* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download [Ca2+]c dynamics in spontaneously firing dopamine neurons of the
Activity-dependent plasticity wikipedia , lookup
Central pattern generator wikipedia , lookup
Neuromuscular junction wikipedia , lookup
Neural coding wikipedia , lookup
Biological neuron model wikipedia , lookup
Axon guidance wikipedia , lookup
Subventricular zone wikipedia , lookup
Apical dendrite wikipedia , lookup
Nervous system network models wikipedia , lookup
Multielectrode array wikipedia , lookup
Electrophysiology wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Neurotransmitter wikipedia , lookup
Spike-and-wave wikipedia , lookup
NMDA receptor wikipedia , lookup
Long-term depression wikipedia , lookup
Synaptic gating wikipedia , lookup
Development of the nervous system wikipedia , lookup
Neuroanatomy wikipedia , lookup
Circumventricular organs wikipedia , lookup
Signal transduction wikipedia , lookup
Synaptogenesis wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Optogenetics wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Glutamate receptor wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
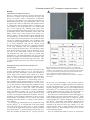
Research Article 2665 Glutamate-mediated [Ca2+]c dynamics in spontaneously firing dopamine neurons of the rat substantia nigra pars compacta Yu Mi Choi, Shin Hye Kim, Dae Yong Uhm and Myoung Kyu Park* Medical Research Center for Regulation of Neuronal Cell Excitability and Department of Physiology, Sungkyunkwan University School of Medicine, 300 Chunchun-dong Jangan-ku, Suwon 440-746, Korea *Author for correspondence (e-mail: [email protected]) Accepted 14 March 2003 Journal of Cell Science 116, 2665-2675 © 2003 The Company of Biologists Ltd doi:10.1242/jcs.00481 Summary The mechanism by which glutamate regulates the cytosolic free Ca2+ concentration ([Ca2+]c) in spontaneously firing dopamine neurons is not clear. Thus we have investigated the glutamate-mediated [Ca2+]c dynamics in the acutely isolated dopamine neurons from the rat substantia nigra pars compacta by measuring [Ca2+]c and spontaneously occurring action potentials (SAPs). The freshly isolated dopamine neurons showed tetrodotoxin (TTX)-sensitive spontaneous firing of 2-3 Hz and the resting [Ca2+]c decreased with abolition of the SAPs. The level of [Ca2+]c was affected by the spontaneous firing rate. In the presence of the Na+ channel antagonist, TTX (0.5 µM), glutamate increased [Ca2+]c by activating different glutamate receptors depending on the glutamate concentration used. Addition of glutamate at low concentrations (<3 µM) raised [Ca2+]c mainly by activating metabotropic glutamate receptors (mGluR), whereas at high concentrations (>10 µM) it raised [Ca2+]c mainly by activating AMPA/kainate Introduction Glutamate-mediated rises in cytosolic free Ca2+ concentration ([Ca2+]c) are of the ultimate importance for neuronal excitability (Nakanishi, 1992; Berridge, 1998); they also play a crucial role in the outgrowth of dendrites and axons, neuronal differentiation, synaptic remodeling and plasticity (Collingridge and Singer, 1990), regulation of mitochondrial metabolism (Duchen, 1999), gene expression (Gallin and Greenberg, 1995; Ginty, 1997), and neurotoxicity (Sattler and Tymianski, 2000). Since glutamate is involved in many such functions and can activate various glutamate receptors, the [Ca2+]c signals related to glutamate receptors in neuronal cells have long been studied in a variety of neuronal cells (Mayer and Miller, 1990; Gallin and Greenberg, 1995). Therefore, it is well known that elevated glutamate, in addition to generating excitatory synaptic potentials and changing membrane potential, increases [Ca2+]c by the activation of AMPA/kainate receptors, NMDA receptors, and group 1 metabotropic glutamate receptors (mGluR), as well as by the secondary opening of voltage-operated Ca2+ channels (VOCCs) (Mayer and Miller, 1990; Nakanishi, 1992). Dopamine neurons located at substantia nigra pars compacta (SNc) are known to generate spontaneous firing and, receptors. The contribution of NMDA receptors to the glutamate-mediated [Ca2+]c rises was largest at intermediate concentrations of glutamate. Activation of mGluR elicited a Ca2+ release from intracellular Ca2+ stores and continuous Ca2+ influx out of the cell. The spontaneous firing activities were highly enhanced by submicromolar levels of glutamate and abolished at levels above 10 µM. From these results, we conclude that at low glutamate concentrations the [Ca2+]c in the dopamine neurons is mainly governed by mGluR and the firing activities, whose rate is regulated at submicromolar glutamate concentrations, but at higher glutamate concentrations [Ca2+]c is dominantly affected by AMPA/kainate receptors. Key words: Glutamate, AMPA receptor, NMDA receptor, Metabotropic glutamate receptor, Calcium, Dopamine neuron, Substantia nigra pars compacta glutamate, as a major excitatory neurotransmitter, is reported to modulate the firing patterns of the SNc dopamine neurons through many kinds of glutamate receptors (Overton and Clark, 1997; Meltzer et al., 1997; Kitai et al., 1999). Subthalamic and pedunculopontine nuclei and neurons in prefrontal cortex provide glutamatergic inputs to the dopamine neurons and can regulate firing patterns and increase their frequency of SAPs (Cardozo, 1993; Meltzer et al., 1997; Kitai et al., 1999; Grillner and Mercuri, 2002). In addition to these inputs, dopamine neurons can be tonically exposed to glutamate at variable concentrations owing to ambient glutamate levels, local synaptic activities of glutamatergic neurons and pathologic conditions such as seizures, injuries, and hypoxia (Benveniste et al., 1984; Lerma et al., 1986; Sah et al., 1989; HerreraMarschitz et al., 1996; Obrenovitch and Urenjak, 1997). The increased SAP frequency could modulate target cells by changing the dopamine release at the axon terminals as well as at the somatodendritic trees, particularly in dopamine neurons (Nedergaard et al., 1988; Jaffe et al., 1998; Chen and Rice, 2001). The VOCCs and Ca2+-dependent ion channels also appear to be critical in the ionic mechanisms by which the cells spontaneously fire, and the glutamate-mediated changes in 2666 Journal of Cell Science 116 (13) [Ca2+]c could affect the electrical activities (Hounsgaard et al., 1992; Kang and Kitai, 1993; Amini et al., 1999). In addition, Ca2+ signals in dopamine neurons appear to play important roles in somatodendritic dopamine releases or dendritic secretions (Nedergaard et al., 1988; Jaffe et al., 1998). Thus the glutamate-mediated Ca2+ signals appear to be important in maintaining the functions of dopamine neurons. However, how each glutamate receptor is activated and cooperatively contributes to the [Ca2+]c dynamics is not clear in SNc dopamine neurons. This is partly due to the specific situation of the neurons, which always exist within the brain tissue and so are persistently influenced by networks and nearby cells. Moreover, owing to the high ambient glutamate concentration in cerebrospinal and interstitial fluids (Herrera-Marschitz et al., 1996), it is difficult to observe the effect of glutamate on [Ca2+]c dynamics in vivo or in brain slices, especially at low glutamate concentrations. Thus we have investigated how glutamate activates different glutamate receptors and raises [Ca2+]c at different glutamate concentrations, by using acutely isolated dopamine neurons from the rat SNc. By taking advantage of using freshly isolated cells, we were able to remove network interferences between neurons and/or glial cells and clearly clamp glutamate concentrations even at very low levels. Thus we show that the [Ca2+]c, depending on the glutamate concentration, can be differentially regulated by different glutamate receptors as well as by the rate of spontaneous firing. At low glutamate concentrations (0.3-3 µM), the level of [Ca2+]c was determined mainly by the activation of mGluR as well as the enhanced frequency of spontaneous firing. However, at high glutamate concentrations (>10 µM) [Ca2+]c was affected mainly by the activation of AMPA/kainate receptors. Materials and Methods Preparation of SNc dopamine neurons We used the postnatal day 9 to 16 Sprague-Dawley rats. After decapitation, whole brains were quickly removed and placed in icechilled oxygenated HEPES-buffered saline, which contains (in mM): 135 NaCl; 5 KCl; 1 CaCl2; 1 MaCl2; 25 D-glucose; 10 HEPES; adjusted to pH 7.3 with NaOH. The brain was cut into a midbrain block containing SNc and coronal slices of 300-400 µm thickness were obtained with a vibratome (TPI, USA). Subsequently, the SNc regions of the slices demarcated by dark color were dissected out with a scalpel blade and digested with the fully oxygenated HEPESbuffered saline containing papain (4-10 U/ml, Worthington) for 20-60 minutes at 34-37°C. Next, the tissue segments were rinsed with enzyme-free saline and then gently triturated with a graded series of fire polished Pasteur pipettes (Chung et al., 2000). The isolated cells were plated on poly-D-lysine-coated small glass coverslips that were already fitted for a recording chamber mounted on an inverted microscope equipped with a fluorescence measurement system. Solutions and chemicals The normal bath solution contains (in mM): 140 NaCl, 5 KCl, 10 HEPES, 10 D-glucose, 1 CaCl2, 1 MgCl2. The pH and osmolarity were adjusted to 7.4 and about 300 mOsm with NaOH and sucrose. When we applied glutamate to stimulate NMDA receptors, we added 1 µM glycine in the bath solution. Among the chemicals related to ionotropic/metabotropic glutamate receptors and ion channels, (s)3,5-dihydroxyphenylglycine (DHPG, group 1 mGluR agonist), 6-cyano-7-nitro-quinoxaline-2,3-dione (CNQX, AMPA/kainate receptor antagonist), D(–)-amino-5-phosphonopentanoic acid (AP-5, NMDA receptor antagonist), tetrodotoxin (TTX, Na+ channel antagonist), CPCCOEt (type1 mGluR antagonist), and (R,S)-AMPA were obtained from Tocris, and nifedipine, ω-conotoxin GVIA, and ω-agatoxin IVA were from Alomone Laboratories (Jerusalem, Israel). All other materials were obtained from Sigma. Measuring cytosolic Ca2+ concentration The isolated SNc cells were incubated with 2-5 µM fura-2-AM at room temperature (20-24°C) for 20-35 minutes. After that, the cells were washed with normal physiological salt solution twice. All cells were used within 3 hours of isolation. Single cell fluorescence intensity was measured using an Olympus IX70 inverted microscope (40× objective or 60× water immersion objective), attached with a charge coupled device (CCD) image intensifier camera (Quantix) and Metafluor software (Universal Imaging). We used 340/380 dual excitations with a 400 nm dichroic mirror and emitted light was collected with a long pass filter of 450 nm. The details are described previously (Park et al., 2002). The ratio (340/380) of fluorescence intensities measured at the cell bodies was calibrated with the maximum and minimum ratio values obtained after exposing to 15 µM ionomycin and 10 mM Ca2+ or 10 mM EGTA, by using a dissociation constant of 150 nM for Ca2+-fura2 at room temperature (Neher and Augustine, 1992). Measuring electrical activities The patch clamp system (EPC-9, HEKA) was used to measure spontaneous firing activities. Patch pipettes were made by a Sutter puller and pipette tips were polished with Narishige Forge. The resistance of patch pipettes was 2-3 MΩ. We made whole cell or cellattached configurations in the current clamp mode. In cell-attached patch experiments, the electrical signals were continuously sampled at 2 kHz (1 kHz filter) and stored in an IBM-compatible computer for further analysis. In this case, patch pipettes were filled with the bath solution. The electrical signals were much the same in the extracellular recordings (Grace and Bunney, 1983a; Grace and Bunney, 1983b). Frequency conversion of SAPs was performed with Igor ver. 4 and some of data were analyzed with Origin ver. 6.0. When recorded in the whole-cell configuration, patch pipettes were filled with the internal solution whose compositions are (in mM): 125 Kgluconate; 5 KCl; 8 NaCl; 0.1 CaCl2; 1 MgCl2; 0.75 EGTA; 10 HEPES; 2 Mg-ATP; adjusted to pH 7.3 with KOH. Immunocytochemistry The acutely isolated cells on glass coverslips were rinsed twice by phosphate-buffered saline (PBS) and fixed with 4% paraformaldehyde for 40 minutes at room temperature. After fixation, the cells were washed with PBS and then incubated in the PBS with 1% bovine serum albumin (BSA) and 0.1% Triton X-100 for 60 minutes. After that, the cells were incubated for 2 hours in PBS containing tyrosine hydroxylase antibodies (Pel-Freez, Rogers, Arkansas, USA, diluted 1:100), 1% BSA, and 0.1% Triton X-100. Next, the cells were rinsed three times with PBS and incubated with fluorescein isothiocyanate (FITC)-conjugated goat anti-rabbit IgG (Molecular Probes) diluted by 1:200 in PBS. After incubation for 1 hour at room temperature, the fluorescent antibodies were removed by washing three times with PBS. The fluorescence images were obtained in a Zeiss 510 confocal laser scanning microscope with a 488 nm excitation line and 505-545 nm emission filter. Statistics Paired student’s t-test was used and P-values less than 0.05 were regarded as significantly different. Glutamate-mediated [Ca2+]c changes in dopamine neurons 2667 Results Identification of dopamine neurons When we isolated cells from the selectively dissected SNc brain slices, a majority of the cells showed a very large soma having 3-6 neurites, which is characteristic of dopamine neurons (Fig. 1Aa). But there were small cells having only two bidirectional neurites (Fig. 1Ab, small bipolar cells) as well as atypical cells such as a small body with multiple neurites or a large body with two opposite neurites. Among the 104 cells isolated, 83 were large multipolar, 5 were small bipolar and 16 were atypical cells. Although many papers have already reported that dopaminergic neurons have a large body and multiple neurites (Grace and Bunny, 1983a; Grace and Bunny, 1983b; Grace, 1988; Cardozo, 1993; Chung et al., 2000), in order to confirm whether the large multipolar cells are dopaminergic, we stained cells with the antibody (PelFreez, USA) for tyrosine hydroxylase (TH), which is the main enzyme for synthesizing dopamine. After incubation with secondary antibodies tagged with FITC, we found that 76 out of 83 large multipolar cells (91.5%) showed strong fluorescence intensity as shown in Fig. 1, suggesting that most of the large multipolar cells are dopaminergic neurons. However, among the atypical cells, a few cells having a large soma showed positive staining with TH antibodies (3/16), but none of the small bipolar neurons was stained with TH antibodies (0/5 cells). Fig. 1 shows two typical large multipolar (Fig. 1Aa) and small bipolar cells (Fig. 1Ab) and fluorescence images after staining with TH antibodies. Only the large multipolar cells showed strong fluorescence. Thus we have performed experiments only in these large multipolar neurons. Spontaneous firing activities and cytosolic Ca2+ concentration It is well known that dopamine neurons in substantia nigra generate spontaneous firing in vivo, in vitro and even in isolated cells (Grace and Bunney, 1983a; Grace and Bunney, 1983b; Grace, 1988; Cardozo, 1993; Uchida et al., 2002). Thus, in order to test whether the neurons we isolated produce SAPs, we used patch-clamp techniques. When we made the whole-cell configuration in a current-clamp mode, we observed SAPs with an average frequency of 2-3 Hz (n=10). The membrane potential fluctuated between –64 and –50 mV (n=10, data not shown). Interestingly, this spontaneous firing was also detected in a cell-attached configuration, whose shapes were similar to those that Grace and Bunney previously reported in extracellular recording conditions (Grace and Bunney, 1983b) (Fig. 2). In the cell-attached configuration we continuously recorded spontaneous firing activities for a long time without much fluctuation. One more advantage of this recording configuration is that it does not disturb natural concentrations of Ca2+ buffers and soluble signalling molecules, thereby we mainly recorded them in this condition. To investigate the relationship between spontaneous firing activities and [Ca2+]c, we recorded them in the fura-2-loaded cells at the same time. As shown in Fig. 2, when we applied 0.5 µM TTX, the spontaneous firing was completely blocked (Fig. 2Aa,b) and [Ca2+]c decreased at the same time (Fig. 2Ac). After TTX washout, the SAPs and [Ca2+]c slowly restored to Fig. 1. Identification of dopaminergic neurons. (A) Transmitted images. Among the three neurons isolated, two large multipolor cells (marked a), were dopaminergic, whereas a small bipolar cell (marked b) was not (stained with tyrosine hydroxylase (TH) antibodies and FITC-conjugated secondary antibodies). Bar, 20 µm. (B) Classification of acutely isolated cells from the SNc according to their shapes. the previous level. Interestingly, in this particular cell there were some fluctuations of firing frequencies after washout of TTX, and the changes in [Ca2+]c were exactly mirrored by the frequency changes in SAPs. This is a good example showing that resting [Ca2+]c in spontaneously firing dopamine neurons is highly dynamic and easily affected by the frequency of SAPs. We observed a similar phenomenon in eight cells. Since there was no neurotransmitter in the bath solution, we suspected that VOCCs were playing a major role in Ca2+ influx in the spontaneously firing cells. Thus we used a non-specific voltage-operated Ca2+ channel antagonist, Cd2+. As soon as 100 µM Cd2+ was applied, SAPs suddenly disappeared and then slowly restored to the previous level while the [Ca2+]c remained at the decreased level (Fig. 2Ba,b,c, n=5). This suggests that the SAPs at resting conditions could activate VOCCs to allow Ca2+ influx and help to keep [Ca2+]c elevated in SNc dopamine neurons. Next we examined what kinds of VOCCs are expressed in 2668 Journal of Cell Science 116 (13) Fig. 2. Electrical activities of acutely isolated SNc dopamine neurons. Spontaneous firing activities were recorded from the cell-attached current-clamp cells (A,Ba). Their firing activities were converted into the frequency-time graph (A,Bb). The [Ca2+]c using fura-2 was measured from the same cells (A,Bc). (A) The spontaneous firing activities were completely blocked by 500 nM TTX. Note that the change in [Ca2+]c was mirrored by the firing rate. (B) The spontaneous firing was initially blocked by a nonspecific Ca2+ channel antagonist, 100 µM CdCl2. But it recovered after some time even in the presence of CdCl2. Note the persistent decreased [Ca2+]c level after a full recovery of the spontaneous electrical activities. 200 20 25 0 0 5 10 15 20 [Ca ]c (nM) 800 600 400 100 µM CdCl2 2+ 10 µM Nif 1 µM ω-conoGVIA 1 µM ω-aga IVA 200 0 200 0 0 5 10 15 Time (min) 20 25 0 5 10 15 10 15 20 25 Time (min) E 800 400 5 Time (min) F 600 0 25 20 25 100 80 60 40 20 Ni f 15 KC l 10 Relative [Ca2+]c change (%) 5 Time (min) [Ca2+]c (nM) 200 0 0 D 400 100 µM CdCl 2 0 400 600 L-,N-,P/Q-type [Ca 2+ ] blocker 200 1 µM ω-cono GVIA ω-aga IVA 400 600 ω-cono GVIA 10 µM Nif 1 µM ω-aga IVA 800 2+ 600 C 800 [Ca ]c (nM) B 800 [Ca2+]c (nM) [Ca2+]c (nM) A 0 Time (min) Fig. 3. KCl-elicited [Ca2+]c rises through voltage-operated calcium channels. After application of 60 mM KCl for 12 seconds (䉱), the KCl solution was reapplied under the presence of L-type Ca2+ channel (10 µM nifedipine, A), N-type Ca2+ channel (1 µM ω-conotoxin GVIA, B), and P/Q-type Ca2+ channel (1 µM ω-agatoxin IVA, C) antagonists, a cocktail of the above three antagonists (D), or 100 µM CdCl2 (E). (F) Summary of the inhibitions of the KCl-elicited [Ca2+]c rises by VOCC antagonists; nifedipine (36.0±3.0% of control, n=10), ω-conotoxin GVIA (69.5±8.8%, n=5), ω-agatoxin IVA (92.3±6.6%, n=4), cocktail of the above three antagonists (23.2±4.1%, n=3), and CdCl2 (15.0±2.7%, n=4). *Significant difference (paired student t-test, P<0.05). Nif, nifedipine; ω-cono GVIA, ω-conotoxin GVIA; ω-aga IVA, ω-agatoxin IVA. Glutamate-mediated [Ca2+]c changes in dopamine neurons A Glutamate 2669 B 300 µM 100 µM 10 µM 3 µM 1 µM c 1 min 30 µM CNQX 50 µM AP- 5 10 µM Nif f Nif 1 µM 1 µM ω-conoGVIA ω-conoGVIA ω-agaIVA ω-agaIVA 200 µM CdCl2 CdCl2 ω-a ga IVA ω-cono GVIA CNQX Nif+ω -cono GVIA + ω -aga IVA 0 Nifedipine 20 AP-5 40 Glutamate Relative [Ca2+]c change (%) 100 60 1 10 100 1000 Glutamate-mediated [Ca2+]c rises and related receptors In the presence of 0.5 µM TTX, we applied glutamate at various concentrations and measured [Ca2+]c. As shown in Fig. 4, the [Ca2+]c started to rise from submicromolar concentrations (0.3 µM) and the peak value was obtained at near 100 µM with a half activation dose of 3.9±0.1 µM. To investigate what kinds of receptors or ion channels are involved, we used specific antagonists for various glutamate receptors and VOCCs. The representative traces are shown in Fig. 5A where the black curves are the [Ca2+]c rises in response to 100 µM glutamate and the red curves are those obtained in the presence of specific antagonists in the same cells. Interestingly the glutamate-induced [Ca2+]c rise was most significantly inhibited by the specific AMPA/kainate receptor antagonist, CNQX (30 µM, n=16). The NMDA receptor antagonist, AP-5, and VOCC antagonists slightly inhibited the glutamate-mediated g [Ca2+]c rises (n=13, P<0.05 in paired student’s t-test). In the case of nifedipine, this L-type Ca2+ channel antagonist did not inhibit as much as it did in the KCl-induced [Ca2+]c rise (Fig. 3F). B 80 300 glutamate (µM) e d 600 0.1 200 nM b 900 0 100 µM Glutamate a 1200 30 s SNc dopamine cells (Fig. 3). To eliminate SAPs we added 0.5 µM TTX to all the solutions and depolarized membrane potential with a brief exposure (12 seconds) to 60 mM KCl. After the first stimulation of cells with 60 mM KCl, we stimulated cells again with the same KCl solution in the presence of various VOCC antagonists, such as nifedipine (Ltype Ca2+ channel antagonist, Fig. 3A), ω-conotoxin GVIA (Ntype Ca2+ channel antagonist, Fig. 3B), and ω-agatoxin IVA, (P/Q-type Ca2+ channel antagonist, Fig. 3C), a cocktail of the three antagonists (Fig. 3D), or 100 µM Cd2+ (Fig. 3E). The average block effects were summarized in Fig. 3F. In the SNc dopamine neurons, despite the dominant contribution of L-type Ca2+ channels to the rise in [Ca2+]c, other channels appear to participate in the KCl-induced [Ca2+]c rises. A [Ca2+]c (nM) Fig. 4. Dose dependence of glutamate-induced [Ca2+]c rises. (A) The [Ca2+]c rises in response to different glutamate concentrations in the same cell. (B) The concentration-response curve of the glutamate-induced [Ca2+]c rises. The concentration of the half-maximum [Ca2+]c rise (EC50) is 3.9±1.0 µM. All the solutions contain 1 µM glycine. Each point represents the average of 4-20 cells (mean ± s.e.m.). 200 nM 1500 Fig. 5. Glutamate-induced [Ca2+]c increases. (A) Effects of antagonists for ionotropic glutamate receptors (a,b) and VOCCs (c-g) on the 100 µM glutamate-elicited [Ca2+]c rises. The black curves are [Ca2+]c rises in response to 100 µM glutamate and the red curves are [Ca2+]c rises in the presence of specific antagonists. (a) 30 µM CNQX; (b) 50 µM AP5; (c) 10 µM nifedipine; (d) 1 µM ω-conotoxin GVIA; (e) 1 µM ω-agatoxin IVA; (f) cocktail of the above VOCC antagonists; (g) 200 µM CdCl2. (B) Summary of a-f experiments (means ± s.e.m., n). CNQX, 39.5±6.7% of control, n=16; AP-5, 93.3±3.9%, n=13; nifedipine, 82.3±3.9%, n=7; ω-conotoxin GVIA, 93.9±0.3%, n=5; ω-agatoxin IVA, 100%, n=3; cocktail of the above three antagonists, 67.5±2.4%, n=3; CdCl2, 27.7±4.8%, n=6. All the solutions contain 1 µM glycine. *Significant difference (paired student t-test, P<0.05). 2670 Journal of Cell Science 116 (13) 10 µM Nif [Ca2+ ]c (nM) A 200 calcium-free 0 0 [Ca2+ ]c (nM) B 10 min a 100 µM AMPA 1 µM ω-cono GVIA b 1 µM ω-aga IVA 200 0 0 min 10 ω-aga IVA ω-cono GVIA Nifedipine AMPA C Relative [Ca2+]c change (%) AMPA did not appear to directly stimulate intracellular Ca2+ stores. Therefore, it is likely that, if cells were stimulated with high concentrations of glutamate, the SNc dopamine neurons would raise [Ca2+]c through mainly Ca2+-permeable AMPA/kainate receptors (Pellegrini-Giampietro et al., 1997; Metzger et al., 2000) and some VOCCs. Because the intracellular endoplasmic reticulum Ca2+ store is the most important source of Ca2+ for many kind of cells, such as cardiac, skeletal and pancreatic acinar cells (Park et al., 2000; Csordas et al., 2001; Bers, 2002), we tested how this store contributes to the [Ca2+]c rises in SNc dopamine neurons. To this end, we used antagonists for ionotropic glutamate receptors and stimulated cells with glutamate. In this case, the [Ca2+]c rise reached 31.5±3.5% (n=6) of the maximal [Ca2+]c increase that was obtained with 100 µM glutamate (Fig. 7A). In the Ca2+-free solution, glutamate also increased [Ca2+]c by 19.3±4.2% of control levels (n=5, Fig. 7B,D). When we directly stimulated cells with a group 1 mGluR agonist, DHPG, the [Ca2+]c increased to a level similar to that shown in the glutamate-stimulated cells in the Ca2+-free solution (Fig. 7C,D). Fig. 6. AMPA-induced [Ca2+]c increases. 100 µM AMPA was applied at each point indicated by an arrowhead for 12 seconds. (A) Nifedipine (10 µM) inhibited the 100 µM AMPA-elicited [Ca2+]c rises (n=4). In the Ca2+-free solution, AMPA did not rise [Ca2+]c at all (n=3). (B) 1 µM ω-agatoxin IVA (n=2; b) but not 1 µM ωconotoxin GVIA (n=4; a) slightly inhibited the AMPA-induced [Ca2+]c rises. (C) Summary of the above data. Nifedipine, 67.7±12.5%, n=4; ω-conotoxin GVIA, 95.0±0.5%, n=4; ω-agatoxin IVA, 88.8±0.5%, n=2; *Significant difference (paired student t-test, P<0.05). Therefore, to further clarify the Ca2+ influx pathways blocked by CNQX, we directly stimulated AMPA/kainate receptors by adding AMPA to the bath. In this case, AMPA/kainate receptors and VOCCs would be selectively opened. As shown in Fig. 6, nifedipine and other VOCC antagonists did not inhibit the glutamate-induced [Ca2+]c rises as much as they did in the KCl-stimulated cells (Fig. 3), suggesting that the AMPA-mediated [Ca2+]c rise is not solely mediated by VOCCs but involved with other pathways. When we added AMPA to a Ca2+-free solution, there was no detectable change in [Ca2+]c as shown in Fig. 6A (n=4). Thus Metabotropic glutamate receptor-mediated [Ca2+]c rises When we stimulated cells with DHPG in a long time scale, SNc showed a characteristic shape of the [Ca2+]c rise as shown in Fig. 8Aa. After the initial rapid [Ca2+]c rise and drop, the sustained [Ca2+]c elevation was observed in all cells tested (n=9). The initial peak was not affected by removal of extracellular Ca2+(Fig. 8Ab), but only blocked by a specific type 1 mGluR antagonist, CPCCOEt (100 µM, Fig. 9C). Moreover, the sustained [Ca2+]c elevation was completely abolished by removal of extracellular Ca2+ (Fig. 8Ab), suggesting two kinds of Ca2+ rising mechanisms by mGluR; the initial Ca2+ release from the intracellular Ca2+ stores and the sustained Ca2+ influx out of the cell. Interestingly this characteristic [Ca2+]c rise was reproduced by the application of glutamate at low concentrations. As shown in Fig. 8Ba,b, 1 or 3 µM glutamate reproduced a [Ca2+]c rise similar to that shown in Fig. 8Aa. In these cells, 100 µM CPCCOEt not only blocked the first component of the [Ca2+]c rise (Fig. 9C) but also effectively inhibited the second sustained component (Fig. 8Ba,b). CNQX (30 µM) or AP-5 (50 µM) had a minimal effect (Fig. 8Ba,b), indicating that at low glutamate concentrations mGluR are dominantly operating in the SNc dopamine neurons. Relative contributions of NMDA, AMPA and mGluR receptors In the previous experiments, we showed that glutamate at low concentrations mainly mimicked the DHPG-induced [Ca2+]c responses but glutamate at high concentrations gave rise to [Ca2+]c mainly through activation of the AMPA/kainate receptors (Fig. 5). Thus, we thought that glutamate could differentially raise [Ca2+]c according to glutamate concentrations. Thus we examined how glutamate raises [Ca2+]c at different glutamate concentrations in the presence of specific antagonists for glutamate receptors such as CNQX, AP-5 and CPCCOEt. Fig. 9 shows the results, where the phenomenon we observed was clearly disclosed. At low Glutamate-mediated [Ca2+]c changes in dopamine neurons 100 µM glutamate 100 µM glutamate B 1200 1400 1000 1200 800 30 µM CNQX + 50 µM AP-5 600 400 200 [Ca2+]c (nM) 1000 800 600 0 15 20 25 0 Time (min) 1000 800 30 µM DHPG 400 200 0 5 10 15 20 25 Time (min) Relative [Ca2+]c change (%) D 1200 0 10 15 20 25 Time (min) 100 µM glutamate 600 5 100 80 60 40 DHPG 10 DHPG Ca2+-free 5 Glutamate 2+ Ca -free 0 [Ca2+]c (nM) Ca -free 200 0 C1400 2+ 400 CNQX + AP5 [Ca2+]c (nM) A 2671 20 0 Fig. 7. The [Ca2+]c increases by the activation of metabotropic glutamate receptors. (A) 100 µM glutamate-mediated [Ca2+]c rises in the absence/presence of ionotropic glutamate receptor antagonists (30 µM CNQX + 50 µM AP-5, n=6). (B) 100 µM glutamate-mediated [Ca2+]c rises with and without calcium in the bath solution (n=5). (C) 100 µM glutamate- and DHPG (mGluR agonist)-mediated [Ca2+]c increases (n=10). (D) Summary of the above data. 100% (control) means the peak values of [Ca2+]c increases to 100 µM glutamate. CNQX and AP-5, 31.5±3.5% of control, n=6; glutamate in Ca2+-free solution, 23.7 ± 3.5%, n=5; DHPG in Ca2+-free solution, 19.3±4.2%, n=5; DHPG (31.3±1.9%, n=10. All the solutions contain 1 µM glycine. glutamate concentrations (0.3-3 µM), CPCCOEt dominantly inhibited the glutamate-elicited [Ca2+]c rises, whereas at high glutamate concentrations CNQX effectively blocked the [Ca2+]c rises. The blocking effect of AP-5 on the glutamateinduced [Ca2+]c rise was not much concentration-dependent but largest at near 3 µM glutamate. In Fig. 9D, we summarized relative contributions of each glutamate receptor to the glutamate-induced [Ca2+]c rises at various concentrations by analyzing antagonist effects in several cells. Contribution of spontaneous firing to glutamatemediated [Ca2+]c rises In Fig. 2, we show that SAPs were important in maintaining the resting [Ca2+]c, and changes in SAP frequency could affect the level of [Ca2+]c. Thus we tried to investigate how SAP frequency affects [Ca2+]c. To raise spontaneous firing frequency without activating glutamate receptors, we gradually raised KCl concentration from 5 mM until SAPs disappeared, and measured [Ca2+]c at the same time. As shown in Fig. 10 (left), the elevation of KCl in a bath solution raised the frequency of spontaneous firing as well as [Ca2+]c. The frequency reached a peak at 10-12 mM KCl and the SAPs disappeared at 15 mM KCl, probably as a result of too much depolarization of membrane potential. It also suggests that SAPs can be generated between optimal ranges of the membrane potential. One interesting finding in this figure is that the [Ca2+]c was rising while the frequency was increasing. However, as soon as SAPs disappeared, after addition of 15 mM KCl, the [Ca2+]c decreased, despite the higher KCl concentration. This suggests that SAP is more important in [Ca2+]c rises than simple depolarization of membrane potential in our experimental conditions. To test how the steady state depolarization (due to KCl) activate VOCCs, we raised KCl concentration in the presence of 0.5 µM TTX. But in this case, the [Ca2+]c rise was much smaller than that observed in spontaneously firing cells (data not shown, n=5). Therefore it is likely that the frequency of SAPs is an important factor in the regulation of [Ca2+]c in SNc dopamine neurons. Next, we tested how glutamate changes [Ca2+]c and spontaneous firing frequency. Although glutamate is known to raise spontaneous firing activity (Meltzer et al., 1997), the concentration dependence of glutamate has not been reported yet. On the right-hand side of Fig. 10, we gradually raised glutamate concentration in a bath, starting at 0.3 µM. Surprisingly glutamate at very low concentrations strongly raised the SAP frequency as well as [Ca2+]c (n=6). Glutamate very sensitively and dramatically increased the SAP frequency compared with the increase caused by KCl elevation (Figs 3, 10). Moreover, the [Ca2+]c rise by glutamate in the presence of TTX was much smaller (data not shown, n=5), suggesting that spontaneous firing is also an important factor in contributing to the [Ca2+]c rise in the SNc dopamine cells. Thus we could 2672 A 10 µM DHPG b 10 µM DHPG Fig. 8. Two phases of the [Ca2+]c rise by activation of the metabotropic glutamate receptors. (A) Application of 10 µM DHPG (a) elicited characteristic [Ca2+]c rises: the initial transient Ca2+ rise and long-sustained [Ca2+]c elevation (n=7). Disappearance of the later persistent [Ca2+]c elevation (b) under the Ca2+free solution (n=7). (B) Effects of glutamate receptor antagonists on the late phase of glutamate-induced [Ca2+]c increases. All the solutions contain 1 µM glycine. Note the glutamate concentration. 50 nM a Journal of Cell Science 116 (13) 1 min near 100 µM (Fig. 4). When cells are stimulated with high glutamate 2+ influx is concentrations, the main Ca Ca2+ free mediated by the activation of AMPA/kainate receptors and VOCCs (Figs 5, 6). The intracellular Ca2+ stores also contribute to the glutamate-induced [Ca2+]c rise by about 30% of the maximal response (Fig. 7). The 3 µM glutamate 1 µM glutamate activation of metabotropic glutamate receptors leads to the initial rapid Ca2+ rise and later sustained elevation of [Ca2+]c (Fig. 8). In Fig. 9, we clearly demonstrate that different glutamate receptors differentially 100 µM CPCCOEt participate in [Ca2+]c dynamics according to glutamate concentrations. At low glutamate 100 µM CPCCOEt concentrations, the [Ca2+]c is mainly 30 µM CNQX 30 µM CNQX regulated by activation of mGluR (Figs 8, 9), whereas at high glutamate 50 µM AP-5 50 µM AP-5 concentrations it mostly depends on the activation of AMPA/kainate receptors (Figs 5, 6, 9). Moreover, the spontaneous firing rate of the cell is another key factor 1 min 1 min governing the [Ca2+]c level in the SNc dopamine neurons, whose sensitivity is conclude that glutamate at low concentrations not only elevates modulated at the level of ambient glutamate concentrations [Ca2+]c by activation of mGluR but also gives rise to [Ca2+]c (Figs 2, 4, 10). by enhancing SAP frequency. B a 200 nM 100 nM b Discussion In this study, we have investigated the [Ca2+]c dynamics in spontaneously firing single dopamine neurons and dissected the Ca2+ influx pathways relating to glutamate receptors. The freshly isolated dopamine neurons from SNc showed regular SAPs of 2-3 Hz (Figs 2, 10) and their frequency was highly affected by submicromolar concentrations of glutamate, which is similar to the ambient glutamate concentration reported in vivo (Lerma et al., 1986; Herrera-Marschit et al., 1996). The frequency of SAPs is closely related to the level of [Ca2+]c and the resting [Ca2+]c is slightly elevated due to the resting level of spontaneous firing (Figs 2, 10). During this firing, VOCCs are partially activated at resting condition and allow Ca2+ influx out of the cells (Figs 2, 3). Among the many VOCCs, L-type Ca2+ channels appear to contribute mostly to the depolarization-induced Ca2+ rises in the SNc dopamine neurons (Fig. 3). Glutamate, surprisingly, could raise [Ca2+]c at submicromolar concentrations and peak at Cell types in substantia nigra pars compacta The SNc dopamine neurons are well known to have multiple neurites and a large cell body (Juraska et al., 1977; Grace and Bunny, 1983a; Hajos and Greenfield, 1993; Cardozo and Bean, 1995; Kitai et al., 1999). SNc also contains non-dopaminergic neurons, which are different in shape and electrophysiological properties from the dopamine neurons. When we isolated cells from the SNc area, which we deliberately tried to confine, 79% of the isolated cells were large multipolar cells and among them 91% were dopaminergic neurons (Fig. 1). If we include three TH+ cells among the atypical cells, the total dopaminergic neurons in SNc would be 76% of the dissociated cells. This suggests that the majority of cells within the SNc area are dopaminergic but a substantial portion of the cells in SNc are not dopaminergic. However, in this calculation we could not exclude the possibilities that some of cells may originate from the nearby areas of the SNc during the isolation procedures and some cells vulnerable to the dissociation procedures would be selectively lost. Glutamate-mediated [Ca2+]c changes in dopamine neurons A 10 µM glutamate B 100 µM glutamate 2673 100 µM 10 µM 3 µM 3 µM 200 nM 0.3 µM 1 µM 1 µM 0.3 µM 2 min C 10 µM 100 µM glutamate 3 µM 1 µM 0.3 µM CPCCOEt D Relative Contribution(%) AP-5 CNQX mGluR NMDAR AMPAR 100 80 60 40 20 0 0.3 µM 1 µM 3 µM 10 µM 100 µM Glutamate Fig. 9. Inhibition of [Ca2+]c rises by antagonists for AMPA/kainate, NMDA, and metabotropic glutamate receptors at different glutamate concentrations in SNc dopamine neurons. The black curves indicate the glutamate-induced [Ca2+]c rises at the indicated concentrations, and the red curves indicate the glutamate-induced [Ca2+]c rises in the presence of 30 µM CNQX (A), 50 µM AP-5 (B), 100 µM CPCCOEt (C), respectively. (D) Different contributions of AMPA/kainate, NMDA, and metabotropic glutamate receptors to the [Ca2+]c rises according to glutamate concentrations. Each point was calculated by analysis of the data from 4-20 cells. All the solutions contain 1 µM glycine. Contribution of VOCCs to [Ca2+]c rises in dopamine neurons In SNc dopamine neurons, VOCCs appear to participate in pacemaker-like oscillations of membrane potential and many types of VOCCs are reported to be present in SNc neurons (Cardozo and Bean, 1995; Kang and Kitai, 1993; Takada et al., 2001). However it has not been reported what types of VOCCs are important in [Ca2+]c homeostasis. According to the wholecell patch-clamp recordings in acutely isolated cells from 310-day old rats (Cardozo and Bean, 1995), P/Q-type, N-type and L-type VOCCs are near equally activated by strong depolarization pulses. However, the immunohistochemical data obtained from brains from the adult Wistar rats (Takada et al., 2001) show the predominant presence of L-type Ca2+ channels at dendrites and cell bodies of the dopamine neurons. Our experimental data obtained from 7-16-day old rats indicate that L-type VOCCs are a major contributor to [Ca2+]c changes in the cell body. They are closer to the data obtained from the adult rats. Roles of NMDA-, AMPA/kainate, and metabotropic glutamate receptors in [Ca2+]c dynamics Neurons in the brain can be exposed to highly variable concentrations of glutamate and the glutamate concentration in the cerebrospinal fluid is as high as several micromolar concentrations (Lerma et al., 1986; Sah et al., 1989; HerreraMarschitz et al., 1996). Although resting neurons appear to be exposed more or less to a micromolar concentration due to strong buffering activities of glial cells, some glutamate receptors are reported to be tonically active at resting glutamate concentrations (Sah et al., 1989). Since maintenance of basal Ca2+ concentrations could be essential in neuronal functions, understanding of glutamate-mediated Ca2+ influx pathways would be important. In SNc dopamine neurons, modulation of firing patterns by glutamate is relatively well-studied (Metzler et al., 1997; Kitai et al., 1999) but the Ca2+ signals relating to glutamate have not been thoroughly studied yet. In this experiment, we have dissected glutamate-mediated Ca2+ influx pathways at variable levels of glutamate concentrations. One interesting finding is that the Ca2+ influx pathway, which is closely linked with mGluR, is operating at low glutamate concentrations and this pathway may be tonically active and contributes to resting [Ca2+]c. In Fig. 8, we showed the two components of the mGluR-linked [Ca2+]c rises. Among them the fast component is clearly attributed to the intracellular Ca2+ stores and probably plays an important role in acute responses. This is in agreement with the reports that SNc neurons have the inositol 1,4,5-trisphophate mediated Ca2+ responses and it acts as an inhibitory signals (Fiorillo and 2674 Journal of Cell Science 116 (13) A glutamate ( µ M) KCl (mM) 5 10 7 12 15 0.3 5 1 3 10 4 (mV) 0 100 sec B Frequency (Hz) 3 8 5 6 12 0.3 15 1 5 4 2 0 0 C 10 7 100 200 300 400 500 TIME (sec) 600 10 500 [Ca 2+ ]c (nM) 10 Fig. 10. Glutamate at submicromolar concentrations increases the frequency of spontaneous firing and [Ca2+]c in SNc dopamine neurons. (A) Spontaneous firing activities of the dopamine cell were recorded in the cell-attached current-clamp configuration. The concentration of KCl was gradually increased and thereafter the glutamate concentration increased too. (B) Frequency plot of the data in A. When stimulated with KCl, a maximum frequency was obtained at 1012 mM. Glutamate more dramatically raised the firing frequency dose-dependently but the spontaneous firing suddenly disappeared after exposure to 10 µM glutamate. (C) The [Ca2+]c changes from the same cell. The increased spontaneous firing reflects the increase in [Ca2+]c. Glutamate has a much stronger effect on the spontaneous firing and [Ca2+]c than KCl. The solution contains 1 µM glycine. 3 400 1 300 5 200 10 7 12 15 5 0.3 100 0 0 100 200 300 400 500 TIME (sec) William, 1998; Morikawa et al., 2000; Seutin et al., 2000). By contrast, the second component, persistent elevation of [Ca2+]c, appears to be important in keeping [Ca2+]c at some levels elevated in vivo, since submicromolar concentrations of glutamate elicited the responses and interstitial fluid contains glutamate at micromolar concentrations (Lerma et al., 1986; Herrera-Marschitz et al., 1996). For this Ca2+ influx pathway, store-operated Ca2+ channels or/and unidentified Ca2+ permeable channels would be responsible (Guatteo et al., 1999; Fagni et al., 2000). Regarding AMPA/kainate receptors in dopamine neurons, we have somewhat different results from the general view that NMDA receptors are more important than AMPA/kainate receptors in the glutamate-mediated Ca2+ influxes. In Fig. 5, we showed that the contribution of NMDA receptors to the [Ca2+]c rise, when stimulated with glutamate at high concentrations, was relatively smaller than that of AMPA/kainate receptors. However, recently there have been accumulating reports that [Ca2+]c can be increased through AMPA/kainate receptors themselves and some types of AMPA/kainate receptors are highly Ca2+-permeable and play a role in AMPA-mediated neurotoxicity (Metzger et al., 2000; Pellegrini-Giampietro et al., 1997). Among them, it has been reported that joro spider toxin can selectively inhibit Ca2+permeable AMPA receptors (Blaschke et al., 1993). In Fig. 5, we showed that AMPA/kainate receptors are most important in the [Ca2+]c rise in response to the high glutamate concentration. Thus we suspected the presence of Ca2+permeable AMPA receptors in SNc dopamine neurons. However, when we tested joro spider toxin in our experimental conditions, it did not inhibit the AMPA-mediated Ca2+ influx (data not shown, n=7), suggesting the presence of joro spider toxin-resistant Ca2+-permeable AMPA receptors (Meucci et al., 1996; Meucci and Miller, 1998) or other unknown pathways. Spontaneous firing and Ca2+ signals Glutamate not only conveys electrical signals at 600 synapses but also regulates spontaneous firing patterns in SNc dopamine neurons. Thus the roles of many kinds of glutamate receptors have long been studied. So far, all the glutamate receptors such as NMDA, AMPA/kainate receptors, and mGluR are known to be more or less involved in modulating spike patterns or shapes of spikes (Pucak and Grace, 1994; Metzler et al., 1997; Kitai et al., 1999). However, to our knowledge, it is not clear what concentration of glutamate could modulate the spontaneous firing or change spike patterns. Surprisingly we found that glutamate at submicromolar concentrations strongly increases the firing rate of SNc neurons. Since firing rate (Figs 2, 10) is directly related to the level of [Ca2+]c, glutamate at low concentrations could effectively maintain [Ca2+]c at slightly elevated levels. Generally, in pancreatic acinar cells, immune cells, or cardiac myocytes, the basal elevation or continuous oscillations of [Ca2+]c are recently regarded as a wakeup signal in which it activates subcellular organelles to boost metabolic processes or trigger production of antibodies (Duchen, 1999; Bers, 2002; Csordas et al., 2001; Lewis, 2001; Park et al., 2000; Park et al., 2001). However, in the SNc dopamine neurons the physiological role of the slightly elevated [Ca2+]c at resting conditions, as a result of spontaneous firing, remains elusive but it may play an important role in many biological processes. This work was supported by the Neurobiology Research Program from the Korea Ministry of Science and Technology (M1-0108-000027) and by the Korea Science and Engineering Foundation (KOSEF) grant through the Medical Research Center for Regulation of Neuronal Cell Excitability at Sungkyunkwan University. References Amini, B., Clark, J. W. and Canavier, C. C. (1999). Calcium dynamics underlying pacemaker-like and burst firing oscillations in midbrain Glutamate-mediated [Ca2+]c changes in dopamine neurons dopaminergic neurons: a computational study. J. Neurophysiol. 82, 22492261. Benveniste, H., Drejer, J., Schousboe, A. and Diemer, N. H. (1984). Elevation of the extracellular concentrations of glutamate and aspartate in rat hippocampus during transient cerebral ischemia monitored by intracerebral microdialysis. J. Neurochem. 43, 1369-1374. Berridge, M. J. (1998). Neuronal calcium signaling. Neuron 21, 13-26. Bers, D. M. (2002). Cardiac excitation-contraction coupling. Nature 415, 198205. Blaschke, M., Keller, B. U., Rivosecchi, R., Hollmann, M., Heinemann, S. and Konnerth, A. (1993). A single amino acid determines the subunitspecific spider toxin block of alpha-amino-3-hydroxy-5-methylisoxazole-4propionate/kainate receptor channels. Proc. Natl. Acad. Sci. USA 90, 65286532. Cardozo, D. L. (1993). Midbrain dopaminergic neurons from postnatal rat in long-term primary culture. Neuroscience 56, 409-421. Cardozo, D. L. and Bean, B. P. (1995). Voltage-dependent calcium channels in rat midbrain dopamine neurons: modulation by dopamine and GABAB receptors. J. Neurophysiol. 74, 1137-1148. Chen, B. T. and Rice, M. E. (2001). Novel Ca2+ dependence and time course of somatodendritic dopamine release: Substantia nigra versus striatum. J. Neurosci. 21, 7841-7847. Chung, J., Chang, S., Kim, Y. and Shin, H. (2000). Zinc increases the excitability of dopaminergic neurons in rat substantia nigra. Neurosci. Lett. 286, 183-186. Collingridge, G. L. and Singer, W. (1990). Excitatory amino acid receptors and synaptic plasticity. Trends. Pharmacol. Sci. 11, 290-296. Csordas, G., Thomas, A. P. and Hajnoczky, G. (2001). Calcium signal transmission between ryanodine receptors and mitochondria in cardiac muscle. Trends. Cardiovasc. Med. 11, 269-275. Duchen, M. R. (1999). Contributions of mitochondria to animal physiology: from homeostatic sensor to calcium signalling and cell death. J. Physiol. 516, 1-17. Fagni, L., Chavis, P., Ango, F. and Bockaert, J. (2000). Complex interactions between mGluRs, intracellular Ca2+ stores and ion channels in neurons. Trends. Neurosci. 23, 80-88. Fiorillo, C. D. and Williams, J. T. (1998). Glutamate mediates an inhibitory postsynaptic potential in dopamine neurons. Nature 394, 78-82. Gallin, W. J. and Greenberg, M. E. (1995). Calcium regulation of gene expression in neurons: the mode of entry matters. Curr. Opin. Neurobiol. 5, 367-374. Ginty, D. D. (1997). Calcium regulation of gene expression: isn’t that spatial? Neuron 8, 183-186. Grace, A. A. (1988). In vivo and in vitro intracellular recordings from rat midbrain dopamine neurons. Ann. N. Y. Acad. Sci. 537, 51-76. Grace, A. A. and Bunney, B. S. (1983a). Intracellular and extracellular electophysiology of nigral dopaminergic neurons – 1. identification and characterization. Neuroscience 10, 301-315. Grace, A. A. and Bunney, B. S. (1983b). Intracellular and extracellular electophysiology of nigral dopaminergic neurons – 2. action potential generating mechanisms and morphological correlates. Neuroscience 10, 317-331. Grillner, P. and Mercuri, N. B. (2002). Intrinsic membrane properties and synaptic inputs regulating the firing activity of the dopamine neurons. Behav. Brain. Res. 130, 149-169. Guatteo, E., Mercuri, N. B., Bernardi, G. and Knopfel, T. (1999). Group I metabotropic glutamate receptors mediate an inward current in rat substantia nigra dopamine neurons that is independent from calcium mobilization. J. Neurophysiol. 82, 1974-1981. Hajos, M. and Greenfield, S. A. (1993). Topographic heterogeneity of substantia nigra neurons: diversity in intrinsic membrane properties and synaptic inputs. Neuroscience 55, 919-934. Herrera-Marschitz, M., You, Z. B., Goiny, M., Meana, J. J., Silveira, R., Godukhin, O. V., Chen, Y., Espinoza, S., Pettersson, E., Loidl, C. F. et al. (1996). On the origin of extracellular glutamate levels monitored in the basal ganglia of the rat by in vivo microdialysis. J. Neurochem. 66, 17261735. Hounsgaard, J., Nedergaard, S. and Greenfield, S. A. (1992). Electrophysiological localization of distinct calcium potentials at selective somatodendritic sites in the substantia nigra. Neuroscience 50, 513-518. Jaffe, E. H., Marty, A., Schulte, A. and Chow, R. H. (1998). Extrasynaptic vesicular transmitter release from the somata of substantia nigra neurons in rat midbrain slices. J. Neurosci. 18, 3548-3553. Juraska, J. M., Wilson, C. J. and Groves, P. M. (1977). The substantia nigra of the rat: a Golgi study. J. Comp. Neurol. 172, 585-600. 2675 Kang. Y. and Kitai, S. T. (1993). A whole cell patch-clamp study on the pacemaker potential in dopaminergic neurons of rat substantia nigra compacta. Neurosci. Res. 18, 209-221. Kitai, S. T., Shepard, P. D., Callaway, J. C. and Scroggs, R. (1999). Afferent modulation of dopamine neuron firing patterns. Curr. Opin. Neurobiol. 9, 690-697. Lerma, J., Herranz, A. S., Herreras, O., Abraira, V. and Martin, D. R. (1986). In vivo determination of extracellular concentration of amino acids in the rat hippocampus. A method based on brain dialysis and computerized analysis. Brain Res. 384, 145-155. Lewis, R. S. (2001). Calcium signaling mechanisms in T lymphocytes. Annu. Rev. Immunol. 19, 497-521. Mayer, M. L. and Miller, R. J. (1990). Excitatory amino acid receptors, second messengers and regulation of intracellular Ca2+ in mammalian neurons. Trends Pharmacol. Sci. 11, 254-260. Meltzer, L. T., Christoffersen, C. L. and Serpa, K. A. (1997). Modulation of dopamine neuronal activity by glutamate receptor subtypes. Neurosci. Behav. Rev. 21, 511-518. Metzger, F., Kulik, A., Sendtner, M. and Ballanui, K. (2000). Contribution of Ca2+-permeable AMPA/KA receptors to glutamate-induced Ca2+ rise in embryonic lumbar motoneurons in situ. J. Neurophysiol. 83, 50-59. Meucci, O. and Miller, R. J. (1998). Dissociation between the Joro spider toxin sensitivity of recombinant alpha-amino-3-hydroxy-5-methyl-4isoxazolepropionic acid receptors and their ability to increase intracellular calcium. Neuropharmacology 37, 1431-1443. Meucci, O., Fatatis, A., Holzwarth, J. A. and Miller, R. J. (1996). Developmental regulation of the toxin sensitivity of Ca2+-permeable AMPA receptors in cortical glia. J. Neurosci. 16, 519-530. Morikawa, H., Imani, F., Khodakhah, K. and Williams, J. T. (2000). Inositol 1,4,5-triphosphate-evoked responses in midbrain dopamine neurons. J. Neurosci. 20, RC1031-RC1035. Nakanishi, S. (1992). Molecular diversity of glutamate receptors and implications for brain function. Science 258, 597-603. Nedergaard, S., Bolam, J. P. and Greenfield, S. A. (1988). Facilitation of a dendritic calcium conductance by 5-hydroxytryptamine in the substantia nigra. Nature 333, 174-177. Neher, E. and Augustine, G. J. (1992). Calcium gradients and buffers in bovine chromaffin cells. J. Physiol. 450, 273-301. Obrenovitch, T. P. and Urenjak, J. (1997). Altered glutamatergic transmission in neurological disorders: from high extracellular glutamate to excessive synaptic efficacy. Prog. Neurobiol. 87, 39-87. Overton, P. G. and Clark, D. (1997). Burst firing in midbrain dopaminergic neurons. Brain Res. Rev. 25, 312-334. Park, M. K., Petersen, O. H. and Tepikin, A. V. (2000). The endoplasmic reticulum as one continuous Ca2+ pool: visualization of rapid Ca2+ movements and equilibration. EMBO J. 19, 5729-5739. Park, M. K., Ashby, M. C., Erdemli, G., Petersen, O. H. and Tepikin, A. V. (2001). Perinuclear, perigranular and sub-plasmalemmal mitochondria have distinct functions in the regulation of cellular calcium transport. EMBO J. 20, 1863-1874. Park, M. K., Lee, K. K. and Uhm, D. Y. (2002). Slow depletion of endoplasmic reticulum Ca2+ stores and block of store-operated Ca2+ channels by 2-aminoethoxydiphenyl borate in mouse pancreatic acinar cells. Naunyn Schmiedebergs Arch. Pharmacol. 365, 399-405. Pellegrini-Giampietro, D. E., Gorter, J. A., Bennett, M. and Zukin, R. S. (1997). The GluR2 (GluR-B) hypothesis: Ca2+-permeable AMPA receptors in neurological disorders. Trends. Neurosci. 20, 464-470. Pucak, M. L. and Grace, A. A. (1994). Regulation of substantia nigra dopamine neurons. Crit. Rev. Neurobiol. 9, 67-89. Sah, P., Hestrin, S. and Nicoll, R. A. (1989). Tonic activation of NMDA receptors by ambient glutamate enhances excitability of neurons. Science 246, 815-818. Sattler, R. and Tymianski, M. (2000). Molecular mechanisms of calciumdependent excitotoxicity. J. Mol. Med. 78, 3-13. Seutin, V., Mkahli, F., Massotte, L. and Dresse, A. (2000). Calcium release from internal stores is required for the generation of spontaneous hyperpolarizations in dopaminergic neurons of neonatal rats. J. Neurophysiol. 83, 192-197. Takada, M., Kang, Y. and Imanishi, M. (2001). Immunohistochemical localization of voltage-gated calcium channels in substantia nigra dopamine neurons. Eur. J. Neurosci. 13, 757-762. Uchida, S., Akaike, A. and Nebekura, J. (2002). Dopamine activates inward rectifier K+ channel in acutely dissociated rat substantia nigra neurons. Neuropharmacology 39, 191-201.