically used in these studies, so that the results are equivocal for evaluating the three hypotheses. Only rarely have the effects of attention on contrast-response functions been examined1–9. For single neurons in areas V4 and MT, several studies reported that voluntary visual attention affected spiking activity, primarily on the basis of contrast gain1,10,11; attention modulated the later component of responses (B200–450 ms after stimulus onset), even for highcontrast stimuli1,12, but not as strongly as for low- to moderate-contrast stimuli1. Recent recordings from a large number of V4 neurons, however, found a variety of attention effects on neuronal contrastresponse functions. The attention effect on each neuron was partially consistent with contrast, response or activity gain, yielding a statistical tie across the population, but slightly favoring response and activity gain7. Behavioral results are also mixed with respect to the three hypotheses. Consistent with contrast gain, a recent study4 reported that voluntary attention improved orientation discrimination for lowto moderately high-contrast stimuli, corresponding to the dynamic range of the psychometric function (behavioral performance plotted as a function of contrast). However, attention did not affect performance for high-contrast stimuli, corresponding to the saturated portion of the psychometric function. If attention only operates through mechanisms that induce contrast gain (Fig. 1a), then attention should be generally Department of Psychology and Institute for Neuroscience, Northwestern University, 2029 Sheridan Road, Evanston, Illinois 60208, USA. Correspondence should be addressed to S.S. ([email protected]). Received 5 September; accepted 27 November; published online 17 December 2006; doi:10.1038/nn1821 NATURE NEUROSCIENCE VOLUME 10 [ NUMBER 1 [ JANUARY 2007 117 b Contrast gain Attended Difference between Attended and Ignored © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience 0 0 Ignored Stimulus contrast Response gain Attended Activity gain Ignored 0 Stimulus contrast Difference between Attended and Ignored Neural response a Neural response ARTICLES 0 Stimulus contrast Stimulus contrast Figure 1 The contrast, response and activity gain hypotheses of the way voluntary visual-spatial attention boosts neural responses. (a) According to the contrast gain hypothesis, attention increases the effective contrast of attended stimuli, shifting the contrast-response function to the left. (b) According to the response and activity gain hypotheses, attention multiplicatively increases neural responses to attended stimuli, either with (activity gain) or without (response gain) attention effects on spontaneous neural activity. Upper, hypothetical contrast-response functions for attended and ignored conditions. Lower, difference between attended and ignored contrast-response functions. ineffective for high-contrast stimuli. Contrary to this prediction, robust behavioral attention effects are generally found with high-contrast stimuli, as such stimuli are more readily detected and processed when presented at the focus of attention8,9,13. Furthermore, some behavioral results provide evidence for multiplicative attention effects consistent with response gain (for example, attention improved the discrimination of contrast, orientation and spatial frequency more for a higher-contrast than for a lower-contrast stimulus)8,9 or evidence for the involvement of both contrast gain and response gain5. In sum, for both neuronal spike rates and behavioral performance, the evidence to date has been mixed as to whether voluntary visual attention primarily affects neural activity based on contrast1,3–6,10,11, response5,7–9 or activity7 gain. The three hypotheses have not previously been examined at the level of the neural population. This examination is important for two reasons. First, it is intrinsically difficult to compare the contrastresponse functions of individual neurons with behavioral results because it is unclear how neural signals from different visual areas (or sub-areas) contribute to performing a specific behavioral task14. An examination of neural population responses will identify the primary population activity that is induced by a given stimulus and determine how that population activity is affected by allocation of attention to the stimulus. Second, neural population activity takes on emergent characteristics that are not reflected in the spike rates of individual neurons. Response synchronization is one such example, and there is evidence that attention modulates synchronization. For example, visual attention led to increased 35–90-Hz (gamma-frequency) synchronization among V4 neurons that were responding to an attended stimulus12. For another example, attention to tactile (as opposed to visual) stimuli increased the synchronization among somatosensory neurons15. Because the synchronization can increase the impact of the involved neurons on postsynaptic targets16, these results suggest that response synchronization is one of the mechanisms that are important for attentional selection17,18. Recent evidence suggesting that the coding of stimulus contrast in V1 involves synchronization19 is also consistent with a function for neural response synchronization in modulating the strength of stimulus representation. Because response synchronization is not limited by the saturation of neuronal spike rates at high contrasts19, we hypothesized that attentional modulations of the 118 coherence of neural population responses might provide a mechanism that is especially effective for increasing the salience of high-contrast stimuli. This would complement the attentional modulations of neuronal spike rates, which may be potentially limited by response saturation at high contrast. Our strategy for investigating the effects of attention on the coherence of neural population activity in humans was to monitor SSVEPs that were elicited by flickering stimuli20. These electroencephalographic (EEG) measures can index the increased synchronization of neural responses even if the firing rate of individual neurons is saturated because of high contrast. Hypothetically, if attention induced contrast gain in individual neurons, while also inducing multiplicative response (or activity) gain in terms of increased phase locking of population activity to stimulus flicker, SSVEP amplitudes might be sensitive enough to demonstrate response (or activity) gain. Indeed, some of the data reported in a prior SSVEP study2 are suggestive of multiplicative attention effects on SSVEP amplitudes. Here we rigorously tested the attentional response and activity gain hypotheses for neural population activity by recording frequency-tagged SSVEPs from both attended and ignored stimuli simultaneously (thus controlling for influences on SSVEPs that were unrelated to attention), analyzing the scalp topography of attention effects (crucial for evaluating the attentional response and activity gain hypotheses) and examining the effects of attention on response synchronization. The response and activity gain hypotheses predict that attention directly boosts visual responses that are elicited by the attended stimulus (Fig. 1b). Evidence in support of these hypotheses must demonstrate that there is a multiplicative attention effect on population activity that is stimulus selective. Therefore, we recorded frequency-tagged SSVEPs from numerous scalp locations to identify patterns of activity that were specific to each of the competing stimuli. We then sought to demonstrate that attentional boosts in SSVEP amplitudes occur with a topographic pattern that is indicative of the selective enhancement of sensory activation and that this enhancement is multiplicative (Fig. 1b). The presence or absence of attention effects on subthreshold activity allowed us to distinguish between response and activity gain. Care must be taken to ensure that SSVEP modulations can clearly be attributed to selective attention. This requires that attended and ignored conditions be equated in terms of arousal and the type of visual processes engaged. Accordingly, we presented two flickering circular gratings, one to be attended to and the other to be ignored, in the left and right visual hemifields (Fig. 2a). When the observer attended to one grating, he or she simultaneously ignored the other grating, such that the level of arousal and general task requirements were comparable while we recorded SSVEPs from both the attended and ignored gratings simultaneously21. We made two gratings flicker at different frequencies so that the Fourier band-power value for each frequency could be extracted from the EEG activity to measure coherent neural responses to the respective gratings separately21–23. This frequency-tagging method allowed us to simultaneously monitor SSVEPs from attended and ignored stimuli so that we could determine whether attentional modulation of stimulus selective population electrophysiological activity (recorded as EEG) reflected contrast gain (Fig. 1a), response gain or activity gain (Fig. 1b). We found that (i) each grating elicited SSVEPs that were localized in the contralateral posterior scalp region, (ii) voluntary visual attention selectively and multiplicatively boosted these stimulus-induced SSVEPs in a manner that was consistent with response gain and (iii) this attentional boosting was at least partially attributable to the enhancement of neural response synchronization by attention. VOLUME 10 [ NUMBER 1 [ JANUARY 2007 NATURE NEUROSCIENCE ARTICLES Figure 2 Stimuli and a trial sequence. (a) Two circular gratings were presented in opposite hemifields. Both gratings were flickered (one at 12.50 Hz and the other at 16.67 Hz) between a dark phase and a light phase; for illustration, the dark phase is shown on the left and the light phase is shown on the right. (b) Each trial was initiated by a button press, followed by a central arrow (attention cue) indicating the grating to be attended, a fixation screen and then a 4.8-s presentation of the flickered gratings. a 5.9° © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience 4.47° b Rest (until button press) Attention cue (2 s) Fixation (1 s) Maintain central fixation and attend to cued grating during flickering (4.8 s) RESULTS We elicited SSVEPs by using circular gratings flickering at 12.5 Hz and 16.67 Hz. Two gratings appeared simultaneously in the left and right visual hemifields (Fig. 2a), with flicker frequency randomly assigned to each hemifield on each trial. Each trial lasted 4.8 s, during which the observer maintained fixation at a central marker while voluntarily attending to the grating that was indicated by a central arrow that was presented prior to each trial (Fig. 2b). We contrasted SSVEPs that were derived from the second-harmonic EEG responses (see Methods) for attended and ignored conditions. The SSVEP topographies for the responses to the 16.67-Hz grating (Fig. 3a) showed that the maximal SSVEP amplitudes occurred contralateral to the stimulus over posterior cortical regions and that attention boosted these localized responses. The topography of the Figure 3 Topographic plots of the secondharmonic SSVEPs in response to the two gratings, averaged across observers and stimulus contrasts. (a) 16.67-Hz grating. (b) 12.50-Hz grating. Colorscale data were interpolated based on a fine Cartesian grid. SSVEP amplitudes have been standardized so that the positive and negative values indicate responses above and below the mean level, respectively, in z units. Left, SSVEP topographies when the relevant grating was presented to the right visual field. Right, SSVEP topographies when the relevant grating was presented to the left visual field. Separate rows show conditions in which the relevant grating was attended to (upper), ignored (middle) or the difference between attended and ignored conditions (lower). In all cases, maximal responses and maximal attention effects occurred at contralateral posterior scalp locations. NATURE NEUROSCIENCE VOLUME 10 [ a 12.50-Hz 16.67-Hz grating grating SSVEP differences between the attended and ignored conditions showed that the contralateral posterior focus of attentional enhancement closely resembled the SSVEP topographies for the attended and ignored conditions. This topographic similarity between SSVEP responses and SSVEP enhancement suggests that the attentional modulation occurred in the same brain regions that were selectively activated by the grating. A parallel set of results for SSVEPs that were synchronized to the 12.50-Hz grating (Fig. 3b) showed a similar pattern, except that the SSVEP amplitudes were generally larger. These findings demonstrated that the stimuli elicited focal SSVEPs at contralateral posterior scalp regions and that voluntary spatial attention boosted these localized visual responses. We next evaluated whether this attentional boost of stimulus-evoked population electrophysiological activity was consistent with contrast gain (Fig. 1a), response gain or activity gain (Fig. 1b). To evaluate SSVEP contrast-response functions, we used data from ten scalp locations (five on each side of the scalp; Fig. 4a) that were selected to correspond to the foci of maximal sensory activation and attentional boost. We averaged SSVEPs separately for locations contralateral and ipsilateral to the side of stimulation (combining left and right stimulus presentations). For SSVEPs that were elicited by the 16.67-Hz grating, the contralateral contrast-response functions for the attended and ignored conditions showed that the effect of attention monotonically increased with increasing stimulus contrast, with attention having no effect at the lowest contrasts (Fig. 4b). This finding is consistent with the response gain hypothesis (Fig. 1b). To quantitatively confirm the multiplicative attention effect, we first fit both the attended and ignored contrastresponse functions using the Naka-Rushton equation, which is typically used to fit neural contrast response functions (Fig. 4). If attention multiplicatively boosted visual responses, then the difference between the attended and ignored contrast-response functions should also be well fit by the Naka-Rushton equation1. This was indeed the case (Fig. 4b; F1,7 ¼ 41.582, P o 0.001). Attention had negligible effects 16.67-Hz 12.50-Hz grating grating 16.67-Hz grating ignored 1.0 0.8 0.6 0.4 0.2 0.0 – 0.2 –0.4 –0.6 – 0.8 –1.0 Difference between attended and ignored 0.6 0.4 0.2 0.0 – 0.2 16.67-Hz grating attended NUMBER 1 [ JANUARY 2007 b 16.67-Hz 12.50-Hz grating grating 12.50-Hz grating attended 12.50-Hz grating ignored Difference between attended and ignored 12.50-Hz 16.67-Hz grating grating 1.0 0.8 0.6 0.4 0.2 0.0 – 0.2 –0.4 –0.6 – 0.8 –1.0 0.4 0.2 0.0 – 0.2 119 ARTICLES a Stimulus Contralateral electrodes Ipsilateral electrodes Ipsilateral electrodes Ipsilateral SSVEP 4 1 3 0 2 Attended 1 10 100 1 Ignored 0 1 10 100 1 10 100 Difference between Attended and Ignored 4 3 2 1 Attended 0 1 10 100 1 Ignored 0 1 10 Stimulus contrast (%) 100 1 10 Stimulus contrast (%) 100 on the weak responses recorded from ipsilateral locations, which confirmed the spatial selectivity of the attention effect (Fig. 4b). We found essentially the same pattern of results for SSVEPs that were elicited by the 12.50-Hz grating (Fig. 4c). The attentional boosting of SSVEPs was spatially selective (confined to contralateral locations) and the contrast-response functions indicated response gain; the difference between the attended and ignored contrast-response functions grew monotonically with increasing stimulus contrast, conforming to the Naka-Rushton equation (Fig. 4c; F1,7 ¼ 9.107, P o 0.02), with attention having no effect at the lowest contrasts. In addition to these primary results that consistently supported the idea that the effects of attention on SSVEPs have response gain Figure 5 Representative simulation results indicate that an attentional response or activity gain effect could not emerge from averaging neuronal contrast gain effects over a population of neurons with a variety of contrastresponse functions and contrast gain magnitudes. The average attention effect (the difference between attended and ignored responses) is plotted as a function of contrast for each simulated neural population (N ¼ 1,000), characterized by specific distributions of neuronal contrast-response functions and attentional contrast gain magnitudes. The 48 representative attention effect–versus–contrast curves shown are based on an even sampling of population-parameter statistics from the following ranges. Because neurons in higher visual areas tend to saturate at lower contrasts (see ref. 44 for a review), mean C50 was sampled between 0.43 (reported for V1)31 and 0, and its standard deviation was fixed at 0.31 (reported for V1)31. The mean neuronal attentional contrast gain (the mean percentage reduction in C50 due to attention) was sampled between 0% (no attention effect) and 100% (maximal attention effect), and its standard deviation was sampled between 0% and 200%. The mean and standard deviation for the parameter n were 2.5 and 1.4 (reported for V1)31, and those for b were 0.1 and 0.2; a was set to unity. See text and equation (1) for an explanation of these parameters. To clearly show the shape of each attention-effect curve, each curve has been normalized to its peak value. Though not shown, our simulation also produced peaked attention-effect curves that were consistent with contrast gain when we sampled the population-parameter values from wider ranges of means and standard deviations. 120 characteristics, we also found some frequency dependencies. First, SSVEPs were generally stronger for the 12.50-Hz grating than for the 16.67-Hz grating (compare Figures 3a and b). This could be related to differences in perceived contrast. Although the amplitude of luminance modulation was physically identical for the two flicker frequencies, subjectively the 12.50-Hz grating appeared slightly higher in contrast than the 16.67-Hz grating, probably owing to a higher perceptual sensitivity to the slower flicker. Second, the effects of attention were generally greater for the 16.67-Hz grating than for the 12.50-Hz grating (compare Figures 4b and 4c). Indeed, the magnitude of the attention effects might depend systematically on flicker frequency24. We note that the examined EEG second-harmonic responses (33.33 Hz) were in the gamma range for the 16.67-Hz grating and that some evidence suggests that gamma-range synchronization is closely associated with attention12,25. Third, within the contrast range used, SSVEP contrastresponse functions saturated for the 16.67-Hz grating (Fig. 4b), but not for the 12.50-Hz grating (Fig. 4c). Perhaps the higher-frequency grating activated a higher ratio of magno-type to parvo-type neurons than did the lower-frequency grating. Magno-type neurons may disproportionately contribute to the perception of a faster flicker26 (but see ref. 27,28) and they also tend to saturate at a lower contrast29. These frequency dependencies merit further investigation, and it is unclear how SSVEP results might differ for frequencies that were not tested in this experiment. Nevertheless, our SSVEP results provided strong evidence that is pertinent to the response gain hypothesis. Both the 16.67- and 12.50-Hz gratings elicited electrophysiological responses Simulated population attention effects based on neuronal contrast gain Standardized SSVEP 16.67-Hz grating Contralateral electrodes Difference between Attended and Ignored c Standardized SSVEP 12.50-Hz grating © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience Contralateral SSVEP b Figure 4 Second-harmonic SSVEP contrast-response functions. (a) Illustration of the contralateral and ipsilateral electrodes used for this analysis. (b,c) SSVEP contrast-response functions for the 16.67-Hz grating (b) and the 12.50-Hz grating (c), computed from the 10 scalp electrodes illustrated in a, from which strong stimulus responses and attention effects were obtained (Fig. 3). In b and c, the left panels show contralateral responses and the right panels show ipsilateral responses. Solid curves show contrast-response functions obtained when the relevant grating was attended. Dotted curves show contrast-response functions obtained when the relevant grating was ignored. For the contralateral responses, the insets show the contrast dependencies of the attention effects (the difference between the attended and ignored conditions). The continuous curves show fits based on the Naka-Rushton equation; for the 16.67-Hz grating, r2 for the fits are 0.995 (contralateral attended), 0.991 (contralateral ignored), 0.982 (ipsilateral attended) and 0.956 (ipsilateral ignored); for the 12.50-Hz grating, the corresponding r2 are 0.994, 0.987, 0.975 and 0.992. SSVEP amplitudes have been standardized and averaged across observers (see Fig. 3), and the error bars indicate ± 1 s.e.m. Stimulus 1 VOLUME 10 [ NUMBER 1 10 Stimulus contrast (%) [ 100 JANUARY 2007 NATURE NEUROSCIENCE ARTICLES ITPC for SSVEPs (from contralateral focal electrodes) b 0.40 Attended Ignored Attended Ignored Attended Ignored 0.30 Attended Ignored Time averaged ITPC for SSVEPs (from contralateral focal electrodes) © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience a 0.50 Attended 0.40 0.30 0.20 Ignored 0 16.67-Hz grating 500 1,000 1,500 2,000 2,500 3,000 3,500 4,000 4,500 0.50 Attended function of contrast, C; equation (1))31 and that the parameters a (maximum response relative to baseline), C50 (half-saturation contrast), n (steepness of the contrastresponse function) and b (spontaneous baseline response) were normally distributed across the responding population of neurons. 0.40 0.30 RðCÞ ¼ a Ignored 12.50-Hz grating Cn n C n +C50 +b ð1Þ We considered a wide range of population means and standard deviations for the distributions of these contrast-response parameters, including published values for monkey V1 Figure 6 ITPC for second-harmonic SSVEPs averaged across contralateral focal electrodes, which are (ref. 31) and V4 (ref. 7) neurons. In simulating illustrated in Figure 4a. (a) Time-averaged ITPCs (averaged over the entire trial period) when gratings the effects of attention, we assumed that the were attended versus ignored. ITPCs are shown separately for the two flicker frequencies (16.67-Hz and 12.50-Hz) and for the two highest contrasts (40% and 80%). Attention significantly increased ITPC magnitude of attentional contrast gain (the in all conditions; t7 ¼ 2.758, P o 0.03 and t7 ¼ 2.409, P o 0.05 for 40% and 80% contrasts, reduction in the C50 parameter with attention) respectively, for the 16.67-Hz flickered grating, and t7 ¼ 3.608, P o 0.01 and t7 ¼ 2.878, P o 0.03 was normally distributed across the respondfor 40% and 80% contrasts, respectively, for the 12.50-Hz grating. (b) Time course of ITPCs when ing neurons, and we tested a wide range of gratings were attended versus ignored. ITPCs (averaged across 40% and 80% contrasts) are shown population means and standard deviations for separately for the 16.67-Hz grating (upper) and the 12.50-Hz grating (lower). Time zero corresponds the attention effect (the mean percentage to the grating onset. Note that it takes a few hundred milliseconds for the effects of attention on synchronization to emerge after the grating onset. The data have been averaged across observers and reduction in C50 was varied from 0% (no the error bars shown in a indicate ± 1 s.e.m., with individual variability in baseline ITPC removed. attentional contrast gain) to 100% (maximum attentional contrast gain) with the standard deviations ranging from 0% to 1,000%). For that were localized to the contralateral posterior scalp regions, and each simulated neural population, we computed the average attention attention boosted these focal responses in a manner that was consistent effect (the difference between attended and ignored responses) as a function of contrast. All of our simulated population-averaged attention with the response gain hypothesis. effects peaked at mid-range contrasts (Fig. 5), consistent with contrast gain (Fig. 1a). This indicates that the average population responses from DISCUSSION To understand how the voluntary allocation of spatially focused neurons with a variety of contrast-response functions and attentional attention modulates neural activity, previous research has considered contrast gain magnitudes should still produce an overall contrast gain three hypotheses: contrast gain (the effects of attention being equivalent effect. Thus, it is unlikely that the attentional response gain effects we to increasing stimulus contrast), response gain (attention multiplica- observed on SSVEPs were due to the simple averaging of contrast gain tively boosting stimulus-evoked neural activity) and activity gain effects from individual neurons. (attention multiplicatively boosting the net neural activity). As disAn alternative possibility is that attention increases the phase cohercussed above, to date the evidence at the level of single neurons has ence of neural responses at the population level, in addition to producing been mixed. Here we investigated these hypotheses at the neural contrast gain or a mixture of contrast, response and activity gain at the population level by using flickered stimuli and monitoring the corre- neuronal level. Because SSVEPs are measured as frequency-locked sponding SSVEPs. Voluntary visual attention multiplicatively increased electrophysiological responses to flickered stimuli, increased SSVEPs the synchronized electrophysiological activity, which likely arose from could arise from increased amplitudes or from an increase in the contralateral neocortical regions in response to these stimuli. As coherence of underlying neural responses32,33. One way to evaluate attention did not affect the baseline activity, we have demonstrated whether attention increased neural response synchronization is to attentional response gain at the neural population level. examine intertrial phase coherence (ITPC)34,35. ITPC provides an There are at least two possible ways by which attention could increase amplitude-independent measure of the degree to which stimulus-evoked SSVEPs in accord with response gain, even if attention increased EEG responses are phase-locked to the stimulus volleys. For example, neuronal spike rates predominantly in accord with contrast gain. when the neuronal responses underlying SSVEPs are not synchronized, First, a response gain pattern might emerge for a neural population there will be substantial random variability in the phase lags among when responses are averaged across neurons with a variety of contrast- those responses, producing SSVEPs with large variability in phase from response functions (showing different half-saturation contrasts and one stimulus volley to another. In contrast, when the neuronal responses maximum responses) and various magnitudes of attention-based con- underlying SSVEPs become more synchronized to stimulus dynamics, trast gain. Note that we cannot make any firm predictions about SSVEPs there will be less random variability in the phase lags among on the basis of single-cell spike rate activity because SSVEPs reflect those responses, producing coherent SSVEPs with less variability in aggregate local field potentials, which are more closely associated with phase from one stimulus volley to another. ITPC provides a measure the synaptic activity of a neural population than with spike rates19,30. of this phase consistency in terms of the degree to which EEG responses Nevertheless, we conducted a simulation analysis to show that atten- of a specific frequency are time-locked to stimulus dynamics. tional response gain at the population level cannot emerge from a linear ITPC values vary from 0 (a complete absence of synchronization) to 1 summation of attentional contrast gain on single-cell spike rate activity. (perfect synchronization). Voluntary attention increased the average ITPC in the contralateral We assumed that each neural contrast-response function was reasonably approximated by the Naka-Rushton equation (a neural response, R, as a focal electrodes by B10% for both 16.67-Hz and 12.50-Hz gratings at 0.20 0.20 40 80 40 80 Stimulus contrast (%) Stimulus contrast (%) 16.67-Hz grating 12.50-Hz grating NATURE NEUROSCIENCE VOLUME 10 500 1,000 1,500 2,000 2,500 3,000 3,500 4,000 4,500 Time (ms) Grating onset [ 0 NUMBER 1 [ JANUARY 2007 121 ARTICLES 0.40 Attended 0.30 ITPC for SSVEPs (from contralateral focal electrodes) © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience Ignored 0.20 1 0.40 0.30 10 100 1,000–4,800 ms, attended 1,000–4,800 ms, ignored 400–1,000 ms, attended 400–1,000 ms, ignored 200–400 ms, attended 200–400 ms, ignored 50–200 ms, attended 50–200 ms, ignored 0.20 1 10 100 Stimulus contrast (%) the higher end of the stimulus contrast range (40% and 80%; Fig. 6a). The time course of the ITPC (averaged across 40% and 80% contrasts) shows that the effect of attention emerged within 300–500 ms (Fig. 6b), which is within the temporal range of voluntary attention effects reported for single-cell spike rates1,12,36,37. A previous single-cell study12 reported faster (within 50–150 ms) attentional boosting of gamma-range synchronization (and reduction of low-frequency synchronization) between single-cell spikes and local field potentials in V4. It is possible that it takes longer for synchronization to develop over the large population of neurons that generate SSVEPs. Additionally, the two studies examined different types of neural synchronization; whereas the single-cell study found attentional modulations of local neuronal synchronizations within intrinsic frequencies, we found attentional enhancement of population synchronization to stimulus dynamics. Future research is necessary to clarify the relationship between these two types of synchronization. We also acknowledge the need to be cautious in comparing SSVEP results with single-cell results. Specifically, single-cell studies have examined attention effects when stimuli are presented within the neurons’ classical receptive fields, whereas SSVEPs combine responses from a large population of neurons, including those for which the stimulus falls on the border of, or outside, their classical receptive fields. Future single-cell research, in which attention effects are examined using large stimuli spanning multiple receptive fields, would be necessary to understand how our SSVEP results relate to attention effects on single neurons. An important aspect of our results is that the attentional boosting of ITPC increased with contrast, indicating that attention enhanced population synchronization most strongly for high-contrast stimuli (Fig. 7). This response gain–type attentional enhancement of ITPC occurred throughout the time period of sustained attention (200–400ms, 400–1,000-ms and 1,000–4,800-ms intervals), except for the initial period (50–200 ms), in which no attentional modulation of ITPC occurred (Fig. 7). Overall, converging evidence from our simulation (Fig. 5) and ITPC analyses suggests that the observed attention-based multiplicative boost in SSVEPs is attributable to attentional modula- 122 Figure 7 Contrast dependence of ITPC for second-harmonic SSVEPs (averaged across contralateral focal electrodes, see Fig. 4a) when gratings were attended versus ignored. Upper, time-averaged (50–4,800 ms) ITPC contrast-dependence functions. Lower, ITPC contrast-dependence functions shown separately for different time periods during sustained attention. The data have been averaged for the 16.67-Hz and 12.50-Hz gratings and across observers. The continuous curves indicate fits based on the Naka-Rushton equation; in the upper panel, r2 values for the fits are 0.991 (attended) and 0.987 (ignored) for the 50–4,800-ms period; in the lower panel, r2 are 0.692 (attended) and 0.933 (ignored) for the 50–200-ms period (black), 0.946 (attended) and 0.951 (ignored) for the 200–400-ms period (orange), 0.985 (attended) and 0.967 (ignored) for the 400–1,000-ms period (blue) and 0.994 (attended) and 0.985 (ignored) for the 1000–4,800-ms period (green). Note that, except for the earliest interval that shows no attention effect, ITPC contrast-dependence functions for all of the other periods show a response gain–type profile. tion of neural response synchronization. Further research will be necessary to understand how voluntary attention increases neural response synchronization, but a recent computational study suggests that one possible mechanism might be for attention to reduce the driving current to inhibitory neurons18. Our results might be criticized on the grounds that natural images are not flickered. In fact, rapid luminance modulations frequently occur in both natural and artificial environments—rippling water, motion through a textured environment, rapid saccades, micro saccades that always occur even during voluntary eye fixation38, TV displays and artificial illuminations that generate periodic flicker. Thus, one might argue that flickered stimuli are more ecologically valid23 than the briefly flashed stimuli that are often used in visual attention research. Furthermore, our ITPC analysis has shown that attention increases phase-locking of SSVEPs to rapid changes in luminance, suggesting that attention increases the synchronization of neural population responses to dynamic signals from an attended stimulus. Because retinal stimulation is nearly always dynamic, it is plausible that the synchronization-based multiplicative attention mechanism demonstrated here is also operational in natural situations. To conclude, visual sensitivity is enhanced for stimuli presented at the focus of attention. Many previous studies have investigated the neural mechanisms underlying the beneficial effects of attention on performance. The present findings add to this body of research by implicating separate functions for the effects of attention on neural population activity and neuronal spike rate activity. On a fine spatial scale—that is, when attended and ignored stimuli activate many of the same neurons—attention appears to increase the influence of the attended stimulus relative to the influence of the ignored stimulus in determining neuronal activity. This mechanism has been formalized in the biased-competition model36,39. In this model, attention can affect the relative weighting of signals from attended and ignored stimuli and can mediate attention effects on high-contrast and low-contrast stimuli if both attended and ignored stimuli fall within a single receptive field. For example, when a high-contrast preferred stimulus and a high-contrast null stimulus both fall within a single neural receptive field, attention can boost the neuron’s response by preferentially weighting the signal from the preferred stimulus and can reduce the neuron’s response by preferentially weighting the signal from the null stimulus; the response would be intermediate if both stimuli were ignored. Some suggest that this biased-competition mechanism is closely associated with contrast gain because both selective attention and the relative contrast of competing stimuli similarly modulate the relative influence from within-receptive-field stimuli10,11. A biased competition–contrast gain mechanism can thus VOLUME 10 [ NUMBER 1 [ JANUARY 2007 NATURE NEUROSCIENCE © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience ARTICLES mediate attentional boosts in visual sensitivity a b for low- to moderate-contrast stimuli in general (Fig. 1a) and also for high-contrast stimuli when the competing stimuli are near enough to activate a single receptive field. Note that increasing neuronal activity via response or activity gain would be ineffective for selecting among within-receptive-field stimuli; in that case, input modulation would be necessary to accomplish attentional selection, either via contrast gain or via response (or activity) gain occurring in the previous processing stage in which the competing stimuli fall in Figure 8 Examples of probe displays for the control experiment. (a) A target-present display (the target is separate receptive fields. The biased competition–contrast gain the pair of identically oriented oblique lines at the location of the left grating). (b) A target-absent display (oblique lines are oriented differently within each pair). Vertical distractors were added so that grouping mechanism, however, would be ineffective did not create an easily detectable emergent feature. when attended and ignored stimuli were both high contrast and presented relatively far apart, so as not to fall within a single neuron’s receptive field. For Experimental procedure. The observer initiated each trial by a button press example, in our experiment, the two gratings were presented in (Fig. 2b). A central arrow (attention cue) then appeared for 2 s to indicate opposite visual hemifields. The attended and ignored gratings would which grating (left or right) the observer was to attend to during that trial. have activated separate receptive fields in nearly all of the cortical visual Following a 1-s fixation screen, the two flickering gratings appeared. During the areas throughout the ventral pathway (V1, V2, V4, through the subsequent 4.8-s period, the observer voluntarily attended to the cued grating, inferotemporal cortex) thought to be closely associated with object while maintaining eye fixation at the central fixation marker and attempting to perception and pattern visibility40–42. Even in the inferotemporal withhold blinks. We directly manipulated attention by instructing the observers to sustain cortex, where visual neurons have very large receptive fields, neurons attention to the cued grating and ignore the other grating. We chose this respond primarily to contralateral stimuli regardless of the direction of method, rather than indirectly manipulating attention by using a visual task, so attention37. In such circumstances, our results implicate a synchroniza- that SSVEP modulations could be attributable to voluntary spatial attention tion-based attention mechanism that multiplicatively enhances popu- without potential task-related confounding factors. Behavioral4,13,43,44 as well as lation neural activity. fMRI45 results have provided evidence that human observers can reliably In summary, a biased competition–contrast gain mechanism can allocate attention when instructed to do so. The fact that we obtained modulate neuronal spike rates for both low- to moderate-contrast substantial attentional boosting of stimulus-selective contralateral SSVEPs stimuli and spatially proximate stimuli (regardless of contrast). Sub- provides evidence that our observers successfully allocated attention to the populations of neurons showing response and activity gain properties cued grating. To verify that our manipulation of attention produced behavioral effects that may extend attention effects across a broader range of contrasts7. were independent of grating contrast, we conducted the following control However, an additional synchronization-based response gain mechanexperiment. The trials were identical to those in the SSVEP experiment ism, operating at the neural population level, allows robust attentional (Fig. 2b), except that we presented a cue instructing the observer to attend selection of the high-contrast stimuli typically encountered in everyday to both gratings on half of the trials and we measured the effects of attention life even when attended and ignored stimuli are too far apart to be using a probe display that was flashed for 100 ms. We presented the probe detected by the same neuron’s receptive field. Both neuronal and display (Fig. 8) 500 ms after the grating onset, as the time course of ITPC synchronization-based mechanisms of attention may operate concur- (Fig. 6b) indicated that observers fully allocated their attention by 500 ms. The rently to fulfill the demands of attentional regulation in diverse target in the probe display was defined by a pair of oblique lines, identical in orientation, that occurred at the location of one of the gratings. The target environmental circumstances. METHODS Observers. Eight observers with normal or corrected-to-normal vision participated as paid volunteers after giving informed written consent. The group included five men and three women. Their ages ranged from 23 to 45 years. Stimuli. We displayed stimuli on a 19-inch CRT monitor set to a 100-Hz refresh rate. We presented two identical square-wave modulated circular gratings (1.1 cycles per degree in fundamental spatial frequency) to the left and right visual hemifields (Fig. 2a). We always flickered the two gratings at different frequencies (12.50 Hz and 16.67 Hz, with assignment to visual hemifield randomized across trials). We induced flicker by modulating the luminance of the concentric rings symmetrically (darker and lighter) against the mid-gray (64.7 cd m–2) background, which prevented the creation of negative afterimages. Because visual neurons are primarily driven by luminance changes, we define the contrast, C, of the flickered gratings as, C ¼ (Llight – Ldark) (Llight + Ldark)–1, where Llight and Ldark indicate the luminance during the light and dark phases, respectively. NATURE NEUROSCIENCE VOLUME 10 [ NUMBER 1 [ JANUARY 2007 always occurred at the location of the attended grating when the observer attended to only one grating (cue validity was 100%). Because we presented the targets among feature-matched distractors, they did not attract attention, nor were they easily detectable when the observer distributed attention across the display46. Thus, the experiment provided a sensitive measure of attention allocation, where successfully focused attention should have resulted in faster and more accurate target detection at the location of the cued grating. We adjusted the contrast of the probe display for each grating contrast and for each observer so that the target-detection performance in the distributed-attention condition was equivalent for all of the grating contrasts. We tested three of the eight observers who participated in the SSVEP experiment, using 5%, 20% and 80% randomly intermixed grating contrasts, with 384 trials per observer. All of the observers produced both faster response time and greater accuracy in the focused-attention condition (while attending to only one grating) than in the distributed-attention condition (while attending to both gratings) for all of the grating contrasts. Average response times were: 769 ms (focused) versus 982 ms (distributed) for the 5% contrast grating (a 213-ms advantage), 831 ms versus 950 ms for the 20% contrast grating (a 119-ms advantage) and 781 ms versus 972 ms for the 80% contrast grating (a 191-ms advantage). Average accuracies were: 85% (focused) versus 73% (distributed) for the 5% contrast grating 123 © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience ARTICLES (a 12% advantage), 85% versus 74% for the 20% contrast grating (an 11% advantage) and 89% versus 73% for the 80% contrast grating (a 16% advantage). Thus, these results verified that our instructional manipulation of attention produced similar behavioral effects at different grating contrast levels, suggesting that our observers allocated a comparable amount of attention to gratings of different contrasts. In the SSVEP experiment, there were 32 trial types: 2 directions of attention (left or right), 2 assignments of flicker frequencies (12.50 Hz on the left and 16.67 Hz on the right, and vice versa) and 8 contrast levels (0.00625, 0.0125, 0.025, 0.05, 0.10, 0.20, 0.40, or 0.80). We ran each trial type 20 times while randomly varying and counterbalancing the 3 factors across trials. We tested each observer for a total of 640 trials in blocks of 160 trials. We initially ran several practice trials and also gave breaks as necessary. Data recording and analysis. We recorded EEG activity using tin electrodes embedded in an elastic cap at locations distributed relatively evenly across the scalp. For 59 EEG channels, the right mastoid served as the reference. We used four additional channels for monitoring vertical and horizontal electro-oculographic (EOG) activity. We lowered electrode impedances to 5 kO, amplified signals with a bandpass of 0.3–200 Hz and digitized them at 1,000 Hz. We rejected individual trials from further analysis on the basis of blink or muscle-activity artifacts detected by vertical EOG activity. In addition, to retain only the trials in which central eye fixation was maintained, we recursively rejected trials with the highest horizontal EOG activity until the average horizontal EOG activity for each condition (defined by each combination of attention allocation, flicker-frequency assignment, and contrast) for each observer was less than 5 mV during the entire 4.8-s trial period. This criterion approximately corresponds to maintained central fixation within a visual angle22,47 of 0.51. Following these artifact-rejection procedures, we retained a mean of 89% of the trials. To exclude the initial transient response to the grating onset, we analyzed EEG waveforms recorded from 526 ms to 4,621 ms after grating onset (except for the time course analysis of ITPC which included the entire trial period). This yielded 4,096 (212) data points per trial. Reducing the number of EEG data points from each trial to a power of 2 is optimal for fast Fourier transform (FFT) analysis. We averaged EEG waveforms from the 59 scalp electrodes separately for each condition and for each observer and then we re-referenced the average waveforms to the average of left and right mastoid recordings. To extract SSVEP activity that was synchronized to the stimulus flicker, we subjected each re-referenced average waveform (corresponding to a specific condition) from each scalp electrode to an FFT. We then computed the SSVEP amplitude as the Fourier band-power within the range of 0.976 Hz centered at the second harmonic of the stimulus flicker frequency. Because the luminance flicker we used was symmetric about the mid-gray background (Fig. 2), the fundamental-frequency responses (for example, first harmonic) from the non– frequency doubling cortical simple cells would have mostly averaged out in SSVEPs due to the random spatial distribution of the ON and OFF sub-regions of their receptive fields with respect to the flickered stimuli2,48–50. Any remaining power at the fundamental frequencies would then be due to overall unequal activation of the ON and OFF sub-regions, unbalanced amplitudes of ON and OFF responses or both. The effects of attention on such residual responses would be difficult to interpret. In contrast, frequency-doubling responses from cortical complex cells50 are largely spatial-phase invariant and they contribute robustly to SSVEPs48,49. Because the absolute values of EEG signals vary widely from observer to observer, we standardized the data from the eight observers before combining them. Specifically, we z-transformed the SSVEP amplitude from each electrode, in each condition and for each observer based on the observer’s overall average and standard deviation of SSVEP amplitudes across all scalp electrodes and across all of the conditions. ACKNOWLEDGMENTS This work was supported by a US National Institutes of Health Grant EY14110 to S.S. and by National Institutes of Health Grant NS34639 and US National Science Foundation Grant BCS0518800 to K.A.P. 124 COMPETING INTERESTS STATEMENT The authors declare that they have no competing financial interests. Published online at http://www.nature.com/natureneuroscience Reprints and permissions information is available online at http://npg.nature.com/ reprintsandpermissions 1. Reynolds, J.H., Pasternak, T. & Desimone, R. Attention increases sensitivity of V4 neurons. Neuron 26, 703–714 (2000). 2. Di Russo, F., Spinelli, D. & Morrone, M.C. Automatic gain control contrast mechanisms are modulated by attention in humans: evidence from visual evoked potentials. Vision Res. 41, 2435–2447 (2001). 3. Cameron, E.L., Tai, J.C. & Carrasco, M. Covert attention affects the psychometric function of contrast sensitivity. Vision Res. 42, 949–967 (2002). 4. Ling, S. & Carrasco, M. Sustained and transient covert attention enhance the signal via different contrast response functions. Vision Res. 46, 1210–1220 (2006). 5. Huang, L. & Dobkins, K.R. Attentional effects on contrast discrimination in humans: evidence for both contrast gain and response gain. Vision Res. 45, 1201–1212 (2005). 6. Carrasco, M., Ling, S. & Read, S. Attention alters appearance. Nat. Neurosci. 7, 308–313 (2004). 7. Williford, T. & Maunsell, J.H. Effects of spatial attention on contrast response functions in macaque area V4. J. Neurophysiol. 96, 40–54 (2006). 8. Morrone, M.C., Denti, V. & Spinelli, D. Color and luminance contrasts attract independent attention. Curr. Biol. 12, 1134–1137 (2002). 9. Lee, D.K., Itti, L., Koch, C. & Braun, J. Attention activates winner-take-all competition among visual filters. Nat. Neurosci. 2, 375–381 (1999). 10. Reynolds, J.H. & Chelazzi, L. Attentional modulation of visual processing. Annu. Rev. Neurosci. 27, 611–647 (2004). 11. Martinez-Trujillo, J. & Treue, S. Attentional modulation strength in cortical area MT depends on stimulus contrast. Neuron 35, 365–370 (2002). 12. Fries, P., Reynolds, J.H., Rorie, A.E. & Desimone, R. Modulation of oscillatory neuronal synchronization by selective visual attention. Science 291, 1560–1563 (2001). 13. Posner, M.I., Snyder, C.R. & Davidson, B.J. Attention and the detection of signals. J. Exp. Psychol. 109, 160–174 (1980). 14. Maunsell, J.H. & Cook, E.P. The role of attention in visual processing. Phil. Trans. R. Soc. Lond. B 357, 1063–1072 (2002). 15. Steinmetz, P.N. et al. Attention modulates synchronized neuronal firing in primate somatosensory cortex. Nature 404, 187–190 (2000). 16. Azouz, R. & Gray, C.M. Dynamic spike threshold reveals a mechanism for synaptic coincidence detection in cortical neurons in vivo. Proc. Natl. Acad. Sci. USA 97, 8110–8115 (2000). 17. Niebur, E. & Koch, C. A model for the neuronal implementation of selective visual attention based on temporal correlation among neurons. J. Comput. Neurosci. 1, 141–158 (1994). 18. Buia, C. & Tiesinga, P. Attentional modulation of firing rate and synchrony in a model cortical network. J. Comput. Neurosci. 20, 247–264 (2006). 19. Henrie, J.A. & Shapley, R. LFP power spectra in V1 cortex: the graded effect of stimulus contrast. J. Neurophysiol. 94, 479–490 (2005). 20. Regan, D. Human Brain Electrophysiology: Evoked Potentials and Evoked Magnetic Fields in Science and Medicine (Elsevier, New York, 1989). 21. Morgan, S.T., Hansen, J.C. & Hillyard, S.A. Selective attention to stimulus location modulates the steady-state visual evoked potential. Proc. Natl. Acad. Sci. USA 93, 4770–4774 (1996). 22. Müller, M.M. et al. Effects of spatial selective attention on the steady-state visual evoked potential in the 20–28 Hz range. Brain Res. Cogn. Brain Res. 6, 249–261 (1998). 23. Müller, M.M., Malinowski, P., Gruber, T. & Hillyard, S.A. Sustained division of the attentional spotlight. Nature 424, 309–312 (2003). 24. Ding, J., Sperling, G. & Srinivasan, R. Attentional modulation of SSVEP power depends on the network tagged by the flicker frequency. Cereb. Cortex 16, 1016–1029 (2005). 25. Fell, J., Fernandez, G., Klaver, P., Elger, C.E. & Fries, P. Is synchronized neuronal gamma activity relevant for selective attention? Brain Res. Brain Res. Rev. 42, 265–272 (2003). 26. Schiller, P.H., Logothetis, N.K. & Charles, E.R. Role of the color-opponent and broadband channels in vision. Vis. Neurosci. 5, 321–346 (1990). 27. Levitt, J.B., Kiper, D.C. & Movshon, J.A. Receptive fields and functional architecture of macaque V2. J. Neurophysiol. 71, 2517–2542 (1994). 28. Hawken, M.J., Shapley, R.M. & Grosof, D.H. Temporal-frequency selectivity in monkey visual cortex. Vis. Neurosci. 13, 477–492 (1996). 29. Edwards, D.P., Purpura, K.P. & Kaplan, E. Contrast sensitivity and spatial frequency response of primate cortical neurons in and around the cytochrome oxidase blobs. Vision Res. 35, 1501–1523 (1995). 30. Varela, F., Lachaux, J.P., Rodriguez, E. & Martinerie, J. The brainweb: phase synchronization and large-scale integration. Nat. Rev. Neurosci. 2, 229–239 (2001). 31. Geisler, W.S. & Albrecht, D.G. Visual cortex neurons in monkeys and cats: detection, discrimination, and identification. Vis. Neurosci. 14, 897–919 (1997). 32. Rager, G. & Singer, W. The response of cat visual cortex to flicker stimuli of variable frequency. Eur. J. Neurosci. 10, 1856–1877 (1998). VOLUME 10 [ NUMBER 1 [ JANUARY 2007 NATURE NEUROSCIENCE © 2007 Nature Publishing Group http://www.nature.com/natureneuroscience ARTICLES 33. Srinivasan, R., Russell, D.P., Edelman, G.M. & Tononi, G. Increased synchronization of neuromagnetic responses during conscious perception. J. Neurosci. 19, 5435–5448 (1999). 34. Tallon-Baudry, C., Bertrand, O., Delpuech, C. & Pernier, J. Stimulus specificity of phase-locked and non-phase-locked 40 Hz visual responses in human. J. Neurosci. 16, 4240–4249 (1996). 35. Delorme, A. & Makeig, S. EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J. Neurosci. Methods 134, 9–21 (2004). 36. Reynolds, J.H., Chelazzi, L. & Desimone, R. Competitive mechanisms subserve attention in macaque areas V2 and V4. J. Neurosci. 19, 1736–1753 (1999). 37. Chelazzi, L., Duncan, J., Miller, E.K. & Desimone, R. Responses of neurons in inferior temporal cortex during memory-guided visual search. J. Neurophysiol. 80, 2918–2940 (1998). 38. Martinez-Conde, S., Macknik, S.L. & Hubel, D.H. The role of fixational eye movements in visual perception. Nat. Rev. Neurosci. 5, 229–240 (2004). 39. Kastner, S. & Ungerleider, L.G. The neural basis of biased competition in human visual cortex. Neuropsychologia 39, 1263–1276 (2001). 40. Mishkin, M., Ungerleider, L.G. & Macko, K.A. Object vision and spatial vision: two central pathways. Trends Neurosci. 6, 414–417 (1983). 41. Leopold, D.A. & Logothetis, N.K. Multistable phenomena: changing views in perception. Trends Cogn. Sci. 3, 254–264 (1999). NATURE NEUROSCIENCE VOLUME 10 [ NUMBER 1 [ JANUARY 2007 42. Grill-Spector, K. & Malach, R. The human visual cortex. Annu. Rev. Neurosci. 27, 649– 677 (2004). 43. Sperling, G. & Melchner, M.J. The attention operating characteristic: examples from visual search. Science 202, 315–318 (1978). 44. Suzuki, S. Attention-dependent brief adaptation to contour orientation: a high-level aftereffect for convexity? Vision Res. 41, 3883–3902 (2001). 45. O’Craven, K.M., Rosen, B.R., Kwong, K.K., Treisman, A. & Savoy, R.L. Voluntary attention modulates fMRI activity in human MT-MST. Neuron 18, 591–598 (1997). 46. Treisman, A. & Sato, S. Conjunction search revisited. J. Exp. Psychol. Hum. Percept. Perform. 16, 459–478 (1990). 47. Luck, S.J. et al. Effects of spatial cuing on luminance detectability: psychophysical and electrophysiological evidence for early selection. J. Exp. Psychol. Hum. Percept. Perform. 20, 887–904 (1994). 48. Campbell, F.W. & Maffei, L. Electrophysiological evidence for the existence of orientation and size detectors in the human visual system. J. Physiol. (Lond.) 207, 635–652 (1970). 49. Hou, C., Pettet, M.W., Sampath, V., Candy, T.R. & Norcia, A.M. Development of the spatial organization and dynamics of lateral interactions in the human visual system. J. Neurosci. 23, 8630–8640 (2003). 50. De Valois, R.L., Albrecht, D.G. & Thorell, L.G. Spatial frequency selectivity of cells in macaque visual cortex. Vision Res. 22, 545–559 (1982). 125