* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Seizure, neurotransmitter release, and gene expression are closely
Biochemistry of Alzheimer's disease wikipedia , lookup
Synaptic gating wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Nervous system network models wikipedia , lookup
Biology of depression wikipedia , lookup
Limbic system wikipedia , lookup
Environmental enrichment wikipedia , lookup
NMDA receptor wikipedia , lookup
Neuroinformatics wikipedia , lookup
Neuroplasticity wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
Subventricular zone wikipedia , lookup
Synaptogenesis wikipedia , lookup
Neurotransmitter wikipedia , lookup
Signal transduction wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Haemodynamic response wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Neuroanatomy wikipedia , lookup
Aging brain wikipedia , lookup
Neuroeconomics wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Basal ganglia wikipedia , lookup
Optogenetics wikipedia , lookup
Metastability in the brain wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Molecular neuroscience wikipedia , lookup
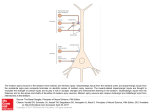
Epilepsy Research 55 (2003) 117–129 Seizure, neurotransmitter release, and gene expression are closely related in the striatum of 4-aminopyridine-treated rats Annamária Kovács a , András Mihály a,∗ , Ágnes Komáromi b , Erika Gyengési c , Magdolna Szente c , Roland Weiczner a , Beáta Krisztin-Péva a , Gyula Szabó b , Gyula Telegdy b a Department of Anatomy, Faculty of Medicine, University of Szeged, P.O. Box 427, H-6701 Szeged, Hungary b Department of Pathophysiology, University of Szeged, P.O. Box 427, H-6701 Szeged, Hungary c Department of Comparative Physiology, University of Szeged, P.O. Box 427, H-6701 Szeged, Hungary Received 22 February 2003; received in revised form 18 June 2003; accepted 22 June 2003 Abstract The present experiments aimed to compare the length of seizure activity with the time-related increase of transmitter release and the induction of c-fos gene expression in the striatum of the rat. Anesthetized Wistar rats were intraperitoneally treated with 7 mg/kg 4-aminopyridine, and the transmitter levels in the striatum were measured by means of in vivo microdialysis, 30, 60, 90, 120, and 150 min following the treatment. Striatal and neocortical electric activity was monitored with depth and surface electrodes, respectively. The expression level of the c-fos gene was estimated by counting the striatal c-fos-immunostained cell nuclei at the time intervals of the microdialysis. 4-Aminopyridine elicited high-frequency seizure discharges in the EEG and significantly increased glutamate, aspartate, GABA, serotonin, noradrenaline, and dopamine levels in the extracellular dialysates. The number of c-fos-stained cell nuclei in the striatum displayed a prolonged increase, showing significantly elevated numbers throughout the experiment. The increase of c-fos expression in time correlated best with the increase of glutamate release, which was also significantly elevated at every sampling time. The GABA release, culminating at 60 min after the seizure onset, correlated best with the cessation of the electrographic seizure. Aspartate, norepinephrine, serotonin, and dopamine displayed transient but significant elevations. We conclude that glutamate plays the essential role (most probably through ionotropic and metabotropic receptors) in the extracellular signaling, which eventually leads to intracellular cascades and c-fos gene expression in the striatum during convulsions. © 2003 Elsevier B.V. All rights reserved. Keywords: 4-Aminopyridine; Glutamate; Seizure; c-fos; Seizure-related glutamate release Abbreviations: 4-AP, 4-aminopyridine; ARG, arginine; ASP, asparatic acid; CIT, citrulline; DA, dopamine; DOPA, 3,4-dihydroxyphenylalanine; DOPAC, 3,4-dihydroxyphenylacetaldehyde; GABA, 4-aminobutyric acid; GLU, glutamic acid; 5-HIAA, 5-hydroxyindole-3-acetate; 5-HT, serotonin (5-hydroxytryptamine); HVA, homovanillic acid; ITF, inducible transcription factor; 3-MT, 3-methoxytyramine; NE, norepinephrine; NMDA, N-methyl-d-aspartate; NO, nitric oxide; NOS, nitric oxide synthase ∗ Corresponding author. Tel.: +36-62-545-670; fax: +36-62-545-707. E-mail address: [email protected] (A. Mihály). 0920-1211/$ – see front matter © 2003 Elsevier B.V. All rights reserved. doi:10.1016/S0920-1211(03)00113-X 118 A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 1. Introduction Excitatory amino acids (EAAs) such as glutamic acid and aspartic acid take part in the induction, maintenance, and propagation of seizures and mediate some of the neuropathological effects of neuronal hyperactivity (Sloviter and Dempster, 1985; Peña and Tapia, 2000). Glutamate (GLU) and aspartate (ASP) are epileptogenic and excitotoxic to nerve cells both in vitro and in vivo (Whetsell, 1996). The release of these EAAs from the nerve terminals may be Ca2+ -dependent or -independent, although the mechanisms are not fully understood. Extracellular K+ stimulates the EAA efflux from striatal neurons (Korf and Venema, 1985), but the GLU release may depend on many other molecular signals, such as the release of nitric oxide (NO; McNaught and Brown, 1998), the activation of presynaptic metabotropic GLU receptors (Cartmell and Schoepp, 2000), 4-aminobutyric acid (GABA) receptors and protein kinases (Perkinton and Sihra, 1999). It was believed for years that the main pathogenetic factor in epilepsies was the impairment of GABAergic transmission (Meldrum, 1975). It turned out later that inhibitory neurotransmission may play an important role in the generation and maintenance of certain seizure types in the hippocampus and in the corticothalamo-cortical circuits (Avoli, 1996; Engel, 1996). Recently, more fundamental processes underlying epileptic phenomena came into focus: the role of neuronal ion channels, and particularly K+ channels, which regulate neuronal excitability and are coupled to several transmitter systems (Hoffmann et al., 1997). Immunohistochemical studies have proved the presence of these channels on the processes of the neuron (Betancourt and Colom, 2000) and we are now only beginning to understand the significance of their local distribution and density on the surface of the neuron. 4-Aminopyridine (4-AP) is a convulsant which blocks some of the voltage-dependent neuronal potassium channels: the KA channel (or A channel), which regulates the spike frequency in postsynaptic structures; the KV channel (or delayed rectifier), involved in the repolarization phase of the action potential; and the KACh receptor-coupled channel (Alexander and Peters, 2000). The effects of 4-AP, therefore, include long presynaptic depolarization and increased transmitter release (Thesleff, 1980). It has been proved experimentally that 4-AP stimulates the release of GLU (Tibbs et al., 1989; Patterson et al., 1995), norepinephrine (NE; Versteeg et al., 1995), serotonin (5-HT; Schechter, 1997), and dopamine (DA; Bonnano et al., 2000). In the hippocampus, 4-AP increases the release of GLU, ASP, and GABA in a dose-dependent manner, and causes typical convulsive EEG changes (Peña and Tapia, 1999, 2000). In contrast, 4-AP did not increase the extracellular concentration of GLU in the striatum of freely moving rats (Segovia et al., 1997). The experimental convulsion evoked by 4-AP is followed by the appearance of the inducible transcription factor (ITF) c-fos in neuronal cell nuclei (Mihály et al., 1997, 2001; Szakács et al., 2003), and by an increased regional cerebral blood flow in those brain areas which participate in the spread of the seizures (Mihály et al., 2000). However, there is no direct in vivo evidence of a causal relationship between neurotransmitter release and the ITF expression induced by 4-AP. Accordingly, the present experiments were designed to measure the extracellular concentrations of neurotransmitters and their metabolites via an in vivo microdialysis technique and to detect the expression of c-fos by means of immunohistochemistry in the same brain area under the same experimental conditions. The striatum was chosen because it plays an important role in the motor coordination and has abundant cortical and subcortical connections which are strongly activated in motor seizures (Kusske, 1979; Hosokawa et al., 1983; Bonhaus et al., 1986). 2. Materials and methods Adult male Wistar rats weighing 200–250 g were housed in a light- and temperature-controlled room (lights on between 6:00 a.m. and 6:00 p.m.; 23 ◦ C) and had free access to food and water. The animals were kept and handled during the experiments in accordance with the standards of the Ethical Committee for the Protection of Animals in Research in Szeged University. The intraperitoneal (i.p.) injection of awake animals with 7 mg/kg 4-AP produced intense motor alterations (tremor, shivering and generalized tonic–clonic seizure; Mihály et al., 1990), and therefore the electrophysiological, microdialysis and immunohistochemical experiments were carried out in A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 separate groups of rats anesthetized with Nembutal (35 mg/kg), injected i.p. The body temperature was maintained at 37 ◦ C by using a heating pad. The anesthesia was maintained with repeated Nembutal (15 mg/kg) injections throughout the experiment. The experiments were performed in separate groups of animals, as listed below. 2.1. Electrophysiology Four animals were used for a direct test of the convulsive effect of 4-AP. The rats were anesthetized with Nembutal (35 mg/kg) and a steel needle depth recording electrode was inserted into the left striatum (interaural 9.48 mm, bregma 0.48 mm, depth 5.50 mm; Paxinos and Watson, 1998) with the help of a stereotaxic apparatus. The EEG electrodes were placed on the surface of the skull (see Fig. 2). The 7 mg/kg 4-AP was injected intraperitoneally to the anesthetized rats and recordings were made at 15-min intervals, with 10-min interruptions, for 150 min. The position of the depth electrode was checked after the experiment, similarly to that of the microdialysis probe. 2.2. Microdialysis Fourteen animals were used for microdialysis: seven were treated with 7 mg/kg 4-AP, while seven control animals were injected with physiological saline. The microdialysis probe was implanted into the right striatum (interaural 9.48 mm, bregma 0.48 mm; Paxinos and Watson, 1998) under Nembutal (35 mg/kg) anesthesia. The probe was located in the caudate–putamen region, and never penetrated the accumbens region or the olfactory tubercle (see Fig. 5). The probe was continuously perfused with modified Ringer solution (composition: 140 mM NaCl, 3.0 mM KCl, 1.2 mM Na2 HPO4 , 1.0 mM MgCl2 , 7.2 mM glucose, 1.2 mM CaCl2 , pH = 6.8–7.2) at a flow rate of 2 l/min, delivered by a CMA 100 microinjection pump. After a 2-h equilibrium period, perfusates were collected in polyethylene vials every 30 min throughout the experiments. Three baseline samples were obtained from every animal before the administration of 4-AP (7 mg/kg, i.p.). The average of these samples were depicted as 100% during the measurements, and the seizure-induced changes were expressed as percentages and handled as such in every animal, because 119 of the inter-animal variances. Immediately following sampling, dialysates were assayed for levels of GLU, ASP, arginine (ARG), citrulline (CIT), and GABA via high-performance liquid chromatography with electrochemical detection (HPLC-EC) following precolumn o-phthalaldehyde-sulfite derivatization at room temperature for 15 min. Amino acids were separated by using a reverse-phase column (250 mm×4 mm, Nucleosil 5C18) and a mobile phase consisting of 100 mM monosodium phosphate and 0.5 mM EDTA with 25% methanol (v/v) in water adjusted to pH 4.95 with 85% phosphoric acid. The total elution time was 20 min. The column temperature was kept at 25 ◦ C. Brain dialysate samples were also assayed for levels of DA, NE, 5-HT, 3,4-dihydroxyphenylalanine (DOPA), 3,4dihydroxyphenylacetaldehyde (DOPAC), homovanillic acid (HVA), 5-hydroxyindole-3-acetate (5-HIAA), and 3-methoxytyramine (3-MT) by HPLC-EC. Samples were separated by using a BST (60 mm × 4 mm) Hypersil 3ODS reverse-phase analytical column and a BST (20 mm × 4 mm) Hypersil 3ODS guard column and a mobile phase consisting of 100 mM Na2 HPO4 , 0.7 mM EDTA, 0.7 mM octane sulfonic acid with 12% methanol (v/v) in water adjusted to pH 2.90 ± 0.05 with 85% phosphoric acid. The column temperature was 25 ◦ C. The total elution time was 15 min. The results are expressed as means ± S.E.M. of the basal levels as percentage. Statistical significance was determined by one-way ANOVA, using Tukey’s Studentized Range (HDS) test. A P value of less than 0.05 was regarded as significant. At the end of the experiments, the rats were perfused transcardially with 4% paraformaldehyde in 0.1 M phosphate buffer, pH 7.2. The brains were postfixed overnight, and 20 m thin coronal sections were cut on a Reichert Jung freezing microtome. The site of the implanted probe was investigated on Nissl-stained frozen sections. Some of these frozen sections were stained with c-fos immunohistochemistry in order to reveal the effect of the tissue damage caused by the probe. 2.3. c-fos immunohistochemistry In separate experiments, 15 male rats (200–220 g) were anesthetized with Nembutal, and injected i.p. with 7 mg/kg 4-AP in physiological saline. Controls (another 15 animals) received the solvent only. 120 A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 The animals were subjected to transcardial perfusion with 4% paraformaldehyde in 0.1 M phosphate buffer (pH 7.4) 30, 60, 90, 120, and 150 min after the treatment: three controls (saline-treated) and three convulsing (4-AP treated) animals were perfused at every time. The Nembutal anesthesia was maintained with repeated i.p. injections throughout the experiment. The brains were dissected and postfixed for 1 h at room temperature. Following postfixation, the brains were cryoprotected overnight in 30% sucrose in 0.1 M phosphate buffer. Frozen serial sections at a thickness of 24 m were cut on a cryostat in the coronal plane, and one of two sections was processed for immunohistochemistry according to the following protocol. Single primary antibody (polyclonal c-fos antibody raised in rabbit, 1:1000; Santa Cruz Biotechnology, CA) was followed by secondary antibody (donkey anti-rabbit IgG, 1:40; Jackson ImmunoResearch, PA), with detection by the peroxidase-anti-peroxidase (PAP; Jackson ImmunoResearch) method (PAP dilution 1:1000), and the peroxidase reaction was developed by using nickel-intensified 3 3 -diaminobenzidine tetrahydrochloride (Sigma, St. Louis, MO) as chromogen. 2.4. Analysis of the immunostained sections The brains of three animals in each experimental group (30, 60, 90, 120, and 150 min following 4-AP and saline injections; 30 animals) were investigated. Sections of identical coronal stereotaxic planes (Paxinos and Watson, 1998) were used. Counting in the striatum was performed blindly of the animal’s treatment, on three histological sections from every animal (i.e., nine tissue samples from each time group). Rectangular areas (the image capturing field of the camera) were selected from the whole striatum (Fig. 1), and the immunoreactive nuclei displaying grayish-black staining were counted by using a NIKON ECLIPSE 600 microscope equipped with a Polaroid DMC digital camera (1600 dpi × 1200 dpi in 8 bits) with 20× objective magnification, with the Image Pro Plus 4 morphometry program (Media Cybernetics, Silver Spring, MD). Following background subtraction, the threshold was adjusted so that paleand deep-stained nuclei could be recognized equally by the counting program. The rectangular image capturing fields were equal to 0.249 mm2 with the magnification used. The cell counts were analyzed by Fig. 1. Sampling method for the counting of the c-fosIR cell nuclei. The rectangles in the striatum represent the image-capturing field of the camera (AOI). This AOI size is used for counting with 20× objective magnification. As shown by the drawing, 13 AOI’s are selected from every section, which cover the whole caudate–putamen region (with the exception of the ventral striatum and pallidum, which were not investigated). A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 means of ANOVA (post hoc test: Bonferroni method). The statistical analysis was performed with the help of the SPSS 9.0 computer program. 3. Results 3.1. Electroencephalography The depth and EEG recordings proved that the ictal activity in the neocortex and striatum began 15–25 min following the 4-AP injection (average latency 18 min). The background activity fell markedly and high-frequency tonic–clonic activity developed (Fig. 2). The amplitude of the ictal discharges increased and reached a maximum of 1.5–2.0 mV. The striatal seizure began 2–4 s later than the neocortical one (Fig. 2). The electric seizure phenomena greatly diminished after 50 min (from the beginning) and were completely absent from 60 min up to the end of the experimental period (Fig. 2). However, in the striatum spiking activity was continuous throughout the experiment: the amplitude of the spikes was less than 1 mV, but their frequency was 3–4 Hz even at the end of the experiment (Fig. 2B). 121 3.2. Microdialysis: transmitter amino acids The GLU concentration of the dialysate was significantly elevated at every sampling time following 4-AP injection. Maximum levels were measured at 120 min. The GLU levels in the control animals were constant (Fig. 3). Measurements of ASP resulted in a different curve: a sharp early rise in ASP concentration at 30 min which returned to the control level, and a later small, but significant elevation at 90 min (Fig. 3). The concentration of GABA displayed a steady increase and reached a maximum at 60 min. This 60-min value was significantly higher than that of the control. After 60 min, it decreased, and remained at an elevated level, which did not differ significantly from the control (Fig. 3). 3.3. Nitric oxide-related amino acids Following the 4-AP injection, the concentration of CIT peaked at 90 min with a significant elevation, then quickly returned to the control level at 120 min (Fig. 3). The level of ARG exhibited a slow elevation, which became significant at 60 min, and remained elevated until the end of the experiment (Fig. 3). Fig. 2. Seizure activity evoked by 4-AP in the striatum and neocortex. (A) The ictal activity begins 17 min after the 4-AP injection. (B) Disappearance of the epileptiform activity by the end of the experiment. Note that frequent, low amplitude spikes are detected with the striatal electrode only. (C) Schematic view of the arrangement of the recording electrodes (ch1: needle electrode inserted into the striatum; ch2, 3, 4: surface electrodes above the neocortex; calibration applies to panels A and B). A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 300 GLU * * * ** * 200 base base % 250 % 122 150 100 50 0 30 60 90 120 350 300 250 200 150 100 50 0 * 150 0 30 time (min) control (5) GABA 4-AP (6) control (5) 300 ** 200 90 120 150 4-AP (4) CIT 250 200 base base % 250 60 time (min) % 300 ASP * 150 150 100 100 50 50 0 30 60 90 120 0 150 30 control (4) control (5) 4-AP (6) base % 250 60 90 120 150 time (min) time (min) 4-AP (4) ARG 200 ** * 150 * 100 50 0 30 60 90 120 150 time (min) control (5) 4-AP (6) Fig. 3. Changes in concentrations of glutamate (GLU), aspartate (ASP), 4-aminobutyric acid (GABA), l-citrulline (CIT), and l-arginine (ARG) in control and 4-AP-treated rat striatum, expressed as percentages of the basal level. The number of dialysis samples (4–6) is given in brackets, and the injection of 4-AP or physiological saline is indicated by arrows. Asterisks denote significant differences: ∗ P < 0.05; ∗∗ P < 0.01. 3.4. Catecholamines, serotonin, and metabolites In the rat striatum, 4-AP treatment resulted in an increased release of all of the catecholamines and their metabolites, except HVA. The concentrations of DA, DOPA, and 3-MT remained increased throughout the whole test period, but only the DOPA concentration increase remained significant until the end of the experiment. DA release was initially highly significant, by 150 min it had decreased but remained elevated compared to the control (Fig. 4). The amounts of striatal DOPAC and 5-HIAA revealed steady increases and at the end of the experiment significant increases were seen (Fig. 4). The extracellular level of NE rose significantly immediately after 4-AP administration, but had fallen to the control level at 60 min and did not change thereafter (Fig. 4). The increase in the 5-HT level was similar to that in DA, showing a long-lasting significant elevation, but its decrease to the control level occurred earlier (Fig. 4). 3.5. c-fos immunoreactivity in the striatum The implantation of the probe elicited tissue damage, and scattered c-fos immunoreactive (c-fosIR) cells were visible around the tissue damage (Fig. 5). These 350 300 250 200 150 100 50 DOPA * * * DA *** 30 60 200 * 100 30 60 90 120 0 150 control (5) control (4) 4-AP (7) DOPAC * * 120 150 base % 300 250 200 150 100 50 0 30 60 90 140 130 120 110 100 90 80 control (4) 0 150 4-AP (6) 30 60 90 120 150 time (min) control (4) 4-AP (5) 500 3-MT * 400 base % * 120 HVA time (min) 300 90 time (min) time (min) base % * 0 0 base % 123 300 * base % base % A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 200 100 4-AP (5) NE 300 200 100 0 0 0 30 60 90 120 0 150 4-AP (6) control (4) 300 5-HT ** 200 100 0 0 30 60 90 120 90 120 150 150 200 180 160 140 120 100 80 4-AP (5) * 5-HIAA * 0 30 60 90 120 150 time (min) time (min) control (4) 60 control (5) base % base % ** 30 time (min) time (min) 4-AP (5) control (4) 4-AP (6) Fig. 4. Changes in concentrations of 3,4-dihydroxyphenylalanine (DOPA), dopamine (DA), 3,4-dihydroxyphenylacetaldehyde (DOPAC), homovanillic acid (HVA), 3-methoxytyramine (3-MT), norepinephrine (NE), 5-hydroxytryptamine (5-HT), and 5-hydroxyindole-3-acetate (5-HIAA) in control and 4-AP-treated rat striatum, expressed as percentages of the basal level. The number of dialysis samples (4–7) is given in brackets, and the injection of 4-AP or physiological saline is indicated by arrows. Asterisks denote significant differences: ∗ P < 0.05; ∗∗ P < 0.01; ∗∗∗ P < 0.001. 124 A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 Fig. 5. Immunohistochemical detection of c-fos in the striatum. (A) An animal perfused after the 4-AP plus microdialysis experiment displays the track of the microdialysis probe (arrow) and scattered c-fos staining around the probe. Note the strong c-fos immunostaining of the neocortex (cx) and piriform cortex (Pi). LV: lateral ventricle. Bar: 1 mm. (B) An animal perfused 90 min after 4-AP injection without microdialysis. The striatum contains strong c-fos immunoreactivity, represented by immunostained cell nuclei. Bar: 200 m. c-fos IR cell number / mm 2 animals were not used for quantitative immunohistochemistry because the tissue damage itself elicited c-fos expression and disturbed the counting. Control animals (not injected with 4-AP) displayed very few c-fosIR cell nuclei scattered in the striatum. 500 450 400 350 300 The animals which received 4-AP treatment under Nembutal anesthesia exhibited a characteristic c-fos expression pattern. The number of c-fosIR cells gradually increased from 30 min, and reached a maximum at 60 min (Fig. 6). A decrease was observed thereafter, * * * 4-AP * 250 200 150 100 50 0 control * * * * * 30 60 90 time (min) 120 150 Fig. 6. The number of c-fos immunoreactive (c-fosIR) cell nuclei in the striatum of anesthetized rats following 4-AP injection without microdialysis. Controls were injected with physiological saline. Every value differs significantly (P < 0.01) from the others. The S.E.M. values are shown at the top of the columns. The number of c-fos immunoreactive cells is largest at 60 min, then displays a gradual decrease, but the 150-min value is still significantly higher than the control. A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 but at the end of the experiment (at 150 min) the level was still significantly higher than the control (Fig. 6). We did not attempt to characterize the cells expressing c-fos protein. The size of the nuclei suggested that all of them were neuronal cell nuclei. The seizure-induced elevations in the number of c-fosIR cells were significant as compared not only to the controls, but also to each other at every time interval (Fig. 6). In the present experiments, no efforts were made for the co-localization of c-fosIR with neuronal antigens, or for the investigation of the relation of c-fosIR to the matrix and striosome compartments. 4. Discussion The effects of experimental seizures on transmitter release have been investigated by a number of authors (Nitsch et al., 1983; Jacobsson et al., 1997; Segovia et al., 1997; Smolders et al., 1997; Morales-Villagrán et al., 1999; Peña and Tapia, 2000). It is clear that acute experimental convulsions increase the extracellular concentrations of various transmitter substances because of the prolonged neuronal depolarization and the elevated extracellular K+ concentration (Macı́as et al., 2001). Nuclear magnetic resonance studies have revealed that seizures in the human neocortex increase the concentrations of transmitter amino acids (Sherwin, 1999). Our experimental data confirm these results and supply further evidence for the complex metabolic action of the seizure. The results also emphasize the importance and compatibility of animal experiments with regard to the human disease. 4.1. Electrophysiology The present data prove that i.p. injection of 4-AP precipitates electrographic seizure in the striatum and neocortex. The beginning of the striatal seizure can be detected 2–4 s after the onset of the neocortical ictal phenomena, indicating the spread of seizure activity from the neocortex towards the striatum. The excitatory glutamatergic afferents of the striatum originate from the neocortex and thalamus (Flores-Hernández et al., 1994) explaining that the neocortical and striatal EEG seizure events showed synchrony and both disappeared 50–60 min after the 4-AP injection. However, neuronal spiking activity persisted in the striatum with 125 higher frequencies than in the neocortex, indicating that the continuous in vivo release of several transmitters and metabolites caused a long-lasting excitation of striatal projection neurons (Flores-Hernández et al., 1994). 4.2. Glutamate and aspartate GLU and ASP are natural ligands and agonists of the ionotropic and metabotropic glutamate receptors (Alexander and Peters, 2000). GLU plays an important role in seizure initiation and spread exerting action not only on ionotropic, but also on metabotropic receptors (Chapman, 1998). The rise of GLU (and ASP) concentration in chronic seizures is well established (for a review, see Chapman, 1998). However, GLU is not always elevated in acute seizures (Nitsch et al., 1983; Segovia et al., 1997). Our present experiments demonstrate fast and highly significant elevation, indicating the transmitter role of GLU in the striatum. Since many of the excitatory afferents of the striatum originate from the cerebral cortex (and thalamus), we may suppose that the elevation in the striatal GLU is a consequence of neocortical hyperactivity and the excessive release of the transmitter from corticostriatal (and thalamostriatal) axon terminals. Interestingly, the GLU level reaches its maximum at 120 min, well after the cessation of the electrographic seizure. This explains the long-lasting spiking activity on the striatal EEG and raises the possibility that besides the postsynaptic depolarization, GLU might regulate the release of other transmitters through metabotropic GLU receptors (Pisani et al., 2002). Recent experimental evidence proves that protein kinase C (the activation of which is coupled to metabotropic GLU receptors) is an important mediator of the phosphorylation of transcription factors which are responsible for c-fos gene expression (Choe and Wang, 2002); so, metabotropic receptors themselves (subtypes 1 and 5; Cartmell and Schoepp, 2000) can participate in the induction of c-fos expression. A significant elevation was detected in the ASP level too, although the dynamics of ASP release displayed a different pattern: an early rise and fall, and then a much smaller (but significant) elevation. This observation is in register with literature data on the increase of ASP release in the striatum following electrical stimulation of the neocortex (Perschak and 126 A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 Cuénod, 1990). ASP, if released from nerve terminals acts mainly on NMDA-type receptors, so it may contribute to neuronal c-fos expression in seizure (Gundersen et al., 2001). On the other hand, ASP serves as important intermediate in energy metabolism of the brain—it is produced mainly from oxaloacetate by the enzyme aspartate aminotransferase, and its production is enhanced in hypoglycemia; the number of ASP immunoreactive neurons increases significantly in the hypoglycemic striatum (Gundersen et al., 2001). 4.3. GABA Pharmacological data suggest that in some acute epilepsy models the GABA levels decrease (Rowley et al., 1995), whereas in other experiments increased GABA levels were observed (Smolders et al., 1997). Recent studies indicate that GABA has an important role in epileptogenesis: GABA-mediated neuronal synchronization initiates seizures in some forebrain areas, mainly through the activation of GABAA receptors (Avoli, 1996). In the present experiments, the GABA levels rose slowly and peaked at 60 min, which indicated that GABA was released from striatal interneurons (Smith and Bolam, 1990) upon the effect of afferent excitatory axon terminals (Heimer et al., 1995). Although there are no literature data concerning the ratio of the GLU- and GABA-containing axon terminals, one explanation of our present observation may be that the number of GLU terminals is far greater than that of the GABAergic terminals; the detectable amount of GABA is, therefore, smaller and an increase to detectable levels takes more time. Interestingly, the GABA peak coincides with the cessation of the electrographic seizure (60 min), indicating the importance of GABAergic neurons in the control of and (probably) protection against striatal seizures. 4.4. Nitric oxide-related amino acids NO regulates many physiological processes and also contributes to the neurotoxicity of GLU in pathological conditions (Bredt and Snyder, 1992). NO is synthesized in the brain by isoforms of NO synthase (NOS) which converts ARG to CIT (Bredt and Snyder, 1992). In the striatum, some of the large, aspiny interneurons contain neuronal NOS (Vincent and Johansson, 1983). We observed a significant, steady, slow increase in ARG, and a sharp CIT peak at 90 min. This probably indicates the slow activation of NOS and the elevation of intrastriatal NO levels. Seizure conditions elevate brain NO levels (Kashihara et al., 1998), and the inhibition of NOS decreases seizure-related GLU release (Rigaud-Monnet et al., 1995). In the present experiments, GLU release increased first (at 30 min), the level of ARG increased 30 min later (at 60 min), then a CIT peak occurred only subsequently (at 90 min). This timing suggests that GLU release activated NOS, which led to the enhanced conversion of ARG to CIT. This result correlates well with the literature data on the NMDA sensitivity of NOS-containing neurons (Kendrick et al., 1996). 4.5. Catecholamines and metabolites It is known from the literature that 4-AP increased the spontaneous release of DA in rat brain slices, which could be a consequence of the depolarizing action of 4-AP on dopaminergic axon terminals (Versteeg et al., 1995). Accordingly, the precursor DOPA and the metabolites DOPAC, 3-MT, and HVA also underwent a slow accumulation, although the HVA levels did not increase significantly; proving the activity-related upregulation of DA synthesis and degradation by monoamino-oxidase and catechol-o-methyltransferase. The DA terminals originate in the substantia nigra, which is activated strongly in seizures (Bonhaus et al., 1986). This activation probably also contributes to the prolonged increase of striatal DA level. DA (probably through D4 receptors) prevents hyperexcitability (Rubinstein et al., 2001) and has a clear-cut neuroprotective role (Bozzi et al., 2000). On the other hand, we have to consider that whilst DA decreases striatal convulsion, it may also suppress the induction of c-fos in striatal neurons, probably through D2 receptors (Adams and Keefe, 2001; Marshall et al., 2001). Thus, the prolonged DA release probably contributed not only to the cessation of the electrographic seizure, but also to the fact, that the number of c-fosIR neurons was medium in the striatum (as compared to the c-fosIR neuron numbers in the cerebral cortex; Szakács et al., 2003). 4-AP increases NE release both in vitro (Versteeg et al., 1995) and in vivo (Morales-Villagrán et al., A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 1999). The time course of NE release in neocortical seizures (Morales-Villagrán et al., 1999) is very similar to that we measured in the present experiments. Striatal NE innervation originates from the locus ceruleus (Heimer et al., 1995), and NE is known to exert anticonvulsant activity, probably through beta receptors (Ferraro et al., 1994). However, this anticonvulsant effect did not last longer than 40–60 min in our experiments, because the increased NE in the dialysates had returned to the base level at 60 min. Nevertheless, we think that DA and NE together are able to exert anticonvulsant and neuroprotective roles, and probably contribute to the cessation of the electrographic seizure and the decrease in c-fos immunoreactivity during the experiments. 4.6. Serotonin and metabolites Serotoninergic afferents to the striatum originate in the dorsal raphe (Heimer et al., 1995). The axons display a high degree of collateralization (Heimer et al., 1995). Similarly to that of DA, 5-HT release is enhanced by 4-AP (Versteeg et al., 1995). Our present results prove that the characteristic features of 5-HT release are similar to those of DA and GLU, indicating that 4-AP acts on axon terminals in the striatum. 5-HIAA, a metabolite of 5-HT, was present in increased amount in the dialysates, indicating the extracellular degradation of the transmitter. 5-HT plays an important role in the striatum, regulating the release of GLU (Di Cara et al., 2001) and DA (Sershen et al., 2000). Seizure experiments demonstrate the modulatory role of 5-HT through different receptors: its inhibitory effect is mediated through 1A receptors to open Ca2+ -independent potassium channels (Colino and Haliwell, 1987). The same receptor can reduce the presynaptic Ca2+ entry into glutamatergic terminals, and thereby inhibit the release of GLU (Schmitz et al., 1997). 127 activity (Labiner et al., 1993; Mihály et al., 1997, 2001). Our experiments indicated that the release of GLU showed the best correlation with the increase of c-fosIR cells in the striatum. Pharmacological experiments in our laboratory proved the important role of ionotropic GLU receptors in the expression of the neuronal c-fos gene (Szakács et al., 2003). Therefore, we think that the gradual increase of the number of c-fosIR cells probably reflected the postsynaptic action of GLU, which was released on a similar time scale. At the end of the experiment, the level of GLU was still high, that of DA was decreasing but still higher compared to the controls. The number of c-fosIR cells displayed maximum at 1 h and a gradual decrease thereafter. We think that this was indicating the primary importance of extracellular GLU in the initiation and maintenance of c-fos expression. On the other hand, GABA, DA, NE, and 5-HT could play role in the termination of the c-fos gene expression by means of inhibition of neuronal activity and the closure of the voltage-dependent Ca2+ channels. Because of the relatively rapid decrease of Fos immunoreactivity in the striatum, we have to consider Fos degradation, too. Fos protein is unstable and is presumably degraded by the proteasome (Acquaviva et al., 2002). The mechanisms are not clear, but it seems, that extracellular signals regulate not only the transcription of the c-fos gene, but also the translocation, viability, and regulatory activity of the Fos protein in the cell nucleus (Acquaviva et al., 2002). The striatum presents an excellent model for the further study the role and significance of these extracellular signals, because the neuronal populations can be identified immunohistochemically, and with double immunostaining methods (Mihály et al., 2001) the localization of Fos protein in separate neuronal types can be studied in detail. 4.7. Seizure-related dynamics of c-fos expression Acknowledgements Our findings concerning the appearance of c-fos in the convulsing striatum are in accord with literature data on seizure-induced expression of ITF’s (Gass et al., 1992; Willoughby et al., 1995). Detection and evaluation of Fos immunoreactivity appears suitable for the histological mapping of epileptic neuronal The work was supported by the Hungarian National Research Fund (OTKA T 32566, T 037224, and T/11029131). The technical support from Mrs. Mónika Herczegh-Kara, Mrs. Márta Dukai, Mrs. Katalin Lakatos, and Mrs. Andrea Imre-Kobolák is greatly appreciated. 128 A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 References Acquaviva, C., Bossis, G., Ferrara, P., Brockly, F., Jariel-Encontre, I., Piechaczyk, M., 2002. Multiple degradation pathways for Fos family proteins. Ann. N.Y. Acad. Sci. 973, 426–434. Adams, A.C., Keefe, K.A., 2001. Examination of the involvement of protein kinase A in D2 dopamine receptor antagonistinduced immediate early gene expression. J. Neurochem. 77, 326–335. Alexander, S.P.H., Peters, J.A., 2000. Receptor and ion channel nomenclature supplement. Trends Pharmacol. Sci. 11, 1–120. Avoli, M., 1996. GABA-mediated synchronous potentials and seizure generation. Epilepsia 37, 1035–1042. Betancourt, L., Colom, L.V., 2000. Potassium channel expression in basal forebrain cholinergic neurons. J. Neurosci. Res. 61, 646–651. Bonhaus, D.W., Walters, J.R., McNamara, J.O., 1986. Activation of substantia nigra neurons: role in the propagation of seizures in kindled rats. J. Neurosci. 6, 3024–3030. Bonnano, G., Sala, R., Cancedda, L., Cavazzani, P., Cossu, M., Raiteri, M., 2000. Release of dopamine from human neocortex nerve terminals evoked by different stimuli involving extraand intracellular calcium. Br. J. Pharmacol. 129, 1780–1786. Bozzi, Y., Vallone, D., Borrelli, E., 2000. Neuroprotective role of dopamine against hippocampal cell death. J. Neurosci. 20, 8643–8649. Bredt, D.S., Snyder, S.H., 1992. Nitric oxide, a novel neuronal messenger. Neuron 8, 3–11. Cartmell, J., Schoepp, D.D., 2000. Regulation of neurotransmitter release by metabotropic glutamate receptors. J. Neurochem. 75, 889–907. Chapman, A.G., 1998. Glutamate receptors in epilepsy. Prog. Brain Res. 116, 371–383. Choe, E.S., Wang, J.Q., 2002. Regulation of transcription factor phosphorylation by metabotropic glutamate receptor-associated signaling pathways in rat striatal neurons. Neuroscience 114, 557–565. Colino, A., Haliwell, J.V., 1987. Differential modulation of three separate K-conductances in hippocampal CA1 neurons by serotonin. Nature 328, 73–77. Di Cara, B., Dusticier, N., Forni, C., Lievens, J.C., Daszuta, A., 2001. Serotonin depletion produces long-lasting increase in striatal glutamatergic transmission. J. Neurochem. 78, 240–248. Engel Jr., J., 1996. Excitation and inhibition in epilepsy. Can. J. Neurol. Sci. 23, 167–174. Ferraro, G., Sardo, P., Sabatino, M., Caravaglios, G., La Grutta, V., 1994. Anticonvulsant activity of the noradrenergic locus coeruleus system: role of beta mediation. Neurosci. Lett. 169, 93–96. Flores-Hernández, J., Galarraga, E., Pineda, J.C., Bargas, J., 1994. Patterns of excitatory and inhibitory synaptic transmission in the rat neostriatum as revealed by 4-AP. J. Neurophysiol. 72, 2246–2256. Gass, P., Herdegen, T., Bravos, R., Kiessling, M., 1992. Induction of immediate early gene encoded proteins in the rat hippocampus after bicuculline-induced seizures: differential expression of KROX-24, fos and jun proteins. Neuroscience 48, 315–324. Gundersen, V., Fonnum, F., Ottersen, O.P., Storm-Mathisen, J., 2001. Redistribution of neuroactive amino acids in hippocampus and striatum during hypoglycemia: a quantitative immunogold study. J. Cereb. Blood Flow Metab. 21, 41–51. Heimer, L., Zahm, D.S., Alheid, G.F., 1995. Basal ganglia. In: Paxinos, G. (Ed.), The Rat Nervous System. Academic Press, New York, pp. 579–628. Hoffmann, D.A., Magee, J.C., Colbert, C.M., Johnston, D., 1997. K+ channel regulation of signal propagation in dendrites of hippocampal pyramidal neurons. Nature 387, 369–375. Hosokawa, S., Kato, M., Kuroiwa, Y., 1983. Topographical distribution of propagation of seizure activity in the basal ganglia during focal motor seizures in the monkey. Neurosci. Lett. 38, 29–33. Jacobsson, S.O., Sellstrom, A., Persson, S.A., Cassel, G.E., 1997. Correlation between cortical EEG and striatal microdialysis in soman-intoxicated rats. Neurosci. Lett. 231, 155–158. Kashihara, K., Sakai, K., Marui, K., Shohmori, T., 1998. Kainic acid may enhance hippocampal NO generation of awake rats in a seizure-stage related fashion. Neurosci. Res. 32, 189–194. Kendrick, K.M., Guevara-Guzman, R., de la Riva, C., Christensen, J., Ostergaard, K., Emson, P.C., 1996. NMDA and kainate-evoked release of nitric oxide and classical transmitters in the rat striatum: in vivo evidence that nitric oxide may play a neuroprotective role. Eur. J. Neurosci. 8, 2619–2634. Korf, J., Venema, K., 1985. Amino acids in rat striatal dialysates: methodological aspects and changes after electroconvulsive shock. J. Neurochem. 45, 1341–1348. Kusske, J.A., 1979. Corticocaudatothalamic interactions in experimental focal epilepsy in the cat. Exp. Neurol. 65, 616– 624. Labiner, D.M., Butler, L.S., Cao, Z., Hosford, D.A., Shin, C., McNamara, J.O., 1993. Induction of c-fos mRNA by kindled seizures: complex relationship with neuronal burst firing. J. Neurosci. 13, 744–751. Macı́as, W., Carlson, R., Rajadhyaksha, A., Barczak, A., Konradi, C., 2001. Potassium chloride depolarization mediates CREB phosphorylation in striatal neurons in an NMDA receptor-dependent manner. Brain Res. 890, 222–232. Marshall, J.F., Henry, B.L., Billings, L.M., Hoover, B.R., 2001. The role of the globus pallidus D2 subfamily of dopamine receptor sin pallidal immediate early gene expression. Neuroscience 105, 365–378. McNaught, K.St.P., Brown, G.C., 1998. Nitric oxide causes glutamate release from brain synaptosomes. J. Neurochem. 70, 1541–1546. Meldrum, B.S., 1975. Epilepsy and gamma-aminobutyric acid-mediated inhibition. Int. Rev. Neurobiol. 17, 1–36. Mihály, A., Bencsik, K., Solymosi, T., 1990. Naltrexone potentiates 4-aminopyridine seizures in the rat. J. Neural Transm. (GenSect) 79, 59–67. Mihály, A., Szente, M., Dubravcsik, Zs., Boda, B., Király, E., Nagy, A., Domonkos, Á., 1997. Parvalbumin- and calbindincontaining neurons express c-fos protein in primary and secondary (mirror) epileptic foci of the rat neocortex. Brain Res. 761, 135–145. Mihály, A., Shihab-Eldeen, A., Owunwanne, A., Gopinath, S., Ayesha, A., Mathew, M., 2000. Acute 4-aminopyridine seizures A. Kovács et al. / Epilepsy Research 55 (2003) 117–129 increase the regional cerebral blood flow in the thalamus and neocortex, but not int he entire allocortex of the mouse brain. Acta Physiol. Hung. 87, 43–52. Mihály, A., Szakács, R., Bohata, C., Dobó, Es., Krisztin-Péva, B., 2001. Time-dependent distribution and neuronal localization of c-fos protein int he rat hippocampus following 4-aminopyridine seizures. Epilepsy Res. 44, 97–108. Morales-Villagrán, A., López-Pérez, S., Medina-Ceja, L., Tapia, R., 1999. Cortical catecholamine changes and seizures induced by 4-aminopyridine in awake rats, studied with a dual microdialysis-electrical recording technique. Neurosci. Lett. 275, 133–136. Nitsch, C., Schmude, B., Haug, P., 1983. Alterations in the content of amino acid neurotransmitters before the onset and during the course of methoxypyridoxine-induced seizures in individual rabbit brain regions. J. Neurochem. 40, 1571–1580. Patterson, T.A., Kim, E.K., Meldrum, M.J., Dawson Jr., R., 1995. Glutamate efflux from rat brain slices and cultures: a comparison of the depolarizing agents potassium, 4aminopyridine and veratridine. Neurochem. Res. 20, 225–232. Paxinos, G., Watson, C., 1998. The Rat Brain in Stereotaxic Coordinates. Academic Press, New York. Peña, F., Tapia, R., 1999. Relationships among seizures, extracellular amino acid changes, and neurodegeneration induced by 4-aminopyridine in rat hippocampus: a microdialysis and electroencephalographic study. J. Neurochem. 72, 2006– 2014. Peña, F., Tapia, R., 2000. Seizures and neurodegeneration induced by 4-aminopyridine in rat hippocampus in vivo: role of glutamate- and GABA-mediated neurotransmission and of ion channels. Neuroscience 101, 547–561. Perkinton, M.S., Sihra, T.S., 1999. A high-affinity presynaptic kainate-type glutamate receptor facilitates glutamate exocytosis from cerebral cortex nerve terminals (synaptosomes). Neuroscience 90, 1281–1292. Perschak, H., Cuénod, M., 1990. In vivo release of endogenous glutamate and aspartate in the rat striatum during stimulation of the cortex. Neuroscience 35, 283–287. Pisani, A., Bonsi, P., Catania, M.V., Giuffrida, R., Morari, M., Marti, M., Centonze, D., Bernardi, G., Kingston, A.E., Calabresi, P., 2002. Metabotropic glutamate 2 receptors modulate synaptic inputs and calcium signals in striatal cholinergic interneurons. J. Neurosci. 22, 6176–6185. Rigaud-Monnet, A.-S., Héron, A., Seylaz, J., Pinard, E., 1995. Effect of inhibiting NO synthesis on hippocampal extracellular glutamate concentration in seizures induced by kainic acid. Brain Res. 673, 297–303. Rowley, H.L., Martin, K.F., Marsden, C.A., 1995. Decreased GABA release following tonic–clonic seizures is associated with an increase in extracellular glutamate in rat hippocampus in vivo. Neuroscience 68, 415–422. Rubinstein, M., Cepeda, C., Hurst, R.S., Flores-Hernandez, J., Ariano, M.A., Falzone, T.L., Kozell, L.B., Meshul, C.K., Bunzow, J.R., Low, M.J., Levine, M.S., Grandy, D.K., 129 2001. Dopamine D4 receptor-deficient mice display cortical hyperexcitability. J. Neurosci. 21, 3756–3763. Schechter, L.E., 1997. The potassium channel blockers 4-aminopyridine and tetraethylammonium increase the spontaneous basal release of [3 H]5-hydroxytryptamine in rat hippocampal slices. J. Pharmacol. Exp. Ther. 282, 262–270. Schmitz, D., Empson, R.M., Glovelli, T., Heinemann, U., 1997. Serotonin blocks different patterns of low Mg2+ -induced epileptiform activity in rat entorhinal cortex, but not hippocampus. Neuroscience 76, 449–458. Segovia, G., Porras, A., Mora, F., 1997. Effects of 4-aminopyridine on extracellular concentrations of glutamate in striatum of freely moving rat. Neurochem. Res. 22, 1491–1497. Sershen, H., Hashim, A., Lajtha, A., 2000. Serotonin-mediated striatal dopamine release involves the dopamine uptake site and the serotonin receptor. Brain Res. Bull. 53, 353–357. Sherwin, A.L., 1999. Neuroactive amino acids in focally epileptic human brain: a review. Neurochem. Res. 24, 1387–1395. Sloviter, R.S., Dempster, D.W., 1985. “Epileptic” brain damage is replicated qualitatively in the rat hippocampus by central injection of glutamate or aspartate but not by GABA or acetylcholine. Brain Res. Bull. 15, 39–60. Smith, A.D., Bolam, J.P., 1990. The neural network of the basal ganglia as revealed by the study of synaptic connections of identified neurons. Trends Neurosci. 13, 259–271. Smolders, I., Van Belle, K., Ebinger, G., Michotte, Y., 1997. Hippocampal and cerebellar extracellular amino acids during pilocarpine-induced seizures in freely moving rats. Eur. J. Pharmacol. 319, 21–29. Szakács, R., Weiczner, R., Mihály, A., Krisztin-Péva, B., Zádor, Z., Zádor, E., 2003. Non-competitive NMDA receptor antagonist drugs moderate seizure-induced c-fos expression in the rat cerebral cortex. Brain Res. Bull. 59, 485–493. Thesleff, S., 1980. Aminopyridines and synaptic transmission. Neuroscience 5, 1413–1419. Tibbs, G.R., Dolly, J.O., Nicholls, D.G., 1989. Dendrotoxin, 4-aminopyridine, and -bungarotoxin act at common loci but by two distinct mechanisms to induce Ca2+ -dependent release of glutamate from guinea pig cerebrocortical synaptosomes. J. Neurochem. 52, 201–206. Versteeg, D.H.G., Heemskerk, F.M.J., Spierenburg, H.A., de Graan, P.N.A., Schrama, L.H., 1995. 4-Aminopyridine differentially affects the spontaneous release of radiolabelled transmitters from rat brain slices in vitro. Brain Res. 686, 233–238. Vincent, S.R., Johansson, O., 1983. Striatal neurons containing both somatostatin and avian pancreatic polypeptide (APP)-like immunoreactivities and NADPH diaphorase activity: a light and electron microscopic study. J. Comp. Neurol. 217, 252–263. Whetsell Jr., W.O., 1996. Current concepts of excitotoxicity. J. Neuropathol. Exp. Neurol. 55, 1–13. Willoughby, J.O., Mackenzie, L., Medvedev, A., Hiscock, J., 1995. Distribution of Fos-positive neuron sin cortical and subcortical structures after picrotoxin-induced convulsion varies with seizure type. Brain Res. 683, 73–87.