* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download When neurons form memories
Neuroanatomy wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Nonsynaptic plasticity wikipedia , lookup
Effects of sleep deprivation on cognitive performance wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Aging brain wikipedia , lookup
Brain Rules wikipedia , lookup
Nervous system network models wikipedia , lookup
Limbic system wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Cognitive neuroscience of music wikipedia , lookup
Optogenetics wikipedia , lookup
Procedural memory wikipedia , lookup
Metastability in the brain wikipedia , lookup
Emotion and memory wikipedia , lookup
Socioeconomic status and memory wikipedia , lookup
Exceptional memory wikipedia , lookup
Prenatal memory wikipedia , lookup
Misattribution of memory wikipedia , lookup
State-dependent memory wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Traumatic memories wikipedia , lookup
Memory and aging wikipedia , lookup
Eyewitness memory (child testimony) wikipedia , lookup
Collective memory wikipedia , lookup
Mind-wandering wikipedia , lookup
Music-related memory wikipedia , lookup
Epigenetics in learning and memory wikipedia , lookup
Synaptic gating wikipedia , lookup
Childhood memory wikipedia , lookup
Sparse distributed memory wikipedia , lookup
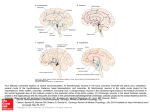
Update TRENDS in Neurosciences Vol.26 No.3 March 2003 123 When neurons form memories Pascal Fries, Guillén Fernández and Ole Jensen F.C. Donders Centre for Cognitive Neuroimaging, Adelbertusplein 1, 6525 EK Nijmegen, The Netherlands Although long-term memory is central among our cognitive functions, the search for a direct neurophysiological correlate to it has proven difficult. The formation of new memories depends on the hippocampus and adjacent cortex, but the final storage is thought to be in a widely distributed neocortical network. Recent experiments, using simultaneous recordings from hundreds of sites in monkey neocortex, have revealed the activation of such a distributed network – probably reflecting the consolidation of long-term memory storage. Long-term memory is a hallmark trait of the functioning of our CNS. The second experience is never exactly the same as the first. Each event leaves a trace in our brain that shapes the way future encounters are handled. However, it appears that the neocortical brain areas that produce our conscious experience of an event cannot at the same time directly store this event for later recall. Rather, an intermediate trace is initially stored in the medial temporal lobe (MTL), an area that includes the archicortical hippocampus and the adjacent cortical areas (the entorhinal, perirhinal and parahippocampal cortices) [1]. Subsequently, memories gradually become independent of the MTL and established in neocortex [2] by an operation called consolidation [3]. Memory consolidation is presumably based on a medial temporal – neocortical interaction and might take place predominantly during periods of rest or sleep, when no new events are processed [4,5]. Distributed memories A central finding in lesion studies of memory is that consolidated, MTL-independent long-term memories are not destroyed entirely by circumscribed lesions in the neocortex [6,7]. This has led to the idea that neocortical memory traces are widely distributed [8,9] and probably encoded in the strength of synaptic connections among neurons across large areas of neocortex [10]. This notion received substantial experimental support, and numerous theoretical modelling studies have demonstrated the remarkable capacity of distributed storage in networks with modifiable synaptic connections [11]. However, although distributed memory storage became the dominant concept in the field, it evaded direct neurophysiological demonstration until very recently. The distributed nature that renders neocortical memory storage so powerful has made it very hard to study. The number of neurons in our brain is Corresponding author: Pascal Fries ([email protected]). http://tins.trends.com daunting and the number of their connections even more so. Demonstrating that an event has changed neuronal interactions requires simultaneous recordings from hundreds of neurons distributed over the cortex of behaving animals, and a sophisticated correlation analysis of the recorded neuronal activity. Kari Hoffman and Bruce McNaughton recently succeeded in this technical tour de force [12]. They implanted four different neocortical areas of macaque monkeys each with 144 electrodes. Each electrode could be positioned independently to isolate the spiking activity of a single neuron. The implanted areas were the posterior parietal cortex, the motor cortex, the somatosensory cortex and the dorsal prefrontal cortex. The monkeys were trained to perform tasks that co-activated neurons in those areas. During the recordings, monkeys first performed the trained tasks and then they rested quietly. Long-range neuronal interactions During task performance, a network of neurons distributed over all four recorded areas was activated, as expected. The remarkable finding was that neurons that had been active together during task performance ‘re-enacted’ their play during the following rest period, despite the fact that no task was performed (Fig. 1). The neuronal activity patterns were indeed generated by the preceding task performance and were not simply a default state of the system. This was demonstrated by recording from neurons during a control rest period just before task performance. The activation patterns shared by the task and the post-task rest could not be found during the pre-task rest. Activation patterns during task performance contain temporal structure because different neurons are involved at different times during the task. Additional analyses revealed that the reactivation of patterns during the post-task rest period preserved some of the temporal order of neuronal activation from the task. This finding is particularly interesting in the light of theoretical work demonstrating that sequence information can be synaptically encoded and recalled by physiologically realistic learning rules [13]. Thus, the sequential reactivation during the rest period suggests that not only a given event, but also the particular sequence of events, is being consolidated. Although the study by Hoffman and McNaughton provided important new insights, it also raises new questions. The most important unresolved issue is probably whether the observed neocortical reactivations are truly a correlate of consolidation of declarative memory or whether Update 124 TRENDS in Neurosciences Vol.26 No.3 March 2003 they are correlates of other forms of learning and memory, such as skill learning. Because the formation of new declarative memories depends crucially on the MTL, cortical reactivations should do so as well if they have a functional role in declarative memory. There seem to be at least two complementary ways of confirming an interaction between the MTL and the neocortex. One approach would be to record simultaneously from both structures and test for correlations between MTL and neocortex related to reactivation during sleep. A second crucial experiment would require a permanent lesion or transient inactivation of the MTL. If neocortical reactivations are indeed a signature of memory traces being activated by MTL, as suggested by Hoffman and McNaughton, these reactivations should disappear and memory performance should decrease in a post-rest test whenever the MTL is effectively inactivated. Pre-task rest Task References Post-task rest TRENDS in Neurosciences Fig. 1. The left half of the figure illustrates the three behavioral conditions studied by Hoffman and McNaughton: the pre-task rest period, the task period and the post-task rest period [12]. The right half illustrates the main results of the neurophysiological recordings. Two neurons are depicted that respond selectively to two different aspects of the task. One of the neurons responds to the red dot on the computer screen that has to be touched by the monkey to obtain juice; the other responds to the actual juice reward. During the pre-task rest period, the monkey is not particularly concerned with red dots or juice, but could be ‘dreaming’ of a banana. Consequently, the ‘red-dot neuron’ and the ‘juice-reward neuron’ do not show a particular coordinated firing pattern. This changes during the task, when the red dot neuron and the juice-reward neuron are repeatedly activated in temporal succession. This sequence of activation is spontaneously reactivated during the post-task rest, when no task is performed but the recent experience from the task period is consolidated into long-term memory. Modified, with permission, from Ref. [14]. q (2001) Nature Publishing Group (http://www.nature.com/). 1 Scoville, W.B. and Milner, B. (1957) Loss of recent memory after bilateral hippocampal lesions. J. Neurol. Neurosurg. Psychiatry 20, 11–21 2 Squire, L.R. and Zola-Morgan, S. (1991) The medial temporal lobe memory system. Science 253, 1380 – 1386 3 Burnham, W.M. (1903) Retroactive amnesia: illustrative cases and a tentative explanation. Am. J. Psychol. 14, 255 – 257 4 Qin, Y.L. et al. (1997) Memory reprocessing in corticocortical and hippocampocortical neuronal ensembles. Philos. Trans. R. Soc. Lond. B Biol. Sci. 352, 1525 – 1533 5 Buzsáki, G. (1998) Memory consolidation during sleep: a neurophysiological perspective. J. Sleep Res. 7 (Suppl. 1), 17 – 23 6 Lashley, K.S. (1929) Brain Mechanisms and Intelligence: a Quantitative Study of Injuries to the Brain, Dover Publications 7 Gabrieli, J.D. (1998) Cognitive neuroscience of human memory. Annu. Rev. Psychol. 49, 87 – 115 8 John, E.R. (1967) Mechanisms of Memory, New York Academic Press 9 Marr, D. (1971) Simple memory: a theory for archicortex. Philos. Trans. R. Soc. Lond. B Biol. Sci. 262, 23 – 81 10 Hebb, D.O. (1949) The Organization of Behavior, Wiley 11 Amit, D.J. (1989) Modeling Brain Function: the World of Attractor Neural Networks, Cambridge University Press 12 Hoffman, K.L. and McNaughton, B.L. (2002) Coordinated reactivation of distributed memory traces in primate neocortex. Science 297, 2070– 2073 13 Abbott, L.F. and Blum, K.I. (1996) Functional significance of long-term potentiation for sequence learning and prediction. Cereb. Cortex 6, 406– 416 14 Engel, A.K. et al. (2001) Dynamic predictions: oscillations and synchrony in top-down processing. Nat. Rev. Neurosci. 2, 704 – 716 0166-2236/03/$ - see front matter q 2003 Elsevier Science Ltd. All rights reserved. doi:10.1016/S0166-2236(03)00023-7 Mouse Knockout & Mutation Database Established in 1995, the Mouse Knockout & Mutation Database (MKMD; http://research.bmn.com/mkmd) is BioMedNet’s fully searchable database of phenotypic information related to knockout and classical mutations in mice. MKMD offers over 7000 entries and includes a new reviews section on mouse models of human diseases and up-to-date fact files for all disease reviews. http://tins.trends.com