* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Neurotransmission in the rat amygdala related to fear and anxiety
Neuromuscular junction wikipedia , lookup
Mirror neuron wikipedia , lookup
Affective neuroscience wikipedia , lookup
Neuroplasticity wikipedia , lookup
Long-term potentiation wikipedia , lookup
Aging brain wikipedia , lookup
Metastability in the brain wikipedia , lookup
Biological neuron model wikipedia , lookup
Neural coding wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Long-term depression wikipedia , lookup
Neurotransmitter wikipedia , lookup
Synaptogenesis wikipedia , lookup
NMDA receptor wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Central pattern generator wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Neuroanatomy wikipedia , lookup
Nervous system network models wikipedia , lookup
Traumatic memories wikipedia , lookup
Spike-and-wave wikipedia , lookup
Nonsynaptic plasticity wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Neuroanatomy of memory wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Circumventricular organs wikipedia , lookup
Chemical synapse wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Emotional lateralization wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Hypothalamus wikipedia , lookup
Optogenetics wikipedia , lookup
Limbic system wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Neurotransmission in the rat amygdala related to fear and
anxiety
Michael Davis, Don Rainnie and Martin Cassell
Michael Davis is at
the Dept of
Psychiatry,
Yale University,
Connecticut Mental
Health Center,
34 Park Street New
Haven, CT0650B,
USA, Don Rainnie is
atthe Dept of
Psychiatry, Harvard
University,
Brockton Veterans
AdmimstratlOn
Medical Center,
Brockton, MA 02401,
USA, and Martin
Cassell IS at the Dept
of Anatomy,
University of 10 wa,
Iowa City, IA 52242,
USA.
An impressive amount of evidence from many different
laboratories using a variety of experimental techniques
indicates that the amygdala Plays a crucial role in the
acquisition, consolidation and retention or expression of
conditioned fear. E lectroPhysiological data are beginning to detail the transmitters and inter-amygdala
connections that transmit information to, within, and
out of the amygdala. In general, treatments that
increase the excitability of amygdala output neurons in
the basolateral nucleus (for examPle, by decreasing
oPiate and GABA transmission, and increasing noradrenergic transmission) improve aversive conditioning,
whereas treatments that decrease excitability of these
neurons (by increasing oPiate and GABA transmission,
and decreasing NMDA and noradrenergic transmission) retard aversive conditioning as well as producing anxiolytic effects in appropriate animal tests. A
better understanding of brain systems that inhibit the
amygdala, as well as the role of its very high levels of
pep tides, might eventually lead to the development of
more effective Pharmacological strategies for treating
clinical anxiety and memory disorders.
Despite its name, the rat amygdala bears only a
passing resemblance to the 'almond' that Karl Burdach
saw on dissections of the human brain nearly 200
208
©
1994. Elsevier SCience Ltd
years ago. Nonetheless,
it contains almost all the
nuclear groups present in the primate amygdala, its
extrinsic and intrinsic connections and neurochemistry
are also remarkably similar, and the evidence for its
role in complex behavior, learning and memory is
generally consistent with the functions attributed to
the human and non-human primate amygdala. In the
past decade, the development of the in vitro slice
preparation and the availability of a wide range of
neurotransmitter
agonists and antagonists, as well as
the availability of detailed anatomical studies on the
intrinsic microcircuitry of individual amygdaloid nuclei,
have led to a considerable increase in the understanding of the synaptic events underlying some of the
behaviors mediated by the rat amygdala. This review
will integrate the current knowledge of synaptic
transmission within the rat amygdala with the effects
of local infusion of drugs into the amygdala on a
specific form of aversive learning: fear conditioning.
Emphasis will be placed on the role of GAB A and
glutamate, because the actions of these neurotransmitters in the amygdala have been strongly implicated
in fear conditioning.
A large and consistent literature indicates that the
amygdala is critically involved in the acquisition and
expression of conditioned fear (for review see Ref. 1).
0166 - 2236/94/$07.00
TINS, Vol. 17, No.5, 1994
Electrical stimulation of the amygdala produces a
pattern of behavioral changes that closely resembles
that produced by stressful or fearful stimuli, and
lesions of the amygdala block innate or conditioned
reactions to stress. Aversive stimuli readily activate
the amygdala, and results obtained after local infusion
of compounds into the amygdala indicate that it is
especially important in the formation, consolidation
and expression of memories of events paired with
these aversive stimuli. A better understanding of the
chemical neuroanatomy
of synaptic transmission
within the amygdala will eventually lead to novel and
superior stratebries for treating clinical anxiety disorders such as post-traumatic stress syndrome and
panic, as well as having general relevance to the study
of learning and memory. However, it should be
emphasized that restricting this review to fear conditioning does not mean that this is the only function of
the amygdala. On the contrary, this complex structure is also involved critically in attention, secondary
reinforcement, reward magnitude and social behavior
(for review see Ref. 2).
B
Intrinsic and extrinsic connections of the
amygdala
Information from all sensory modalities reaches the
amygdala via projections from the cortex and a variety
of subcortical structures (most notably the thalamus
and parabrachial complex) that converge on the
basolateral amygdaloid complex, in particular the
lateral nucleus'. The cortical projections, which arise
from secondary and polymodal association cortices 1,
probably relay cognitive, but otherwise affectively
neutral, information pertaining to sensory stimuli.
Information concerning the aversive properties of
stimuli are probably relayed separately via projections
from the external lateral part of the para brachial
complex-', the dysgranular insular cortex1i, and midline
and intralaminar thalamic nuclei', each of which
receives nociceptive inputs and projects to the
basolateral complex. Direct projections from the
para brachial nucleus and insular cortex to the central
nucleus might also be especially important for relaying
aversive information to the amygdala:>.
The basolateral complex (consisting of the lateral,
basolateral and basomedial nuclei) contains two basic
types of neuronsx:
large, spine-dense
pyramidal
neurons containing glutamate, and providing the major
extrinsic projections of the complex; and spine-sparse
non-pyramidal cells that contain GABA, choline
acetyltransferase
(CAT) and neuropeptides~J,
and
most of which are presumed to be local circuit
neurons, although some also project to the thalamus.
Intracellular
electrophysiolobrical
recordings
from
morphologically identified neurons of the basolateral
complex suggest that the spine-dense and spinesparse neurons (Fig. 1) have characteristic firing
properties and membrane input resistance (Table lJ.
Importantly, the high spine density of projection
neurons in the basolateral complex suggests that they
are capable of integrating a vast array of synaptic
contacts from extrinsic afferent inputs.
There are four main output projections from the
basolateral complex that arise from the pyramidal
neurons of the basolateral nucleus: reciprocal projections back to the cortex including the frontal
cortex. and unidirectional projections to ventral
TINS, Vol. 17, No.5, 1994
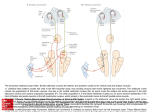
Fig. 1. (A) Photomicrograph of a class I pyramidal neuron
of the basolateral nucleus of the amygdala (BLA). Class I
neurons comprise >75% of the neurons of the BLA, and
are characterized by the spine-laden appearance of the
dendrites. This class I neuron has a prominent apical
dendrite and pyriform cell soma, and was filled with 2%
biocytin during electrophysiological recording, and subsequently visualized with a peroxidase-antiperoxidase
complex using diaminobenzedine as the chromogen.
(8) Photomicrograph of a non-spiny neuron. Scale bars,
100 11m.
caudate~putamen,
the nucleus accumbens, and the
central amygdaloid nucleus 111. The reciprocal cortical
projections might be involved in the conscious perception of fear or anxiety, but this awaits verification in
humans. Projections to the two striatal areas might
relay motivationally significant information to motor
areas necessary for the avoidance of harmful stimuli
or approach to stimuli associated with primary reinforcers. Projections to the central nucleus of the
amygdala, the major intra-amygdaloid target of the
basolateral complex, are critical for autonomic and
somatic responses produced by stimuli that were
previously paired with aversive events.
The central nucleus is organized in a manner similar
to both the dorsal and ventral striatopallidal systems 11. The lateral central nucleus contains mostly
medium sized, spine-dense neurons, many of which
have GABA as their neurotransmitterl~
and contain a
variety of neuropeptides u. Further similarities with
the caudate-putamen
and nucleus accumbens include
the heavy dopaminergic innervation of the lateral
central nucleus 11, direct cortical input to GABA
neunms ]C" and the projections of these GABA
neurons onto the large, spine-sparse output neurons
209
TABLE I. Mean
subtypes
membrane
of amygdaloid
input
resistance
for
several
neurons
Excitatory amino acid transmission
Electrophysiology. Intra- and extracellular recordings
from anesthetized rats have demonstrated both EPSPs
and IPSPs in the amygdala!~, and have confirmed that
Cortex-like
individual amygdaloid neurons can receive and inte5SMO
40-60 MO
Basolateral
grate synaptic inputs from diverse sources20. Intra90MO
SOMO
Lateral
cellular recordings from brain slice preparations have
shown that stimulation of afferent pathways to the
Noncortex-like
111 MO
lateral and basolateral amygdala elicit EPSPs. Stimu113 MO
Central
lation of the stria terminalis, the lateral amygdala, the
Cortex-like neurons have the lowest resistance and can be further
external capsule, or the ventral endopyriform nucleus
subdivided into spine-laden putative projection neurons and
evokes a glutamate-mediated EPSP in the basolateral
spine-sparse putative interneurons. Neurons of the lateral nucleus
generally have a higher input resistance than the neighboring
amygdala:2!-n. These EPSPs consist of a fast combasolateral nucleus indicating the possibility of greater spatial and
ponent mediated by D, L-rx-amino-3-hydroxy-5-methyl-4temporal summation of synaptic inputs onto these neurons.
isoxazole propionic acid (AMP A)lkainate receptors
Neurons of the non-cortex-like central nucleus have the highest
(that is, the fast component is blocked by the
input resistance, suggesting an electronically more compact
antagonist 6-cyano-2, 3-dihydroxy-7 -nitroquinoxaline,
neuron. These neurons are both spine-laden and spine-sparse.
However, they have been tentatively subdivided according to the
CNQX), and a slower component mediated by NMDA
presence (Type A) or absence (Type B) of an afterhyperpolanzreceptors (that is, blocked by the antagonist 2-aminoation following repetitive burst firing.
phosphonopentanoic
acid:2!, APV). Similarly, focal
stimulation of the basolateral amygdala evokes a
glutamate-mediated EPSP in the lateral nucleus:21.2C,.
of the medial central nucleus!5. These medial central
nucleus neurons contain a variety of neuropeptides Hi Thus, most afferents from the projection neurons of
and, in some cases, glutamate!!, and project to a the lateral and basolateral nuclei to the central nucleus
variety of brainstem regions capable of influencing or are glutamatergic, and stimulation of the basolateral
initiating autonomic and somatic components of the amygdala evokes an EPSP in the central nucleus
which has both AMPA/kainate and NMDA receptorfear reaction (Fig. 2). Electrophysiological
studies
mediated components:2li.
suggest that the principal neurons of the two central
Behavior. It has been suggested that fear conditionnucleus subdivisions have high input resistances
ing might be mediated by associative long-term
(Table 1) and can be further differentiated according
potentiation2!-2~
(LTP). In pavlovian fear conditionto the presence (type A, medial neurons) or absence
ing, a neutral stimulus, which has little behavioral
(type B, lateral neurons) of a slow after hyperpolarizeffect by itself, is consistently paired with a strong
ing potential following repetitive firing!K.
aversive stimulus. Following a small number of
The basic intrinsic amygdaloid circuitry potentially
pairings, the neutral stimulus produces effects forinvolved in conditioned associations between neutral
and aver'sive stimuli, based on both anatomical and merly only produced by the strong, aversive stimulus.
electrophysiological recording studies, is shown in This change is not seen when the stimuli are
presented in an unpaired fashion. In associative LTP,
Fig. 3. For simplification, a number of other intrinsic
activation of a weak input to a given postsynaptic cell
and other extrinsic connections, principally subcortical
is paired with activation of a second, strong input
ones, are not included.
Spine-dense
Spine-sparse
Anatomical
Lateral hypothalamus
Conditioned
fear stimulus
\
Sympathetic
activation
----
Dorsal motor n. of vagus
Nucleus ambiguus
Parabrachial
•.
nucleus ----
Ventral tegmental area
Locus coeruleus
----.
Dorsal lateral tegmental n.
Pontine reticular formation
--
Central grey
Unconditioned
fear stimulus
Trigeminal,
•
facial motor n.
Paraventricular
Behavioral test or sign
of fear or anxiety
Effect of amygdala
stimulation
target
n. (hypothal.) ---
Tachycardia, galvanic skin
response, paleness,
pupil dilation,
blood pressure elevation
_
Parasympathetic
activation
._
Ulcers, urination,
defecation, bradycardia
Increased respiration ~
Panting, respiratory distress
Activation of dopamine,
noradrenaline ---and ACh neurons
Behavioral and
EEG arousal,
increased vigilance
Increased reflexes
Increased startle
-
Cessation of
behavior. ------.--.
analgesia
Freezing, conflict test. CER.
social interaction
conditioned analgesia
Mouth open,
----.jaw movements
Facial expressions
ACTH release
of fear
Corticosteroid release
('stress response')
Fig. 2. Schematic diagram of selected outputs of the central nucleus of the amygdala to hypothalamic and brainstem
targets, and possible relationship of these connections to specific signs of fear and anxiety. Abbreviation. CER,
conditioned emotion response. Adapted, with permission, from Ref 1.
210
TINS, Vol. 17, No.5, 1994
Insular cortex
Central nucleus
Secondary sensory cortex,
perirhinal and entorhinal cortex
Basolateral
nucleus
Secondary sensory cortex,
perirhinal cortex
Lateral nucleus
Fig. 3. Schematic diagram of pnnClpal connections within the central, basolateral and lateral amygdaloid nuclei based
on in vitro electrophysiological recording and anatomical data. For simplicity, many other connections, notably
subcortical projections, are not included.
projecting to the same cell. Following a small number
of pairings, the initially weak synaptic input is
potentiated. This potentiation is not seen when an
equal number of the weak and strong inputs are
presented in an unpaired fashion.
In the CAI region of the hippocampus, activation of
the weak input releases excitatory amino acids, such
as glutamate, which bind to both NMDA and AMPA!
kainate receptors on the postsynaptic neuron (for
review see Ref. 30). Binding to the NMDA receptor
has little effect because the highly Ca~+ -permeable
NMDA channel is normally blocked by Mg~+. However, if the postsynaptic neuron is depolarized by a
strong synaptic input activating a sufficient number of
AMPA/kainate receptors, the MgL+ block is removed
and glutamate binding to the NMDA receptor allows
Ca~ + to enter the cell, triggering a series of events
that leads to a lasting potentiation of synaptic efficacy
in the formerly weak input. If glutamate is prevented
from binding to the NMDA receptor by administration
of a competitive NMDA-receptor antagonist, such as
APY, shortly before pairing the weak and strong
stimuli, associative LTP does not occur.
In classical fear conditioning, a neutral stimulus
(conditioned stimulus) could elicit release of glutamate
onto neurons in the amygdala, and this glutamate
could bind to both NMDA and AMPA/kainate receptors. However, this might not produce much of a
behavioral response because at resting membrane
potentials only weak activation of AMPA/kainate
receptors,
and partial blockade of NMDA-channel
permeability by Mg~
occur. However, presentation
of a strong aversive stimulus at about the same time
could further depolarize the neuron, relieve the
remaining :.vlg~ + blockade, and enable Ca~
to enter
the cell. This could trigger events that would increase
TINS, Vol. 17, No.5, 1994
the ability of the conditioned stimulus to activate that
neuron, enabling it then to produce effects similar to
those previously produced only by the strong aversive
stimulus. That Mg~+ appears to only partially block
NMDA currents in the amygdala might explain why
fear conditioning occurs so readily.
Like the hippocampus, both NMDA-dependen(D
and NMDA-independent3~
forms of LTP have been
observed in neurons of the basolateral complex,
depending on the amygdaloid afferent stimulated
(Fig. 4). Therefore, would local infusion of antagonists
of NMDA receptors into the amygdala block the
acquisition of aversive conditioning? Using the fearpotentiated
startle
paradigm,
Miserendino
and
colleagues~9
found that infusion of APY into the
basolateral nucleus caused a dose-dependent blockade
of the acquisition, but not the expression, of conditioned fear. This effect did not seem to result from a
decrease in sensitivity to footshock, a local anesthetic
effect, blockade of visual transmission or permanent
damage to the amygdala. Using similar doses,
Fanselow and colleagues:u found that local infusion of
APY into the basolateral nucleus before training
blocked conditioned freezing measured 24 h later.
Infusion of APY into the immediately adjacent central
nucleus had no effect, implying that the effect was
highly localized. Using a multiple-trial step through
avoidance paradigm, Kim and McGaugh found that
intra-amygdala infusion of IJI.-APY, IJ-APY or (±)-2(carboxypiperazine-4-yl)propyl-I-phosphonic
acid
(CPP) before training, caused a dose-dependent
impairment of retention measured 48 h later:1!. The
potency of the drugs was consistent with their relative
affinities to the NMDA receptor. This effect was not
seen when APY was infused into the striatum,
immediately above the amygdala. Intra-amygdala
211
infusion of DL-APV did not affect footshock sensitivity
or locomotor activity, and the blockade of memory
formation could not be attributed to state-dependent
effects.
Using step-down
inhibitory avoidance,
Izquierdo and colieagues:J'> found that immediate posttraining infusion of APV into either the amygdala,
medial septum, or hippocampus, blocked memory
measured 18 h after training. lJ-2-Amino-5-phosphonovalerate caused amnesia when infused into either
the hippocampus or amygdala immediately after training, but not thereafter:~h.
However, the AMP A/
kainate antagonist CNQX caused amnesia if infused
into the hippocampus or amygdala either immediately,
90 or 180 min after training. By contrast, APV infused
into the entorhinal cortex caused amnesia when given
either 90 or 180, but not immediately or 360, min
after training:Jrd7. These data suggest that a process
sensitive first to APV and CNQX and then only to
CNQX in the amygdala and hippocampus, is critical for
post-training memory processing of this step-down
inhibitory task. Later, an APV -sensitive process in
the entorhinal cortex might come into play.
As mentioned earlier, once learning has occurred,
local infusion of APV into the amygdala does not block
the expression of either fear -potentiated startle or
inhibitory avoidance. In contrast, local infusion of
CNQX into the amygdala dose-dependently blocks the
expression of fear-potentiated startle:JK or retention in
a step-down inhibitory avoidance tes(J9. Once again,
this pattern is similar to LTP where APV does not
reverse LTP once it is established, whereas CNQX
blocks fast synaptic transmission either before or
after LTP.
Thus, excitatory amino acids mediate synaptic
transmission in the amygdala originating from both
cortical inputs and intrinsic amygdaloid connections.
In the amygdala, NMDA receptors
seem to be
involved in the formation of conditioned fear, whereas
AMPA/kainate receptors appear to be involved in the
expression of conditioned fear. As such, these data
are consistent with, but by no means prove, the idea
that an NMDA-sensitive form of LTP in the amygdala
might mediate fear conditioning.
Inhibitory amino acids
ElectroPhysiology. In the basolateral nucleus, stimulation-induced EPSPs are followed by both fast and
slow IPSPs. The fast, GABA-mediated IPSP results
from activation of GABAq receptors, and the slower
IPSP is mediated by activation of GABAg receptorsw
(Fig. 4). The fast IPSP is associated with a large
decrease in resistance, and its possible somatic or
dendritic origin ensures that neurons of the basolateral complex will be effectively inhibited from firing
action potentials, even in the face of strong excitatory
stimuli. The fast onset and short duration of the IPSP
also enables precise moment-to-moment
adjustment
of the cellular response
to excitatory input. In
contrast, the small conductance increase and long
duration associated with the slow IPSP might only
ret-,rulate low-frequency excitatory input during times
of normal neuronal function. Blockade of the fast IPSP
with the GABA; -receptor
antagonist
bicuculline
results in epileptiform burst firing of norn1ally quiescent basolateral neurons, suggesting that the fast
IPSP probably dictates the primary state of excitability in the nucleus. It is possible that both GABAq
212
and GABAg responses
might be mecll,ikcl by
activation of a heterogeneous
population I,f (,'\BA
interneurons lo.n. In addition. both b'j,'J"kll.
a
GABAg-receptor
agonist, and tr<1nS-l-,mll:1Ii,'\dopentane-l,;3-dicarboxylic
acid
(tr<1ns-.'\ll']).
a
metabotropic
glutamate-receptor
a,l!;OIl!st. rl·cluce
glutamatergic transmission via stimul<1tion Ii! thl :'tnaterminalis by an action at presynaptic rl'l'lptl Ir' A reduction in presynaptic GABAw or 111l't;jbl1tl11jJIC
glutamate-receptor
activation might contribute' t I! the
induction of LTP in the basolateral complex. Incb·c!.
kindling results in a decreased sensitivity of prl'synaptic GABAB receptors on terminals of the stri;j
terminalis that are presumed to be glutamatergic L'.
In the central nucleus, focal, low-intensity stimulation of the lateral central nucleus elicits a GABA;mediated inhibition (blocked by bicuculline methiodide) of medial central nucleus neurons, whereas
higher intensity stimulation elicits a GABAg-mediated
inhibition (blocked by phaclofen)2fi. Repetitive stimulation also reveals a slow GABAg-mediated IPSP. In
addition, glutamatergic transmission was blocked by
presynaptic activation of GABAB receptors and A1
adenosine receptors.
Anatomical studies I'> indicate
some terminals of GABA neurons appear to be
presynaptic to cortical terminals in the lateral part of
the central nucleus. The presence of presynaptic A1
adenosine receptors further suggests that, during
periods of high metabolic activity in the adjacent
basolateral nucleus, glutamatergic transmission in the
central nucleus will be restricted. In addition, output
neurons in the central nucleus are heavilv innervated
by GABA terminals of intrinsic originl'>. ~ose and coworkers~(i also demonstrated a strychnine-sensitive
long-lasting IPSP following repetitive stimulation of
the dorsal lateral subdivision of the central nucleus.
This presumed
glycine-mediated
IPSP probably
results from an input arising in the brainstem. In a
nucleus that has such a strong output to the brainstem
cardiovascular regulatory centers, this tight control of
excitability is probably essential. In fact, local infusion
of GABA or chlordiazepoxide into the central nucleus
reduces the severity of stress-induced ulcers, whereas antagonists of GABA receptors increase severity
(for review see Ref. 44).
Behavior, The amygdala has a high density of
benzodiazepine receptors L', which are known to
facilitate GABA transmission. Local infusion of benzodiazepines into the amygdala (for review see Ref. 1)
has anxiolytic effects in the operant conflict test,
social interaction test, measures
of conditioned
freezing and hypoalgesia, the light-dark box test in
mice, and antagonizes the discriminative stimulus
properties of pentylenetetrazol. The anticonflict effect
can be reversed by systemic administration of the
benzodiazepine antagonist flumazenil or co-administration into the amygdala of the GABAA antagonist
bicuculline, and mimicked by local infusion, into the
amygdala, of GABA or the GAB.'\,\ agonist muscimol.
In general, anticonflict effects of benzodiazepines
occur after local infusion into the lateral and basolateral
nuclei, which have the highest densities of benzodiazepine receptors in the amygdala, and not after
local infusion into the central nucleus. More recently,
it has been shown that the anterior part of the
basolateral and central nucleus is especially important
for conflict performance based on both lesion and local
TINS, Vol. 17, No.5. 1994
A
a Control
EPSP
I
b APV 50
c
"
11M
CNOX 10
d APV 50
--r
11M
11M
+
slPSP
~5mV
CNOX 10
11M
50 ms
e CN~X
10
8MI 30
B
11M
~
_
11M
post tetanus
I
OJ
Q.
0
en
o 11M
300
50
11M
APV •
APV 0
250
OJ
.~
Qj
(J)
ru
..0
C
200
150
Q;
0
u
:;a+b
.S-
c+d
OJ
"0
~
............
~
50
-5
0-
C
~
100
OJ
0
5
10
Time (min)
35
15
20
d
30
o0 ,00
000
000CO
25
a
20
b
co ~o
00
c
Q.
E
ru
15
0-
10
0-
5
CJ)
~
~
20 mV
w
oO:Po~
fQ:x9o 0
0
20 ms
-5
t
t
HFS
APV
HFS
0
5
10 15 20 25 30 35 40 45 50
Time (min)
Fig. 4. Synaptic transmission in the basolateral amygdaloid nucleus (BLA). (A, left) Stimulation of the stria terminalis
evokes a multiphasic postsynaptic potential (PSP) in neurons of the BLA. Typically, stimulation (arrow head) elicits an
EPSP followed by a fast IPSP (fIPSP), and subsequent slow IPSP (sIPSP). (A, right) The PSP results from a dual
component glutamate-mediated EPSPand activation of a feedforward inhibitory input. a: A typical response to stria
stimulation. b: 2-Amino-5-phosphonovalerate
(APV) (50IIM) application caused a reduction in the amplitude of both
the fast and slow IPSP. c: CNQX (10 11M)abolished both the EPSPand the flPSP and slPSP to reveal a slow EPSP.d: The
slow EPSP revealed by CNQX is blocked by subsequent addition of APV. e: Antagonism of GABAA receptors with
bicuculline (BM/) enhanced the expression of the slow EPSP.(B) Long-term potentiation (L TP) induced in BLA neurons
following stimulation of the external capsule (EC) is resistant to APV (50 ,11M).(B, left) High-frequency stimulation of the
EC resulted in an increase in the EPSPamplitude (upper trace), the increase was unaffected by 50,uM APV (middle
trace), and blocked by lOOIlM APV (lower trace). (B, right) The 50 11MAPV-resistant LTP persisted for several minutes
following high-frequency stimulation, suggesting that in this pathway NMDA-receptor activation is not needed for
synaptic plasticity. (C) LTP induced in BLA neurons following stimulation of the endopyriform nucleus is sensitive to APV
(50 ,11M).(C, right) In the presence of APV (50 11M),EPSPsevoked before (a), and after (b) high-frequency stimulation of
the endopyriform nucleus are of similar amplitude. Following washout of APV, a similar stimulation paradigm resulted in
an enhanced EPSP.(C, left) The APV-sensitive LTP persisted for several minutes following high-frequency stimulation,
suggesting that NMDA-receptor activation is required for synaptic plasticity in this pathway.
infusion of benzodiazepines. Therefore, taken together,
these results might be sufficient to explain both fearreducing and anxiety-reducing effects of various drugs
given systemically. Local infusion of the benzodiazepine antagonist flumazenil into the amygdala
significantly attenuates the anticonflict effect of the
benzodiazepine agonist chlordiazepoxide given systemicallyHi. This strongly implicates the amygdala in
mediating the anxiolytic effects of benzodiazepines in
normal animals. However, benzodiazepines can still
TINS, Vol. 17, No.5, 1994
have anxiolvtic effects in animals with lesions of the
amygdala 17:10'. In these studies, the lesions produced
anxiolytic effects by themselves. Nonetheless, some
fearful behavior could still be obtained, presumably
because other brain areas can mediate conditioned
fear following damage to the amygdala, and these
areas also seem to be affected by benzodiazepines.
Using inhibitory avoidance, intra-amygdala infusion
of the GABA antagonists bicuculline methiodide FI,
picrotoxin or Ro54864 (4' -purodiazepine) (Ref. 50)
213
immediately after training, produced a dose-dependent
enhancement of retention measured 24-48 h later.
Conversely, infusion of the GABAA agonist muscimol
or the GABAI) agonist baclofen produced retention
deficits. Infusions into the caLtdate nucleus, dorsal to
the amygdala, had no effect.
Thus, it is clear that GABA can potently regulate
cellular excitability in the lateral and basolateral
amygdaloid nuclei by decreasing the release of glutamate or by direct inhibitory actions. These direct
decreases in excitability might explain the depressant
effects of GAB A on aversive conditioning. However,
they might also result from an alteration in the release
of noradrenaline which is known to modulate learning
within the amygdala (for review see Ref. 51). For
example, the facilitatory effect of bicuculline can be
prevented by the f3-adrenergic antagonist propranolol
at a dose which has no significant effect by itself. In
addition, output neurons of the central nucleus, which
project to brainstem targets areas known to be
involved in the autonomic and somatic aspects of
conditioned fear, are tightly regulated by GABA and
other inhibitory transmitters,
disruption of which
might greatly amplify fear and stress.
Selected references
Acknowledgements
Thiswork was
supported by NIMH
Grant MH-47840,
ResearchScientist
DevelopmentA ward
MH-00004, and a
grant from the Air
ForceOffice of
ScientificResearchto
MD., Alzheimer's
Assoc./Harold Wand
GeorgianaSpaght
Memorial Pilot
ResearchGrant No.
PRG-92-119to D.R.
and NIH Grant
NS25139to Me.
214
1 DavIs, M. (1992) in The Amygdala. Neurobiological Aspects
of Emotion, Memory, and Mental Dysfunction (Aggleton,
J. P., ed.), pp. 255-306, John Wiley-liss and Sons
2 Aggleton, J. P, ed. (1992) The Amygdala. Neurobiological
Aspects of Emotion, Memory, and Mental Dysfunction, John
Wiley-Liss and Sons
3 LeDoux, J. E., Cicchetti, P., Xagoraris, A. and Romanski,
L M. (1990) J. Neurosci. 10, 1062-1069
4 Turner, B. and Zimmer, J. (1984) J. Compo Neurol. 227,
320-334
5 Bernard, J. F., Alden, M. and Besson, J. M. (1993) J. Comp
Neurol. 329, 201-229
6 Yasui, Y., Breder, CD.,
Saper, C B. and Cechelto, D. F.
(1991) J. Compo Neurol. 303, 355-374
7 Turner, B. and Herkenham, M. (1991) J. Compo Neurol 313,
295-325
8 McDonald, A. J. (1984) J. Compo Neurol. 222, 589-606
9 McDonald, A. J. and Pearson, J. C (1989) NeurosCi. Lett.
100,53-58
10 McDonald, A. J. (1991) Neurosci. Lett. 44, 15-33
11 Alheid, G. and Heimer, L (1988) Neuroscience 27, 1-39
12 Sun, N. and Cassell, M. D. (1993) J. Compo Neurol 330,
381-404
13 Cassell, M. D. and Gray, T. S. (1989) J. Compo Neurol. 281,
320-334
14 Freedman, L. J. and Cassell. M. D. (1994) Brain Res. 633,
243-252
15 Sun, N., Yi, H. and Cassell, M. D. (1994) J. Compo Neurol
370, 43-64
16 Gray, T. S. (1989) In Autonomic Neuropeptide Connections
of the Amygdala (Tache, Y., Morley, J. E. and Brown, M. R,
eds), pp. 92-106, Springer Verlag
17 Takayama, K. and Miura, M. (1991) Neurosci. Lett. 134,
62-66
18 Schiess, M. C, Asprodini, E. K., Rainnie, D. G. and ShinnickGallagher, P S. (1993) Brain Res. 604, 283-297
19 Brothers, L. A. and Finch, D. M. (1985) Brain Res. 359,
10-20
20 Mello, L E. A. M., Tan, A. M. and Finch, D. M. (1992) Brain
Res. 587, 24-40
21 Rainnle, D. G., Asprodini, E. K. and Shinnick-Gallagher, P.
(1991) J. Neurophysiol 66, 986-998
22 Washburn, M. S. and Moises, H. C (1992) J. Neurosci. 12,
4066-4079
23 Gean, P. W. and Chang, F. C (1992) Synapse 11, 1-9
24 Sugita, S, Shen, K. Z. and North, R. A. (1992) Neuron 8,
199-203
25 Sugita, S, Tanaka, E. and North, R. A. (1993) J. PhyslOl 460,
705-718
26 Nose, I., Higashi, H., Inokuchi, H. and Nishi, S. (1991)
J. Neurophysiol. 65, 1227-1241
27 Clugnet, M. C and LeDoux, J. E. (1990) J. Neurosci. 10,
2818-2824
28 Kim, J. J., DeCola, J. P., Landelra-Fernandez, J. and Fanselow,
M. S. (1991) Behav. NeurosCi. 105, 126-133
29 Mlserendlno, M. J. D, Sananes, C B., Melia, K. R. and
Davis, M. (1990) Nature 345, 716-718
30 Bliss, T. V P and Coilingridge, G. L. (1993) Nature 361,
31-39
31 Gean, P. W., Chang, F. C, Huang, C C, lin, J. H. and Way,
L. J. (1993) Brain Res Bull. 31, 7-11
32 Chapman, P F. and Bellavance, L. L. (1992) Synapse 11,
310-318
33 Fanselow, M. S., Kim, J. J. and Landeira-Fernandez, J. (1991)
Soc. Neurosci. Abstr. 17, 659
34 Kim, M. and McGaugh, J. L. (1992) Brain Res. 585, 35-48
35 Izquierdo, I. et al. (1992) Behav. Neural Bioi. 58, 16-26
36 Jerusallnsky, 0 et al. (1992) Behav. Neural Bioi 58, 76-80
37 Ferreira, M. B. C, Da Silva, R. C, Median, J. H. and
Izquierdo,
I. (1992) Pharmacol. Biochem. Behav 41,
767-771
38 Kim, M., Campeau, S., Falls, W A. and Davis, M. (1993)
Behav Neural Bioi 59, 5-8
39 Izquierdo, I. et al. (1993) Behav. Neural Bioi. 59, 1-4
40 Rainnie, D G, Asprodini, E. K. and Shinnick-Gallagher, P.
(1991) J. Neurophysiol 66,999-1009
41 Sugita, S., Johnson, S. W. and North, R A. (1992) Neurosci
Lett. 134,207-211
42 Asprodinl, E. K., Ralnnle, D. G. and Shinnick-Gallagher, P.
(1992) J. Pharmacol. Exp. Ther 262, 1011-1021
43 Rainnie, D. G. and Shinnick-Gallagher, P. (1992) Neurosci.
Lett. 139, 87 -91
44 Henke, P. G. (1992) In The Amygdala. Neurobiological
Aspects of Emotion, Memory, and Mental Dysfunction
(Aggleton, J. P, ed.), pp. 323-338, John Wiley-Liss and Sons
45 Niehoff, D. L and Kuhar, M. J. (1983) J. Neurosci. 3,
2091-2097
46 Hodges, H., Green, S and Glenn, B. (1987) Psychopharmacology 92, 491-504
47 Kopchla, K. L, Altman, H. J. and Commissaris, R. L. (1992)
Pharmacol Biochem. Behav 43,453-461
48 Yadin, E., Thomas, E., Strickland, C E. and Gnshkat, H. L
(1991) Psychopharmacology 103,473-479
49 Brioni, J. D., Nagahara, A. H. and McGaugh, J. L. (1989) Brain
Res. 487, 105-111
50 Da Cunha, C et al. (1991) Brain Res. 544, 133-1.36
51 McGaugh, J. L. (1992) In The Amygdala. Neurobiological
Aspects of Emotion, Memory, and Mental Dysfunction
(Aggleton, J. P., ed.), pp. 431-451, John Wiley-Liss and Sons
TINS, Vol. 17, No.5, 1994