* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 24 Optogenetics - how to use light to manipulate neuronal networks
Central pattern generator wikipedia , lookup
Behaviorism wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
End-plate potential wikipedia , lookup
Multielectrode array wikipedia , lookup
Neuroethology wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Neuroeconomics wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neurotransmitter wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Mirror neuron wikipedia , lookup
Development of the nervous system wikipedia , lookup
Circumventricular organs wikipedia , lookup
Neural coding wikipedia , lookup
Chemical synapse wikipedia , lookup
Single-unit recording wikipedia , lookup
Neuroanatomy wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Nonsynaptic plasticity wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Biological neuron model wikipedia , lookup
Synaptic gating wikipedia , lookup
Nervous system network models wikipedia , lookup
G protein-gated ion channel wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
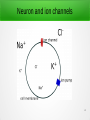
Optogenetics how to use light to manipulate neuronal networks Alexandra Götz 1 http://www.stanford.edu/group/dlab/optogenetics/ 2 Outline ● Background – Neuron and ion channels – Action potential – Light sensitive channels – Channel induction – ReaChr ● Experiment ● Results – ReaChR in Drosophila – CNS neurons – Wing extension reflex – Changes due to social factors ● Summary ● Future challenges 3 Neuron and ion channels 4 Action potential (9) 5 Light sensitive channels (3) 6 Light sensitive channels (3) 7 How are the light sensitive channels induced in the brain? 8 (8) 9 ReaChR Red-activable Channelrhodopsin Improved signaling characteristics e.g. greater photocurrent and faster kinetics λ~590-630nm (can penetrate deeper into tissue) ● Experiments without tissue damage possible ● Drosophila as testsubject? 10 Experiment ● ● ● Study freely moving test subjects Led light with different: – wavelength – cw vs pulsed ReaChR channels in different neuron types (1) 11 ReaChR in Drosophila ● Gr5a receptor neurons ● studying PER (proboscis extension reflex) ● comparison of different channel types ReaChR channel: ● Only light sensitive channel leading to robust response to green (530nm) and red (627nm) light dTrpA1 channel (thermal activated): ● No response at high surrounding T 12 ReaChR in Drosophila continuous illumination: ● PER decays exponentially ➔ depolarization block in Gr5a neurons (cf. thermal channels) pulsed illumination (1Hz): ● repeated spiking when light is activated (2) 13 CNS neuron activation: Testing different neuron types in CNS (central nervous system) with red and blue light-sensitive channels Various behavioral responses expected in channels e.g: wing extension reflex (male courtship song) ● ● response for ReaChR channel (green and red) no response for blue channel (CNS too deep in tissue) LED tuned behavior; experiments tunable with: – light intensity – pulse frequency 14 Wing extension reflex 2 types of neurons known to trigger wing extension reflex, P1 and pIP10, but their role is not nown yet: (1) (2) 15 Wing extension reflex 2 types of neurons known to trigger wing extension reflex, P1 and pIP10, but their role is not nown yet: (1) (2) 16 Wing extension reflex neuron type control type behavior response P1 neuron probabilistic during illumination period 17,5 ± 27,5sec pIP10 deterministic triggered with LED start 0,08 ± 0,04sec But both neuron types with ReaChR led to intensity independant behavior response. 17 Changes due to social factors: Probabilistic behavior of neurons leads to assumption that external factors could lead to changes in response. Test: single-housing males for 7 days Results: ● lower LED intensity leads to wing extension ● higher responses to stimulation Induction of ReaChR in both neurons separately indicates: P1 neurons are responsible for encanced sensitivity 18 Summary ● ReaChR is fitting opsin for Drosophila studies ● high-throughput screening of behavior possible ● reduced visual artifacts due to red illumination ● intensity and frequency dependent behavior ● ● probabilistic (P1) vs. deterministic (pIP10) response social isolation can affect optogenetic behavior 19 Future challenges: ● ● ● ● finding other social impact factors to P1 or other neuron types ReaChR and other opsin engineering for faster kinetis and smaller activation light ranges create red-shifted inhibitory opsins study of interacting neurons activated with different wavelengths 20 Thanks for listening. Questions? 21 References: (1)Inagaki et al., Nat Methods. 2014 March; 'Optogenetic control of freely behaving adult Drosophila using a red-shifted channelrhodopsin' (2) http://www.ncbi.nlm.nih.gov/pmc/articles/PMC4151318/ ; Supplementary info; (12.12.2014) (3)Fenno et al., Annual Rev. Neuroscience 2011; 'The Developement and Application of Optogenetics' (4)Lin et al.; Nat Neurosci. 2013 October; 'ReaChR: A red-shifted variant of channelrhodopsin enables deep transcriptional optogentic excitation' (5)Yizhar et al.; Neuron 2011 July; 'Optogenetics in Neural Systems' (6)http://en.wikipedia.org/wiki/Membrane_potential (7)http://www.stanford.edu/group/dlab/optogenetics/ (8)http://optogenetics.weebly.com/why--how.html (9)http://hyperphysics.phy-astr.gsu.edu/hbase/biology/actpot.html (10)https://www.youtube.com/watch?v=I64X7vHSHOE 22