* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download working memory
Memory consolidation wikipedia , lookup
Haemodynamic response wikipedia , lookup
Nonsynaptic plasticity wikipedia , lookup
Brain Rules wikipedia , lookup
Optogenetics wikipedia , lookup
Neuroanatomy wikipedia , lookup
State-dependent memory wikipedia , lookup
Adult neurogenesis wikipedia , lookup
Synaptic gating wikipedia , lookup
Neuroesthetics wikipedia , lookup
Executive functions wikipedia , lookup
Neurobiological effects of physical exercise wikipedia , lookup
Cognitive flexibility wikipedia , lookup
Cognitive neuroscience of music wikipedia , lookup
Neuropsychology wikipedia , lookup
Alzheimer's disease wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Metastability in the brain wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Biochemistry of Alzheimer's disease wikipedia , lookup
Cognitive psychology wikipedia , lookup
Neuroeconomics wikipedia , lookup
Limbic system wikipedia , lookup
Neuroplasticity wikipedia , lookup
Reconstructive memory wikipedia , lookup
Mental chronometry wikipedia , lookup
Holonomic brain theory wikipedia , lookup
Embodied cognitive science wikipedia , lookup
Cognitive science wikipedia , lookup
Neurophilosophy wikipedia , lookup
Environmental enrichment wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Neuroanatomy of memory wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
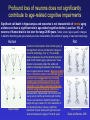
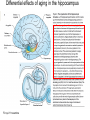
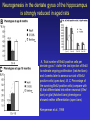
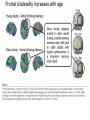
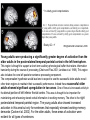
All highly developed nations in the world are experiencing substantial increases in the proportion of elderly adults in the population. The world is experiencing a substantial increase in the proportion of elderly adults in the population. Variations in demographic pyramids in Italy, from 1950 to 2100: Population by age groups and sex (percentage of total population) source ONU, Dipartimento Economico e Affari Sociali Population aged over 65 years Population aged over 80 years Statistics on population aging, analyzed for countries in OECD area for the period 2010/2050 The aging of the population represents both an opportunity and a threat for society. The opportunity comes from the tremendous reserve of human capital and experience represented by older citizens; the threat emerges from the frailties associated with aging and from the disconcerting fact that at this time, adults aged 85 and older have a very high dementia rate (typically in the form of Alzheimer’s disease), with a very high cost to affected individuals and families. AD prevalence (UK) 40-64 years: 1 in 1400 65-69 years: 1 in 100 70-79 years: 1 in 25 80+ years: 1 in 6 At present, it is fair to say that neurocognitive frailty is the biggest threat to successful aging in our society. (Denise C. Park and Patricia Reuter-Lorenz, Annual Review in Psychology, 2009) Adriana Fiorentini, 89 Carlo Maria Martini, 87 Rita Levi Montalcini, 103 Norberto Bobbio, 95 Cecilia Seghizzi, 108 Plasticity and the aging brain As individuals age, many aspects of information processing become less efficient,including speed of processing, working memory capacity, inhibitory function, and long-term memory. At the same time, other aspects of cognitive function such as knowledge storage, are protected and relatively resistant to cognitive aging. There is general agreement in the literature on cognitive aging that memory performance declines from early to late adulthood, and that such age-related losses in performance are much greater in relation to some tasks than to others. Age-related declines are slight in short-term memory span tasks, in which subjects repeat back a short string of words, letters or numbers. Decrements are typically considered slight also in implicit memory tasks. However…… An example of single trial visual perceptual learning An example of single trial visual perceptual learning % of subjects showing the “Eureka” effect "Eureka " effect and age 80,00% 70,00% 60,00% 50,00% 40,00% 30,00% 20,00% 10,00% 0,00% 18-50 65-69 70-74 75-79 80-84 85-89 90-95 Age (years) Berardi et al., unpublished results Age-related losses are substantial in information processing speed and in tasks of declarative memory, particularly spatial memory, and those tasks requiring recollection of events and of the original context in which an event occurred (pattern separation), all tasks involving the hippocampus and other medial temporal lobe structures. Also performance in inhibitory functions and in working memory tasks, relying on prefrontal cortex, declines with age; in particular, performance declines in those tasks which have high memory load and high demand on attention, which also deteriorates with age (Park and Reuter Lorenz, 2009). Similarly, animal models of aging show a decline in medial temporal lobe dependent and prefrontal cortex dependent memory (see Erickson and Barnes, 2003; Burke and Barnes, 2006; Barnes 2011 for review). Fin qui 26 novembre Performance in “dual “ tasks (walking while talking, divided attention,…). esempio Park and Reuter Lorenz, 2009 Digit symbol test (una cifra è associata ad un simbolo ed il soggetto deve studiare l’appaiamento e poi farlo il più rapidamente possibile. Computational span: si dà una piccola operazione da fare, tipo 7+3= e il soggetto deve dare la risposta e ricordare la seconda cifra. Alla fine di tutto, deve dare le seconde cifre nell’ordine in cui le ha incontrate Because these data are cross-sectional, it is possible that the observed differences are due to cohort effects or some other confound. However, data from the Victoria Longitudinal Study (Canada) , (Small, et al. Hultsch et al 1999) show strikingly similar findings for speed of processing, working memory, list recall and vocabulary, suggesting that the results are also found in longitudinal data and are not primarily due to cohort effects. Psychological approach Changes in cognitive processes with age are related to changes in the underlying mechanisms. 1) One dominant construct in the cognitive aging literature has been that of speed of processing. Salthouse (1991, 1996) argued that perceptual speed (measured for instance by the rate at which individuals can make “same/difference” judgments about simple shapes, dot matrices, or strings of letters or digits) is a fundamental, cognitive primitive that accounts for nearly all of the age-related variance on a broad range of cognitive tasks. 2) working memory, which encompasses both the short-term maintenance and active manipulative processing of information, figures prominently in a view of cognitive aging that emphasize declines in executive control processes, namely inhibitory function (Hasher&Zacks 1988, Hasher et al. 2007). According to this view, age-related deficits in cognition stem from the inefficiency of inhibitory processes that normally control the contents of consciousness (i.e., working memory). Older adults show working memory deficiencies and slowing due to selection of irrelevant information into the contents of working memory, along with inefficient deletion of working memory contents that are no longer relevant to task performance (filtering or inhibitory action). Inhibitory dysfunction with age is a source of general attentional dysregulation and accounts for age-related deficits in other cognitive domains such as task switching, response competition, and response suppression. 3) The effortful-automatic distinction (Braver & Barch 2002). Cognitive control operations guide thought and action in accord with task goals, especially when bottom-up, automatic, or prepotent stimulusresponse associations must be overridden. Age-related declines in cognitive control make older adults more susceptible to the influence of automatic, bottom-up processes. Age-related cognitive declines might thus be understood in terms of an age related decline in a range of mechanisms including speed, working memory, inhibition, and cognitive control (Moscovitch&Winocur 1992,West 1996) that show varying degrees of vulnerability in different individuals. Although these mechanisms can all be categorized as executive processes, other sources of decline, such as “dedifferentiation” ( but see later) of cognitive function, have also been considered. Given the broad spectrum of cognitive changes with age, it is unlikely that any single process or unitary mechanism can fully explain age-related deficits across all individuals. How to explain the decline in long term declarative memory, including spatial memory, with particular attention to sensitivity to context and to pattern separation? N.B.: A consensus holds that familiarity, a relatively automatic feeling of knowing that can support recognition-memory judgments, is relatively preserved with aging. By contrast, recollection, which requires the effortful, strategic recovery of contextual detail, declines as we age (Friedman, 2013). Declarative memory depends from the integrity of medial temporal lobe, (hippocampus and surrounding cortical areas), and of plasticity processes in these areas. Is a change in plasticity in these areas related to age dependent decline of declarative memory? There are several papers which have addressed and discussed the functional alterations that occur during normal aging in the medial temporal lobe and the prefrontal cortex and how these ageassociated changes might contribute to the selective cognitive impairments that occur in advanced age (see for instance Berchtold et al. 2008; Burke and Barnes, 2006, Park and Reuter Lorenz 2009; Barnes 2011, Holden and Gilbert, 2012 for review). Many of these studies point out that age-related changes in cognition cannot be accounted for by a generalized loss of neurons: indeed, in humans (Raz et al., 2004; Pakkenberg and Gundersen, 1997; Pakkenberg et al., 2003; West, et al 1994), non-human primates (Merrill et al., 2004; Peters et al., 1994; Gazzaley et al., 1997; Keukeret al., 2003) and rodents (Merrill et al., 1996), significant cell death in the hippocampus and neocortex is not characteristic of normal aging. There are instead age-related reductions in synaptic density and neuropil, resulting in reduction in volume of specific brain structures, but not a general decrease in the number of neurons. Burke and Barnes, 2006 Berchtoldt et al., 2008 On average, there is a correlation between GM amd WM integrity ad cogntitive state. However, many studies failed to find such a correlation Profound loss of neurons does not significantly contribute to age-related cognitive impairments Significant cell death in hippocampus and neocortex is not characteristic of normal aging and does not have a significant role in age-related cognitive decline. Less than 10% of neurons of human brain is lost over the range 20-90 years. Rather, small, region-specific changes in dendritic branching and spine density are more characteristic of the effects of ageing on neuronal morphology Human Rat A common misconception about normal ageing is that significant cell loss and dramatic changes in neuronal morphology occur. a | This example shows progressive loss of the dendritic surface in aged human dentate gyrus granule cells. These data do not accurately reflect the subtle and selective morphological alterations that actually occur in aged neurons, however. Age-associated loss of dendritic extent in the dentate gyrus and CA1 was exaggerated by including healthy aged individuals and those with dementia in the same experimental group, and not using stereological controls. c | Hippocampus Representative hippocampal CA1 neurons from young rats (2 months) and old rats (24 months). There is no reduction in dendritic branching or length with age in area CA1. In the subiculum of non-human primates, however, significant reductions in spine density with age have been observed between the ages of 7 and 28 years. Hippocampus Burke and Barnes, 2006 One exception: Memory Impairment in Aged Primates Is Associated with Focal Death of Cortical Neurons and Atrophy of Subcortical Neurons Also cholinergic input to area 8A is reduced in aged monkeys Smith et al., 2004 At the level of neuronal morphology, investigations on dendritic branching and spine density suggest that age associated alterations are also region-specific: morphology of prefrontal cortex neurons seems to be more vulnerable to the effects of aging than that of hippocampal neurons (see Burke and Barnes, 2006). Thus, normal aging is accompanied by differential and often subtle changes in gray and white matter, with different regions and even different subregions being differentially affected by normal and pathological aging; this is evident, for instance, at the level of the hippocampus (see Small et al., 2011 for a review). Very different is of course the situation in presence of age associated neurodegenerative diseases leading to dementia, such as Alzheimer’s disease (AD). In summary, advancing age is accompanied by a pattern of differential brain changes that are observed in both cross-sectional and longitudinal investigations (see Raz and Rodriguez, 2006 for a review). Overall, there is age related cortical thinning and shrinkage of some neural structures and there is loss of subcortical neurons in the diffuse projecting systems (dopaminergic, cholinergic). There are disproportionate declines in the prefrontal cortex, moderate declines in temporal cortex and only mild declines in occipital regions. As far as neural plasticity is concerned, normal aging causes evident changes at multiple levels: in addition to the reduction in synaptic density, which by itself could affect synaptic plasticity by making it more difficult to attain the sufficient amount of cooperatively active synapses necessary to induce synaptic efficacy changes, and to changes in Ca2+ conductances and homeostasis, aging alters forms of synaptic plasticity related to declarative memory processes such as hippocampal LTP. Hippocampal LTP is altered in aged animals at the level of both induction and maintenance, with maintenance most severely affected (see Burke and Barnes 2006 for review). Most basic neuronal properties remain rather constant with age both in the hippocampus and in the PFC Aged animals do have alterations in the mechanisms of plasticity that contribute to cognitive functions. LTP decline and spatial memory decline (Barnes maze) correlate Burke and Barnes, 2006 Even in normal aging there are subtle changes in the biological processes that underlie memory, and in the relationship between how good an animal’s memory is and how functional their synapses are. But the real message in these experiments is that old animals can and do learn. None of the older animals exhibited what could be considered behaviorally to be “dementia”, nor were their biological mechanisms that allow memory traces to be laid down completely dysfunctional. Age-related changes in gene expression The maintenance of LTP requires gene expression and de novo protein synthesis; therefore, it is not surprising that aged animals and humans also show alterations in these processes. Following behavioural induction, the proportion of cells expressing a gene in a specific brain structure can be measured with fluorescence in situ hybridization. Panel b shows confocal images of fluorescence in situ hybridization for Arc mRNA in the dentate gyrus of a young rat and an old rat. Granule cells are shown in red and Arc mRNA in yellow. After spatial exploration, more granule cells are positively labelled for Arc in young than old rats. No such changes in CA3, for instance In humans, prominent change in baseline gene expression occurred in the sixth to seventh decades across cortical regions, suggesting that this period is a critical transition point in brain aging, particularly in males. Burke and Barnes, 2006; Berchtoldt et al., 2008 Epigenetic factors in aging Young and old animals show differences in DNA methylation. In the resting old brain, granule cells, for example, had overall higher methylation levels, meaning that it was less likely that plasticity genes could be transcribed from the DNA (Barnes and Sweatt, see Barnes 2001). There may well be a number of changes that accumulate during aging via epigenetic means, and that dysregulation of these processes may contribute in a fundamental way to normal age-related cognitive change. Peleg et al. (2010) found that during contextual fear conditioning learning, aged mice (16 months old) exhibited a specific deregulation of histone H4 lysine 12 (H4K12) acetylation and failed to initiate a hippocampal gene expression program associated with memory consolidation. This was in very good correlation with behavioural data showing a deficit in long-term fear memory. Interestingly, restoration of physiological H4K12 acetylation reinstated the expression of learninginduced genes and led to recovery of cognitive performance. The maintenance of place maps also differs between young and old animals. In normal young rats, a place map for a given environment can remain stable for months. Therefore, when a rat is returned to the same environment, the same place map is retrieved. A similar stability of CA1 place maps in aged rats is observed within and between episodes of behaviour in the same environment. Occasionally, however, if the old rat is removed from the environment and returned later, the original place map is not retrieved and an independent population of place cells may be activated even in a familiar room. Spatial maps of an environment DO NOT remain locked to context in old rats Exploration 24h later, re-exploration Young Old In the second case, the old animal DOES NOT remember having been in this place, DOES NOT possess a spatial map of it and may think to be in another place. Reactivation of hippocampal neurons during sleep is impaired in aged rats Gerrard et al., 2008 Differential effects of aging in the hippocampus Fin qui 11 novembre Aging effects on hippocampal neurogenesis Aging strongly reduces hippocampal neurogenesis (dentate gyrus) across several species (see Amrein et al., 2010; Kempermann et al., 1998, 2002 and 2010). (aside note: Recent evidence by Frisen group in Sweden (Spalding et al, 2013) found definitive evidence for DG neurogenesis in humans. By modelling the process of carbon 14 presence in brain cells, the team estimated that we generate around 1400 new neurons every day in the DG, roughly the same percentage as in rodents; most of them die, but some are integrated into DG circuits). The brain, time-stamped. Atmospheric 14C that was released during nuclear bomb tests between 1945 and 1963 has been incorporated into the DNA of dividing cells, providing a time-stamp. This has been used to prove adult hippocampal neurogenesis in humans, thereby confirming a particular type of structural and functional brain plasticity involved in higher cognitive function. (News and views, Kemperman et al., 2013) Neurogenesis in the dentate gyrus of the hippocampus is strongly reduced in aged rats A, Total number of BrdU-positive cells per dentate gyrus 1 d after the last injection of BrdU to estimate ongoing proliferation (hatched bars) and 4 weeks later to assess survival of BrdUpositive cells (open bars). B, C, Percentage of the surviving BrdU-positive cells (compare with B) that differentiated into either neuronal (filled bars) or glial (hatched bars) phenotype or showed neither differentiation (open bars). Kemperman et al., 1998 Aging effects on hippocampal neurogenesis In humans, indirect (doublecortin expression in hippocampal cells from post mortem samples) evidences for reduced neurogenesis in aged subjects comes from the study of Knoth et al. (2010) and by volumetric reductions in hippocampal formation found in human aged subjects (Apostolova et al., 2011; Small et al., 2011). Frisen study (2013) suggests that the ability to create new neurons declines less in aged humans than in aged mice: instead of the nearly 10-fold decrease between young and middle-aged mice, the Swedish team found only a four-fold decrease in humans. Riduzione della neurogenesi nel giro dentato con l’età: uomo e roditore Kheirbeck and Hen, 2013 Ruolo della neurogenesi ippocampale nella memoria: Vedi lezioni sui meccanismi di plasticità neuronale Small et al., 2011 The human dentate gyrus plays a part in ‘pattern separation, a cognitive operation that allows similar stimuli flowing through the hippocampal circuit to be represented with distinct neural codes allowing correct storage of the spatial and temporal contex of a memory (effects of block of neurogenesis on pattern separation, GageBussey e vanPraag). Impairments in these tasks have been documented in ageing subjects (see also van Praag) and have been interpreted as confirming previous fMRI findings that implicated the dentate gyrus in normal ageing. Although the computational process underlying pattern separation occurs within the dentate gyrus itself, impaired performance might also be expected with upstream lesions in the entorhinal cortex. A recent view of hippocampal dysfunction in aging (Barnes, 2011). Until recently, most neuroimaging and neuropsychological tests have evaluated the hippocampal formation as a singular structure, but it is in fact a complex circuit made up of functionally and molecularly distinct subregions. Moreover, the complexity of the hippocampal formation extends beyond its internal circuit organization. Hippocampal vulnerability to insults of different nature is regionally specific. Recent gene-expression studies have established that each hippocampal subregion has a distinct molecular profile, and this ‘molecular anatomy’ provides a partial substrate for regional vulnerability. The relatively high expression of certain NMDA receptors in CA1 helps to explain its vulnerability to excitotoxicity in the context of hypoxia and ischaemia associated with vascular disease whereas the high levels of corticoid receptors in the dentate gyrus confer vulnerability to the effects of reductions in the level of circulating corticosteroids. Different regions of the hippocampus have different involvements in cognition. Although ageing is itself not a disease, cognitive decline that occurs during ageing has deleterious consequences, and because it occurs without prominent cell loss it is considered a prime example of a functional disorder. Nevertheless, the effect of ageing on hippocampal function is often confounded by diseases, in particular Alzheimer’s disease and vascular disease, which commonly occur in older subjects and can cause hippocampal dysfunction independent of ageing. When attempting to isolate the hippocampal pattern of dysfunction reflective of ageing per se, it is therefore important to exclude the effect of Alzheimer’s disease and vascular disease fMRI have been applied to ageing human subjects (with or without Alzheimer’s disease and with or without vascular disease), ageing rhesus monkeys, ageing mice and mice that express disease-causing mutations in the amyloid precursor protein (FAD). Besides confirming the differential link of Alzheimer’s disease to the entorhinal cortex and vascular disease to CA1, results from these cross-species studies suggested that ageing itself differentially affects the dentate gyrus AD VD AGING Regional vulnerability and metabolic state differentiate disorders that affect the hippocampal formation. Although multiple hippocampal subregions can be affected in disorders, by comparing patterns of alterations that are observed by functional and structural MRI it is possible to isolate individual subregions differentially affected by each disorder. Furthermore, functional imaging techniques that are sensitive to metabolic state have suggested that some hippocampus-based disorders are characterized by hypometabolism (shown in blue), whereas others are abnormally hypermetabolic. a | Alzheimer’s disease, vascular disease and ageing all contribute to hippocampal alterations in late life. A direct comparison suggests that the entorhinal cortex (EC) is differentially associated with Alzheimer’s disease and the CA1 with vascular disease, whereas the ageing process per se seems to differentially target the dentate gyrus (DG). Hypometabolism has been localized to the EC in Alzheimer’s disease, to CA1 in vascular disease and the DG in ageing. NB (for the next lectures): While aging and Alzheimer’s disease may in some cases be “superimposed”, there is not a simple linear trajectory where we all end up with the disease. Rather, aging has a different neural signature, supporting the contention that aging and Alzheimer’s disease are distinct. Staub et al., 2006: in participants with amnestic MCI, compared with age-matched controls, results showed a significant decrease in white matter volume in the region of the parahippocampal gyrus that includes the perforant path. There was also significant atrophy in both the entorhinal cortex and the hippocampus. “Compensatory” plasticity in aging In parallel to diminished neural plasticity, there are numerous examples of what can be considered compensatory plasticity during the aging process. This shows up mostly in terms of activation of a larger and more elaborate pattern of acrivation of brain areas, and in particular of the prefrontal cortex, in aged with respect to young subjects performing the same task; interestingly, this more extensive activation correlates with better performance in the elders (see Park and Reuter Lorenz, 2009 for a recent review). Frontal bilaterality increases with age Is this additional activity important for the better performance of old subjects? To answer this question, repetitive TMS (rTMS) has been used to transiently disrupt prefrontal cortex function. These studies have provided especially compelling evidence in favor of contralateral activity enhancing cognitive function in older adults. rTMS is a technique that transiently disrupts neural function by applying repetitive magnetic stimulation to a specific area of the brain, creating highly focal and temporary “lesions.” In a seminal study conducted by Rossi et al. (2004), young and older adults studied pictures while rTMS was applied to the subjects’ left or right dorsolateral prefrontal cortex (DLPFC) (encoding); the subjects then made recognition judgments while rTMS was again applied to the left or right DLPFC (retrieval). One of the most interesting finding from this work was that young adults’ memory retrieval accuracy was more significantly affected when the rTMS was applied to the right compared to the left hemisphere during retrieval. In contrast, older adults’ retrieval was equally affected by the rTMS during retrieval, whether it was applied to left or right, suggesting that the activation in both hemispheres was useful for performing the recognition task. Similar results to those described for memory have been found for linguistic abilities (Wingfield and Grossman, 2006) and motor abilities Heuninckx et al., 2008); interestingly, one of the additional brain areas recruited by elders with good performance in the linguistic task is an area activated in young subjects in situations of particularly complex sentence processing, which stress working memory demands. Y > elderly good comprehenders Elderly GC > Y Wingfield and Grossman, 2006 Young adults were producing a significantly greater degree of activation than the older adults in the posterolateral temporal-parietal cortex in the left hemisphere. This region is thought to support a short-term auditory-phonological buffer that retains information transiently during the course of processing (Chein and Fiez 2001; Jonides et al. 1998). This region also includes the core left posterior sentence processing component. The compensation hypothesis would lead one to expect to see the successful older adults recruit other brain regions to maintain their successful performance. Indeed these successful older adults showed significant upregulation in two areas. One of these is increased activity in the dorsal portion of left inferior frontal cortex. This area is thought to be important for maintaining and rehearsing stored verbal information in working memory. The other is the right posterolateral temporal-parietal region. The young adults also showed increased activation in this area but only for sentences that especially stressed working-memory demands (Cooke et al. 2002). For the older adults, these areas of activation were evident for all types of sentences. This additional activation, which young people use in challenging situations, would compensate for the lower activation in the core left hemisphere posterior language areas found in aged people. The result is as good a performance in elder as in young subjects. The aging brain can maintain a relatively high level of performance despite the biological changes associated with aging. The stability in performance is maintained by engaging novel brain regions and implementing novel (cognitive) strategies in response to a constant (cognitive) goal. Comment on “dedifferentiation” This highlights the adaptive and plastic nature of the neurobiology of brain function that supports optimizing goal attainment with available resources even when in older adulthood these resources may become diminished. Thus, the aging brain can maintain a relatively high level of performance through neural plasticity processes, optimizing goal attainment with the available resources. Park and Reuter-Lorenz point out that compensatory plasticity in the form of more elaborate patterns of brain area activation is not unique to aging but that it is the brain’s response to cognitive challenge: it is more likely to find a more elaborate pattern of brain area activation in aging brains even for simple tasks simply because tasks which for young brains result to have a low level of complexity bring forth significative cognitive challenge for older brains Neural plasticity in the form of compensatory changes in aging “the corpus of these data suggests that the brain is a dynamic organism seeking to maintain homeostatic cognitive function.” Park and Reuter-Lorenz, 2009 In contrast to the age-related declines in cognitive function and brain structure, functional brain activity increases with age, particularly in the frontal cortex. The proposed scaffolding theory of aging and cognition suggests that this increased functional activity is due to compensatory activation—the recruitment of additional circuitry with age that shores up declining structures whose function has become noisy, inefficient, or both. The Authors propose that the Prefrontal cortex, the most flexible structure in the brain, is largely responsible for the scaffolding processes in the aging brain. Scaffolding is the brain’s response to cognitive challenge and is not unique to aging. Aging simply results in more frequent cognitive challenges at lower levels of intensity. The aged brain is in effect less efficient at generating scaffolding, and significant pathology (as occurs in advanced Alzheimer’s disease) may entirely limit scaffolding operations. Enhancing neural plasticity in old age, both that underlying learning and memory and that underlying compensatory activation, might benefit cognitive performance in elders through multiple mechanisms Cognitive scaffolding is promoted by cognitive activity. “….. growing evidence suggests that humans develop scaffolds as a result of stimulating experiences. (Parl and Reuter-Lorenz, 2009” It might be possible to enhance plasticity in old age (that underlying learning and memory, that underlying compensatory activation) by being active. Predictors of maintaining cognitive function in older adults: The Health ABC (Aging and Body Composition) Study Yaffe et al., Neurology, 2009 Elders who maintain cognitive function have a unique profile that differentiates them from those with minor decline. Importantly, some of these factors are modifiable and thus may be implemented in prevention programs to promote successful cognitive aging. See you next lecture