* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Endocannabinoid system wikipedia , lookup
Multielectrode array wikipedia , lookup
Single-unit recording wikipedia , lookup
Functional magnetic resonance imaging wikipedia , lookup
Neuropsychology wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Holonomic brain theory wikipedia , lookup
History of neuroimaging wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
Brain Rules wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Aging brain wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Neuroplasticity wikipedia , lookup
Nervous system network models wikipedia , lookup
Electrophysiology wikipedia , lookup
Neuroeconomics wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Haemodynamic response wikipedia , lookup
Development of the nervous system wikipedia , lookup
Central pattern generator wikipedia , lookup
Synaptic gating wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Neural oscillation wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Neuroanatomy wikipedia , lookup
Circumventricular organs wikipedia , lookup
Hypothalamus wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Metastability in the brain wikipedia , lookup
Colin Pittendrigh wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Optogenetics wikipedia , lookup
Channelrhodopsin wikipedia , lookup
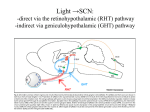
Cover Page The handle http://hdl.handle.net/1887/20930 holds various files of this Leiden University dissertation. Author: Houben, Mathias Bernard (Thijs) Title: Regulation of behavioral activity by the suprachiasmatic nuclei Issue Date: 2013-06-06 Chapter 1 General introduction 1 Preface General introduction Since the earliest days of life on earth, living creatures have been exposed to a dynamic, constantly changing environment. Only those organisms with the necessary attributes to adapt were able to reproduce successfully and spread their genes forward in the tree of life. Many of the challenges facing living organisms from those early days up to the present are due to chance and unpredictable. A number of environmental factors is not governed by chance however, but change in a cyclic and predictable pattern because they are driven by the daily rotation of the earth around its axis and its yearly trajectory around the sun. The day-night rhythms in the amount of light, temperature, humidity and many other parameters require constant adaptation of the organism in order to survive. A plant that is dependent on sunlight as the energy source for photosynthesis should store enough energy during the day to drive vital processes during the night. A nocturnal rodent that forages for food has to make sure that it is safely back in its burrow and away from open fields before the sun rises in the morning. The adaptive value of predicting and anticipating cyclic environmental changes led to the evolution of a built-in timekeeping mechanism, a biological clock, that keeps track of the time of day and is present in nearly every form of life. A second environmental rhythm that is equally essential to survival is the 365 day seasonal rhythm caused by the rotation of the earth around the sun. The yearly cycle in the duration of day and night and the amount of solar energy reaching the earths’ surface leads to strong seasonal differences in temperature, weather patterns and food availability. This requires organisms to time reproduction, energy expenditure and other physiological functions to the right time of year. And just as with 24 hour rhythms, anticipation of these seasonal changes greatly improves the chances for survival and reproduction. Plants should blossom in the right season to allow seeds to fully ripen before the winter. Animals should time conception of their offspring so they are born when there is enough food available to feed them. Although the yearly cycle of the seasons occurs on a very different timescale as the daily cycle of light and darkness, it is also measured by the biological clock that keeps track of time of day by measuring the seasonally changing duration of daylight. There are many examples showing that life has adapted to a rhythmic world and how nearly every form of life is constantly anticipating and changing to prepare for the next phase of the cycle and for the next season. Although humans, through the invention of alarm clocks and electric lighting, are no longer fully dependent on the solar light-dark cycle for timing their activity and rest, they too possess a built-in biological clock driving their daily physiological and behavioral rhythms. As we progress during the day, we eat, drink, and perform a wide 8 Circadian rhythms The first evidence for the presence of an internal timekeeping mechanism was presented in a letter published in 1729 by the French geophysicist Jean-Jacques d'Ortous de Mairan (4). In this letter, he described a simple experiment performed on the Mimosa plant, that has the interesting property that it folds its leaves during the night and opens them in the daytime. De Mairan aimed to test if the leaf movement was a response to the presence of light during the day, and placed the plant in a dark closet to see if it would keep its leaves in the night position. To his surprise, he found that the plant continued to open and close its leaves in the absence of any light. Although his experiment was far from perfect, it did provide the first known evidence that organisms have a built-in clock that continues ticking in constant conditions. An idea that has been confirmed in many plant and animal species since then, from the cellular level (gene expression, cell division) to physiological processes (hormone 9 1 General introduction array of physical and mental activities. As we rest and sleep during the night, we give our body and brain a precious time-window to recover, process the nutrients and experiences of the previous day and allow our physiological systems to prepare for the next active period. Throughout this repeating daily cycle, it is our biological clock system that coordinates that our physiological, mental and metabolic systems are at all times optimally prepared for the changing demands posed by the daily rhythm of our lives. In recent years, the importance of this endogenous clock system for health and the association of rhythm disturbances with disease have received growing attention. From clinical practice in many types of disease, it is evident that disturbances of the sleep-wake rhythm occur as illness develops or progresses over time. In the case of psychiatric disorders with an episodic nature such as schizophrenia, bipolar disorder or depression, disruption of the sleep-wake cycle often precedes or coincides with episodes of increasing severity of the symptoms (1-3). Although the causality in the association between disease and disturbed rhythms is uncertain in many cases, there is an increasing recognition that through nighttime light exposure, jet lag and shift work, the urban lifestyle of the modern 24-7 society exposes our endogenous biological clock system to disruptive inputs that can impair its’ function and may ultimately compromise our health. This thesis consists of two parts. The first part consists of five studies in which we used the mouse as a model organism for investigating the mechanisms that enable mammals to modify their behavior and physiology in order to deal with daily and seasonal rhythms. The second part consists of three experimental studies in which we explore the relation between disruption of the circadian system and diseases, including psychiatric disorders, metabolic disease and renal failure. General introduction Figure 1. Circadian rhythm in behavioral activity 2 Waking and sleeping in humans and animals is 3 scheduled according to the time of day. In laboratory animals, we can measure the timing 4 of behavioral activity by placing a movement 5 sensor or a running wheel in the cage of the 6 animal. Here, the activity of a laboratory mouse 7 (Mus musculus, C57Bl6 strain) in a running wheel was recorded during 18 days and plotted as an 8 actogram. In an actogram, each line shows 24 9 hours of behavioral activity, and subsequent 10 days are plotted below each other. For the first 9 11 days, The animal was exposed to an artificial 24 hour day, consisting of 12 hours of light (white 12 background) alternated by 12 hours of darkness 13 (grey background). As mice are nocturnal 14 animals, they sleep during most of the light 15 phase and run in their wheel in the darkness. 16 After day 9, the lights are kept off and the animal is left in continuous darkness. Although 17 the animal can no longer tell the time of day 18 from the light in its’ environment, the rhythm in 0 4 8 12 16 20 24 behavioral activity continues. Without any Time [hours] external time-source, activity starts slightly earlier each day, and the ‘free-running’ rhythm obtains a period that is slightly shorter 24 hours. Without a daily ‘resetting’ of the clock by the external light-dark cycle, it generates a period close to, but not exactly, 24 hours in constant conditions. Therefore, the rhythm is called a ‘circadian’ rhythm (from the latin ‘circa’ = about, and ‘dies’ = day). Unpublished data. 1 Time [days] 1 levels, body temperature, metabolism), to behavior (sleeping/waking, cognitive performance). A common aspect of these rhythms is that although they continue under constant conditions, their period deviates slightly from 24 hours if there are no environmental changes that indicate the time of day. When a mouse has a running wheel in its cage, it will very precisely start running at the same time each night, and will continue running for a large percentage of time until the end of the night (Figure 1). If the mouse with the wheel is then placed in constant darkness, the running wheel rhythm will remain very strong, but the animal will start and stop running a fixed number of minutes earlier each day. Without an external rhythm, the endogenous clock continues ticking, but with a period that is (in case of the mouse) slightly shorter than 24 hours. After the mouse in the wheel, such rhythms that start several minutes earlier or later each day (depending on the species) are said to be ‘free-running’. This observation of the endogenous biological clock running several minutes faster or slower than 24 hours if conditions are constant prompted researcher Frans Halberg to call these rhythms “circadian”, from latin “circa” (about) and “dies” (day). 10 Entrainment of circadian rhythms B Delay No Shift C D Advance 1 2 3 1 Phase shift (hours) Time (Days) A 5 7 9 * * ** 11 Advance 0 -1 -2 13 0 Active period Delay 4 8 12 16 Time (hours) 20 24 0 4 8 12 16 Time (hours) 20 24 0 4 8 12 16 Time (hours) 20 24 -3 6 8 10 12 14 16 18 20 22 0 2 4 6 Circadian Time (circadian hours) Figure 2. Entrainment of circadian rhythms: the Phase Response Curve Much has been learned about the synchronization of the biological clock to the environmental light-dark cycle by placing animals in constant darkness and measuring the effect of brief light pulses on the timing of their behavior. Here, three actograms are shown of a mouse in constant darkness. On day 7, each animal was exposed to 30 minutes of light (red asterisk) at different times relative to their behavioral activity. A. When the light pulse is timed during the resting period, the timing of behavioral activity on the days following the pulse is not shifted. B. When the light pulse occurs in the first hours after the animal woke up and became active, the activity in the days following the pulse starts later; a phase delay has occurred. C. When the light pulse occurs towards the end of the active phase, the activity on subsequent days starts earlier. To visualize this phase advance, a red line was fitted through the start of activity on the days preceding the light pulse, and a blue line was fitted through the start of activity on the days following the light pulse. D. By performing many such phase shifting experiments with light pulses at different times, a phase response curve (PRC) can be constructed that plots the average shift in behavior as a function of the time of the pulse (10). Here, the PRC of the mouse is drawn schematically. The time of the pulse is determined in Circadian Time (CT), in which CT12 is defined as the start of behavioral activity on the day of the pulse, and one circadian hour equals the total duration of the free-running period (about 23.5 hours in mice), divided by 24. As is evident from the actograms in B and C as well as the PRC in D, mice respond with large phase delays to light pulses at the start of the active period and with much smaller phase advances to light pulse at the end of the active period. Unpublished results. 11 1 General introduction The endogenous circadian clock needs to adjust to the external cycle to keep accurate track of time. How the endogenous circadian clock stays synchronized, or entrained, to the 24 hour environment became a main question in the mid twentieth century, culminating in the first international conference on biological rhythms in Cold Spring Harbour in 1960. Progress in the field was driven by the systemic exploration of the properties of animal circadian rhythms, led by a number of key researchers, including Jürgen Aschoff, Rutger Wever, Patricia deCoursey and Colin Pittendrigh. In a range of species, including birds, insects and rodents, they recorded and manipulated behavioral rhythms in the field and in the lab. In doing this, they established and described a number of key general properties of circadian rhythms (5, 6). One is that light has a strong effect on the timing, or phase, of circadian rhythms and is the most important signal responsible for keeping the internal biological clock synchronized to the 24 hour environment (7-9). By keeping the animals in constant conditions, and testing the effect of single pulses of light on the behavioral rhythms it was shown that the effect of light on circadian rhythms is dependent on the phase of the biological clock (10, 11). In the evening and beginning of the night, light delays (slows down) the biological clock, causing the animal to wake up and start activity later on the days after 1 General introduction the light pulse (Figure 2). In the end of the night and the beginning of the morning, a light pulse has the reverse effect and advances (speeds up) the clock, leading to earlier times of waking up and falling asleep in the days after a pulse. During the day, light pulses have little to no phase-shifting effect on the clock. The time-dependent effect of light on the clock can be summarized in a so-called phase-response curve and explains how exposure to environmental light in the morning and evening keeps the biological clock adjusted to the environment. The molecular mechanisms at the basis of circadian rhythmicity The 1953 discovery that DNA is the carrier of our genetic blueprint started the era of molecular genetics (12). Using constantly improving analysis methods, scientists throughout the life sciences set forth to make sense of the vast amount of information stored in the genome, and to understand the connections and mechanisms connecting this information to the properties of cells, tissues and the organism as a whole. In 1971 the DNA revolution caught up with chronobiology for the first time in a publication describing the discovery of a gene in the fruit fly (Drosophila melanogaster), that is involved in generating and regulating circadian rhythms (13). Fruit flies are a favorite organism for molecular biologists because they are fast breeders and have DNA that can easily be manipulated and investigated. Like many other insects, fruit flies start their life as larva that metamorphose into an adult fly in the pupal stage. They emerge from the pupa in an event called eclosion. The time at which the adults emerge from the pupa has a strong circadian rhythm that could be easily measured using devices and techniques developed in the lab of Colin Pittendrigh. In the experiments, random DNA damage was induced in many fruit flies before mating, followed by a screen for abnormalities in the circadian rhythm in eclosion time of the offspring. With this strategy, the researchers discovered that damage in different parts of a specific gene on the flies X chromosome led to strong changes in the circadian rhythm of the offspring. One group had a very fast clock of 19 hours, one group a very slow clock of 28 hours, and the third group had no circadian rhythm at all in constant conditions. These rhythm changes were present not only in the time of eclosion but also in the behavior of larva and adults. The newly discovered gene was called “Period” and was the first of many similar genes discovered in many species of animals, plants, fungi and bacteria. Together, these genes form the essential basis for the intracellular mechanisms responsible for generating the basic day-night rhythm that allow organisms to be prepared at all times for the challenges posed by living in a rhythmic world. In the 4 decades since the discovery of the first clock gene, an enormous amount of detail has been unraveled about the genes, molecules and their interactions that make up the molecular circadian clock. Despite numerous differences between species, a 12 The suprachiasmatic nuclei In stark contrast with the accumulating knowledge on the general properties of circadian rhythms and their entrainment, very little was known in the mid twentieth century about the underlying mechanisms of the biological clock and where it was located. It took until 1972 before the first evidence emerged that the mammalian biological clock is in fact located in a specific anatomical substrate. This evidence consisted of two papers in which brain lesions were made in a small region at the bottom of the rat hypothalamus, directly above the optic chiasm, the area where the optic nerves reach the brain. In the first study, a lesion of this suprachiasmatic area caused the disappearance of the behavioral rhythms in drinking and running in a running wheel (14). The second study investigated the daily rhythm in corticosterone production by the adrenal gland, and found that this endocrine rhythm was absent in animals with a suprachiasmatic lesion (15). While these lesion studies showed that an intact suprachiasmatic nucleus is necessary for the expression of circadian rhythms, it was not yet conclusive evidence that the SCN is also the location of the biological clock itself. It took several follow up studies to make this point as well. First of these was the 1979 paper by Inouye and Kawamura (16). In this study, they implanted 13 1 General introduction uniform principle has emerged that explains how different sets of clock genes and their products interact in a very similar way that makes them function as a pacemaker, generating a rhythm of approximately 24 hours. The unifying principle is the concept of a transcription-translation feedback loop that has been found at the heart of the intracellular clock in all investigated organisms. In mammals, a set of 5 genes, Period 1,2,3 and Cry1 and 2, all contain so called e-box elements in their promotor region. One CLOCK protein can bind together with one BMAL protein to form a dimer that binds to this e-box and activates transcription of the Per and Cry genes during the day. Over the course of the day, PERIOD and CRY proteins accumulate and also bind together to form dimers. These dimers then find their way to the nucleus, where they interact with the CLOCK/BMAL dimers and inhibit the transcription of the Period and Cry genes during the night. At the end of the night, degradation of PERIOD and CRY proteins diminish the number of inhibitory dimers, giving the CLOCK/BMAL dimers the opportunity to initiate a new cycle. The abundance of BMAL and CLOCK is also rhythmic and regulated in an additional regulatory loop that is thought to stabilize and enhance the rhythm of the Period/Cry feedback loop. In this secondary loop, Bmal1 and 2 transcription is inhibited during the day by REVERBα binding to ROR elements in the Bmal promotor. REV-ERBα levels are controlled by the CLOCK/BMAL complex are lower and the inhibitory effect on Bmal transcription is counteracted by RORα. 1 General introduction electrodes in the rat brain to record the electrical activity of SCN neurons in awake animals. They found that the SCN electrical activity level shows a clear circadian rhythm, with high activity during the day and low activity in the night. The rhythm continued when the animals were placed in constant darkness and persisted even when they surgically severed the connections between the SCN and the rest of the brain, showing that the rhythm was not dependent on synaptic input from other brain regions. In 1982, three different labs independently managed to extract a thin slice containing the suprachiasmatic nucleus from the rat brain, keep this brain slice alive, and use it in an in vitro preparation to do electrophysiological recordings (17-19). All studies showed that the SCN is capable of generating a circadian rhythm in electrical activity even when it is completely isolated from rest of the body. The case for the SCN as the location for the biological clock was made complete by an elegant transplantation study, published in 1990 (20). The study made use of the discovery of a spontaneous genetic mutation in hamsters, that caused hamsters with this ‘tau’ mutation to have a very fast clock, with a period of 22 hours in constant darkness (21). In the study, SCN lesions were made in normal hamsters with a 24 hour rhythm and afterwards, the SCN from ‘tau’ mutant hamsters was transplanted into the SCN lesioned normal hamsters. The result of this treatment was that circadian rhythms were restored in the lesioned hamsters, but they now expressed a 22 hour rhythm showing that the period of the rhythm is determined by the SCN. Together, these studies indicate that the SCN is the location of brain clock responsible for generating circadian rhythms. Anatomical location of the SCN The suprachiasmatic nuclei are located at the bottom of the brain (Figure 3), directly on top of the location where the optic nerves coming from the eye reach the brain and within the hypothalamus (23). Anatomically, the hypothalamus is part of the diencephalon, an ancient vertebrate brain structure connecting the more rostral midbrain and hindbrain to the cerebrum, the most frontal part of the brain (24). The nuclei of the hypothalamus form a central command structure responsible for regulating a number of key physiological and behavioral systems including reproduction, body temperature, metabolism and hunger, water balance & thirst, and activity and rest. The hypothalamus contains mechanisms for sensing the bodies’ temperature, water content and metabolic energy levels. In a very broad sense, the functions of the hypothalamus all have to do with maximizing the animals chances of survival and reproduction by balancing the animals behavior and physiology to each other and adjusting both to the environmental demands. Most functions regulated by hypothalamic nuclei display a strong circadian rhythm, an observation that closely 14 matches the location of the SCN at the base of the hypothalamus, as well as its strong connectivity to the nearby nuclei (25). General introduction A 1 B C V3 OC Suprachiasmatic Nuclei Figure 3. The suprachiasmatic nuclei. The SCN are located at the base of the brain, in an area called the hypothalamus. A. Schematic view of the mouse brain from the side. When the brain is cut at the red line, the SCN (red structures) become visible when seen from the front, as is depicted in figure B. C. Microscopic image of a mouse brain section, showing the hypothalamic area, demarcated by the rectangle in figure B. In the middle of the picture is the ventricle (V3). At the bottom, the white structure (OC) is the optic chiasm, which is the place where the optic nerves enter the brain. Immediately on top of the optic chiasm are the bilateral suprachiasmatic nuclei. In this image, the SCN were stained using an antibody against AVP, a neuropeptide expressed by SCN neurons. A,B redrawn after Paxinos & Franklin (22), C. unpublished result. Light input to the SCN The eyes are connected to the brain via the optic nerves that enter brain at the optic chiasm, located at the base of the hypothalamus. The eyes contain specialized neurons within the retina that are sensitive to light. These photoreceptive rods and cones release neurotransmitters onto nearby retinal ganglion cells and react to light by changing their membrane potential and decreasing the amount of neurotransmitter they release. The ganglion cells have long axons projecting to the brain within the optic nerves, and react to the decrease in transmitters by increasing the rate of action potentials they generate. In the last decade, it has become clear that apart for the rod and cone cells that are essential for vision, the eyes contain a second group of lightsensitive cells. This second group consists of a small percentage of retinal ganglion cells that contain melanopsin, a photopigment that is most sensitive to blue light 15 1 General introduction (26-29). Mice with a genetic defect causing the loss of rods and cones are completely blind, but retain non-visual responses to light such as the restriction of pupil size, suppression of nocturnal melatonin release and entrainment to the 24 hour light-dark cycle (30-32). Different from the rhodopsins and photopsins found in rods and cones, melanopsin allows these ganglion cells to respond in a different way to light that makes them ideally suited to act as a sensor that measures the intensity of the light coming into the eye. Where rods and cones respond almost instantly to changes in the amount and color of the light, their opsins are also quickly bleached by prolonged light exposure. This bleaching makes their responsiveness decrease over time, as evidenced by the amount of time needed to adjust your eyes to sudden darkness. In contrast, when melanopsin containing ganglion cells are exposed to light, they increase their activity much more gradually, reaching a sustained level of activity that reflects the amount of light to which the cells are exposed. The role of melanopsin containing ganglion cells as illumination sensors is reflected by the function of the brain regions they innervate. Where regular retinal ganglion cells transmit signals from rods and cones to the visual cortex via the lateral geniculate nucleus, axons from melanopsin containing ganglion cells innervate several nuclei in the hypothalamus and thalamus. This specific pathway is called the retino-hypothalamic tract (RHT) and connects the melanopsin containing ganglion cells in the retina with the SCN, the intergeniculate leaflet (IGL) and the olivary pretectal nucleus (OPN). These are the nuclei that are essential for regulating circadian rhythms (SCN and IGL) and the pupillary light reflex (OPN) (33, 34). Besides the direct innervation of the SCN by the RHT, light information has second, indirect way of reaching the SCN. This secondary light input originates from the IGL, which has efferent fibers projecting to the SCN that release neuropeptide Y and contribute to the response of the SCN to photic stimuli (33). Axons of the melanopsin containing ganglion cell of the RHT heavily innervate the ventral part of the SCN. When light hits the retina, these axons release a mixture of glutamate and Pituitary adenylate cyclase- activating peptide (PACAP) onto the ventral SCN cells. Glutamate is an excitatory transmitter that increases the firing rate of the cells in the ventral SCN (35, 36). Since SCN cells have a maximal firing rate, the excitatory effect of light on SCN activity is most pronounced during the night, when most cells are not generating spontaneous activity (37). Light exposure in the eye during the night leads to a firing rate increase in the SCN and a shift of the circadian rhythm if the exposure is strong enough and occurs in the early or late night. Both SCN activation and phase shifts can be suppressed by using blockers that interfere with the glutamate receptor found on SCN neurons (38, 39). The neuropeptide PACAP that is co-released with glutamate appears to augment and reinforce the effect of glutamate in the SCN. Although excitation of SCN cells and phase shifting can still 16 take place when PACAP is absent or blocked, both are decreased in the absence of PACAP (40-42). Apart from photic input from the eye and IGL, the SCN also receives inputs from other regions of the brain. These inputs allow the SCN to respond to other cues than light, including activity (43), feeding (44) and sleep deprivation (45). The signaling of such information about the the behavioral state of the animal to the SCN is called ‘non-photic’ input and involves several pathways. One of these originates in the brainstem raphe nucleus, which connects to many areas in the brain and releases serotonin, a central neurotransmitter involved in the regulation of behavior and mood. One of its’ targets is the SCN, where it releases serotonin whenever an animal is active (46). In the SCN, serotonin alters the sensitivity of SCN neurons to light by modulating glutamate release from the RHT and altering the postsynaptic sensitivity to glutamate (47, 48). Furthermore, serotonin release can induce phase shifts that mimic the phase shifts that are induced by non-photic stimuli applied during the resting phase of nocturnal rodents, when light has no effect on the clock (figure 2). Besides serotonin, behavior-associated neuropeptide Y (NPY) release in the SCN by fibers from the intergeniculate nucleus also induces non-photic-like phase shifts, that can be blocked by pre-treating the SCN with an NPI blocker (49). A third signaling pathway that can be considered ‘non-photic’, involves several hormones for which SCN cells express receptors. These hormones that can phase-shift SCN cells or modulate their light-sensitivity, include melatonin (50, 51), estrogen (52) and testosterone (53). Finally, the SCN also receives information about sleep and vigilance state. Where previous studies had shown that locomotor activity can alter SCN electrical activity levels (54, 55), combined EEG and SCN electrical activity recordings showed that SCN electrical activity is also modulated by sleep states (56). Specifically, REM sleep increases SCN electrical activity, while non-REM sleep suppresses it. The anatomical pathways that mediate these specific sleep effects on the SCN likely involve sleepregulatory areas such as the nucleus basalis the pedunculopontine tegmental nucleus and the laterodorsal tegmental nucleus but also the serotonergic projections from the raphe nuclei. Anatomical and functional organization of the SCN Anatomical studies of the SCN in different mammalian species have revealed that within the SCN, subregions exist in which neurons differ in their properties and function. Communication in the circadian system can be conceptually divided into 17 General introduction Non-photic input to the SCN 1 1 General introduction four different aspects: input to the SCN from the eye, input from other brain regions, communication between different cells and regions within the SCN, and the output communication of the SCN to other parts of the brain. Each of these communication pathways utilizes a distinct set of neurotransmitters and peptides. Nearly all neurons of the SCN release Gamma-Amino Butyric Acid (GABA) as their principle neurotransmitter, but regional differences exist within the SCN in the type of neuropeptides that are expressed and release by the SCN neurons (23). Although much remains unknown about the precise roles of the different peptides, it appears that the presence of certain peptides demarcates two main parts of the SCN that each have a specific functional role. The first of these is the so-called core or ventromedial part of the SCN that lies immediately on top of the optic chiasm. This core region contains neurons that express vasoactive intestinal peptide (VIP) and gastrin-releasing peptide (GRP). The neurons of the core region are densely innervated by axons terminals of retinal ganglion cells that release glutamate and PACAP when light activates the retina. Thus, a main function of the core region appears to process photic information from the eye and use this to entrain the circadian rhythm of the SCN to the environmental light-dark cycle. Apart from photic input from the retina, the ventral SCN is also innervated by serotonergic fibers from the raphe nuclei and NPY-releasing fibers from the IGL, enabling the SCN to integrate both photic and non-photic environmental timing cues (57). The VIP positive neurons of the core region project to dorsal SCN neurons and to other hypothalamic areas. Mutant mice that lack either VIP or its receptor show strong deficits in entrainment and have difficulty in maintaining a circadian rhythm in constant darkness (58, 59). Thus, VIP seems to play an essential signaling role in keeping SCN neurons synchronized to each other and the outside world. The dorsal part of the SCN contains neurons expressing arginine vasopressin (AVP), a substance mainly involved in communication timing signals to downstream targets of the SCN that regulate metabolism (60). For the communication of timing information between the ventral and dorsal SCN regions, GABA has been shown to be especially important in a study investigating the adaptation of the rat SCN to a 6 hour delay of the light-dark cycle, an adaptation comparable with a transcontinental flight from western Europe to New York (61). Immediately following such a shift, brain slices containing the SCN were prepared from the brains of the rat, in which the electrical activity rhythm of the SCN was recorded. Instead of the single daily electrical activity peak in the SCN from unshifted rats, the SCN rhythm now displayed a double peak. The first peak was timed in the middle of the day relative to the original light-dark cycle. The second peak corresponded to the middle of the day in the new, 6 hours delayed day. Separating the slices into a dorsal and ventral SCN containing part with a knife-cut showed that the shifted peak originated in the ventral SCN, while the dorsal SCN showed an unshifted 18 Electrophysiological properties of SCN neurons The rhythms generated by the intracellular molecular clock are translated into a rhythmic output signals at the membrane of SCN neurons. These neurons have been found to spontaneously generate action potentials at frequencies of 10 to 15 spikes per second (63, 64). Neurons of the SCN spontaneously generate action potentials for about 3 to 5 hours each day (65-67). Generating this rhythm is cell-autonomous property that does not rely on interactions between neurons. This has been shown in experiments in which SCN neurons were taken out of the brain and investigated in vitro. When these cells were cultured with little or no connectivity to other cells, they were still capable of generating a circadian rhythm in electrical activity (68). As all excitable cells in the body, the generation of an action potential in an SCN neuron requires that the membrane potential is increased up to the threshold potential at which enough voltage gated sodium channels open to initiate an action potential. Like other neurons, SCN neurons fire when their membrane potential is sufficiently depolarized by axonal input from other neurons as illustrated by experiments showing that SCN cells start firing when the retina is exposed to light (69, 70). Apart from these induced potential changes, neurons of the SCN have the uncommon property that during several hours each day, their membrane potential spontaneously depolarizes toward the threshold potential, causing the cell to generate action potentials. This time-window of spontaneous electrical activity falls during the day for most SCN cells, causing a strong circadian rhythm in the electrical activity level of the SCN that can be detected using implanted electrodes in the SCN (Figure 4). Although the intracellular molecular clock is ticking inside nearly every cell in the body, SCN neurons are unique in their ability to communicate this intracellular rhythm to other cells by coupling the molecular clock to the membrane potential. Although many mechanistic details remain unknown, it appears that in the SCN, the expression of several ion channels that modulate membrane potential is under circadian control (71). Importantly, the coupling between molecular clock and membrane potential appears 19 1 General introduction peak. When the same procedure was done 6 days after the shift of the light-dark cycle, the entire SCN had completely shifted and again displayed a single electrical activity peak in the middle of the shifted day. Thus, the cells of the ventral SCN that receive retinal input shift immediately, while dorsal SCN cells that receive less direct retinal input take a couple of days to fully adapt to such a shift. Blocking the GABA-a receptor had the same effect as making the knife-cut between the ventral and dorsal SCN. Furthermore, the data showed that at certain times of the day, GABA can have an excitatory effect on cells in the dorsal SCN. This finding was later confirmed in a study by Choi et al., (62) and is surprising because GABA is generally regarded as a purely inhibitory neurotransmitter in the adult brain. 1 to be bidirectional: studies have shown that blocking the electrical rhythm of SCN neurons appears to suppress the intracellular molecular clock (72). General introduction Network properties of the SCN The SCN contain approximately 10000 neurons in each nucleus (23). These neurons form a network by means of direct synaptic connectivity between them. The network of SCN neurons in turn is part of a bigger network, receiving input from the eye and from other brain regions, and sending output to other parts of the brain. Although SCN neurons are capable of generating circadian rhythms in firing frequency when communication between them is absent or impaired, the rhythms generated by individual cells are imprecise and variable in period (68, 73, 74). Communication within the SCN network appears to add a level of robustness to the SCN clock, allowing it to compensate for the effects of mutations in the Per1 or Cry1 clock genes. Although dissociated SCN neurons and peripheral cells from animals carrying these mutations became arrhythmic, SCN-containing brain slices that preserved the network connectivity remained strongly rhythmic (75). In normal animals, the rhythm produced by the intact SCN is much more precise and stable when these neurons are connected in a network where the rhythmic activity of each neuron is transmitted to connected SCN cells by neurotransmitters that are released from its axon terminals during each action potential. Apart from this neurochemical communication, some of Figure 4. Electrical activity rhythm in the suprachiasmatic nuclei in freely moving mice The neurons of the SCN spontaneously generate action potentials during part of the day, causing a circadian rhythm in spontaneous electrical activity that can be measured by surgically implanting a recording electrode into the SCN. Here, two examples are shown of a 102 hour (4.25 days) recording in the SCN of a freely moving mouse. Action potentials from neurons close to the electrode are detected in the signal, counted, and stored on a computer. The black dots indicate the number of action potentials per second. The behavioral activity of the animals was recorded simultaneously and is depicted below the electrical activity (vertical black lines). The background color indicates the light that is present in the recording cage, white indicates lights were on, grey indicates that lights were off. The animal in example A. was exposed to an artificial 24 hour day, while the animal in example B. was recorded in constant darkness. Note that electrical activity is high during the day and low during the night, while behavioral activity in the nocturnal mice shows a reverse pattern. The recording in B shows that the rhythm in SCN electrical activity continues under constant conditions, in line with the behavior (see also Figure 1.). Unpublished data. 20 SCN Output mechanisms and pathways In order to regulate the diverse physiological and behavioral rhythms, the circadian timing signal generated by the SCN needs to be communicated to other brain areas and organs. Axons from SCN neurons terminate in several brain regions. Besides direct axonal projections that communicate directly with postsynaptic neurons via neurotransmitters, the SCN also releases several neuropeptides that can communicate circadian time to target neurons even without direct synaptic connections. In an elegant experiment, Silver et al. (81) used tau mutant Hamsters with a 22 hour clock and made them arrhythmic by lesioning the SCN. Then they took SCN tissue from hamsters with a normal, 24 hour period, placed the tissue inside a capsule made of semipermeable polymer. After implanting the capsule in the SCN-lesioned animals, they observed that these animals became rhythmic again, with a 24 hour period. As the polymer capsule allows free diffusion of soluble compounds but prevents the 21 1 General introduction the SCN neurons are also electrically coupled by gap junctions that allow an action potential generated by one cell to directly spread onto the membrane of the connected cell. The function of the communication between the cells of the SCN is to synchronize the rhythm of the individual cells to each other and to the outside world. To achieve this synchronization, SCN cells are capable of adjusting the phase of their circadian rhythm in response to the signals they receive from the network. Output pathways from the SCN contain axons from multiple neurons. The circadian pattern in neurotransmitter release from these pathways is therefore determined by the sum of the circadian release pattern of al individual neurons involved in generating the output. SCN slice recordings show that individual neurons of the SCN generate action potentials for only 3 to 5 hours each day, and are silent for the remaining hours. In contrast, SCN population recordings show a much longer duration of high electrical activity that reflected the full duration of the previous lightdark cycle (65-67). Thus, the SCN network can track seasonal changes in day-length by distributing the timing of the short activity pattern of individual neurons over the day to ensure that the duration of high population activity accurately reflects day-length (76). In a study testing the role of VIP in this ‘photoperiodic encoding’, VIP-deficient mice were unable to adapt to different day-lengths, indicating that VIP plays an essential role in the synchronization process that allows the SCN to adapt to seasonal changes in day-length[Lucassen et al., in press]. In these mice, circadian rhythmicity is weakened by a genetic defect, but similar deficits in network synchronization may also occur as a consequence of aging, when VIP levels in the SCN have been shown to decrease (77, 78). The importance of synchronization of the SCN network is further supported by studies showing that exposing animals to constant light severely disrupts synchronization of SCN neurons and leads to disrupted behavioral rhythms (79, 80). 1 General introduction outgrowth of nerve fibers, this study convincingly indicated that the SCN releases a diffusible signal that is sufficient to drive a circadian rhythm in running wheel activity. Importantly, these diffusible signals only restore the circadian rhythm in behavioral activity, but not the daily neuro-endocrine rhythms in hormones such as corticosterone, and melatonin, for which synaptic output from the SCN is essential (82). Since the discovery that the SCN releases humoral factors than drive behavioral rhythms, several of such substances have been identified. The first of these was transforming growth factor alpha (TGF-α). TGF-α is rhythmically produced by the SCN with a peak during the sleep phase, and the epidermal growth factor (EGF) receptors that respond to TGF-α are present in the sub-paraventricular zone (sPVZ) (83). The sPVZ is an area that is densely innervated by SCN neurons and an essential relay station in the circadian regulation of behavioral activity (84). Infusion of TGF-α in the third ventricle inhibited behavioral activity and mice with a defective EGF receptor displayed excessive daytime locomotor activity and failed to suppress activity when exposed to light. A second SCN-released factor that controlled behavioral activity was prokineticin 2 (PK2) (85, 86). Like TGF-α, PK2 release peaks during the resting phase and suppresses behavioral activity when infused into the third ventricle. The receptors for PK2 are expressed in several SCN-innervated regions, but unlike the receptor for TGF-α not in the sPVZ. Another difference with TGF-α signaling, is that the PK2 receptor is also present in neurons in the dorsal SCN where PK2 has an excitatory effect (87, 88). Mutants missing PK2 or a functional PK2 receptor have severely disrupted circadian rhythms (89, 90). The third SCN-released humoral factor was cardiotrophin-like cytokine (CLC), which is released by a subpopulation of SCN vasopressin neurons and peaks during the sleep phase (91). CLC receptors are located along the the third ventricle and, like TGF-α and PK2, locomotor activity was inhibited when CLC was injected into the ventricle. Conversely, locomotor activity is increased when CLC receptors are blocked during the sleep phase. The last SCN-released humoral factor mention is AVP, which was in fact the first rhythmic, SCN-released substance to be discovered. In a 1981 paper (92), Reppert et al., described a strong rhythm in AVP in the cerebrospinal fluid in cats. In spite of this discovery, AVP was soon discarded as an important SCN output factor because of the subsequent discovery that the “Brattleboro” strain of rats was perfectly capable of generating circadian rhythms in behavior, even though they carried a genetic mutation that prevented the expression of AVP (93). It took until 1992 to reinstate AVP as a relevant SCN output, when it was shown that the rhythmic AVP release by the SCN is coupled to the daily rhythm in blood corticosterone level by inhibiting the hypothalamic nuclei that regulate corticosterone release by the adrenal gland (94). 22 Outline of this thesis The experimental chapters of this thesis can be divided in two parts. In the first part, consisting of chapters 2 to 5, we used electrophysiological recordings in the mouse SCN to investigate the regulation of behavior by the SCN. The second part, consisting of chapters 6 to 8, investigates how disturbances of the circadian system are related to changes in behavioral activity and disease. Part I - Regulation of behavioral activity by the SCN In chapter 2, we investigated how changes in the neuronal network of the SCN allow animals to adapt their daily behavioral activity pattern to seasonal differences in day 23 1 General introduction Anatomical studies have shown that efferent connections from the SCN originate in both ‘core’ VIP and ‘shell’ AVP positive SCN neurons. The distribution of VIP and AVP positive axons overlaps, and they jointly terminate in several brain areas in the hypothalamus, thalamus and basal forebrain (23). Areas innervated by the SCN include the sub-paraventricular zone (sPVZ), essential for circadian behavioral rhythms(84), the sleep-regulating ventro-lateral preoptic area (VLPO) (95), the preoptic areas in the anterior hypothalamus that regulate body temperature (96) and the dorsomedial hypothalamic nucleus (DMH) that regulates metabolism and corticosterone levels. A major function of the SCN is to coordinate that metabolic processes in the body are adjusted to the demands posed by the daily rhythms in behavior and food intake. During the active phase, the body has increased demands for glucose to power muscle and brain activity. In animals and humans, the body is prepared for waking up by the daily peak in the release of corticosteroid hormones by the adrenal glands. In the hours before waking up, corticosteroid levels in the blood start to increase, causing an increase in the blood glucose level that prepares the body for activity. The rhythmic release of corticosteroids is driven by the SCN via a dual pathway: (1) AVP, released by the SCN during the day into the SPVZ and DMH regulates the release of Corticotropin-releasing hormone (CRH) by the paraventricular nucleus, the releasing factor that stimulates the pituitary to release adrenocorticotrophic hormone (ACTH) into the bloodstream; (2) The SCN modifies the sensitivity of the adrenal glands for ACTH via a multisynaptic neuronal connection through the autonomic nervous system (97, 98). Apart from the regulation of blood glucose levels via the corticosterone pathway, the SCN influences glucose production by the liver via the sympathetic nervous system and the orexin system (99-101) and modulates the release of insulin by the adrenal glands via the parasympathetic system (99). 1 General introduction length. We exposed animals to short day photoperiods to simulate winter and long day photoperiods to simulate summer. After animals had adapted their behavior to the artificial summer and winter days, we recorded the SCN neuronal population activity pattern in the SCN in vivo and in vitro and found differences between short and long days. Using a detailed analysis of the activity pattern of both individual SCN neurons and small subpopulations, we tested whether the observed changes in the SCN output pattern is a consequence of changes of the activity pattern of individual neurons, or a result of alterations in synchronization in the SCN network. The experiments described in chapter 3 investigate how SCN activity is related to the time of the transition between the active and rest period. SCN neuronal activity displays a 24 hour rhythm with high activity in the day. Towards the end of the day the neuronal activity level starts to decrease, reaching a trough activity level in the early night, from where it will start increasing again in the late night and early morning. This circadian rhythm in electrical activity is communicated to other brain regions and leads to a division of behavioral activity into an active (waking) and an inactive (sleeping) phase. In mice as well as in humans, a transition between the two phases occurs in the morning and in the evening. To investigate at what precise level of SCN activity the transitions between the waking and resting phase occurred, we implanted mice with microelectrodes in the SCN and performed long-term, simultaneous recordings of SCN neuronal activity and behavioral activity. Using the obtained dataset, we analyzed what SCN firing rate levels correspond to the onset of active period and the onset of the resting period. These in vivo recordings were performed under three different day lengths, to investigate whether there are seasonal differences in the SCN firing rate level at the time of the transition between activity and rest. Chapter 4 investigates how behavioral activity influences the waveform of SCN neuronal activity. Although previous studies have presented evidence that behavioral activity can modulate SCN firing rate levels, it was not known whether this effect depends on specific types and/or intensities of behavior. It was also not clear what the function of these feedback effects may be and whether they have a meaningful impact on the waveform of the SCN output rhythm. To address these questions, we used longterm in vivo SCN recordings in combination with video observation of the animals behavior. Using this data, we analyzed if the presence and strength of SCN MUA changes depends on specific types or intensity of behavior. Furthermore, we performed a set of mild manipulations, to see whether evoked behavioral activity would have a similar effect on SCN firing rate compared to spontaneous activity. After observing that the SCN neuronal activity rhythm governs the transition between the resting and active period and thereby determines the daily duration of the activity period, we investigated in chapter 5 whether the SCN may also be involved in regulating the level of activity within the active period. To test this hypothesis, we 24 Part II - Deterioration of circadian rhythms and disease In chapter 6, we collaborated with the research group of professor Albrecht in Fribourg (Switzerland) to investigate the relationship between the circadian system and the mesolimbic reward system. The reward system of the brain consist of several subcortical structures that are connected in a network that gets activated whenever an animal has a rewarding stimulus, e.g. finding food or having intercourse. The main neurotransmitter in this system is dopamine. The reward system in general, and specifically dopamine, have been linked to psychiatric disorders such as depression and also to substance abuse and addiction. Several lines of evidence suggest a link between the circadian system and mesolimbic dopamine neurotransmission. Depression in patients often coincides with rhythm and sleep disturbances (2, 102, 103), and mutations in clock genes lead to alterations in the response to cocaine (104). Little was known however, about the mechanisms linking the circadian system and the dopaminergic reward system. We investigated a possible link between per2 gene expression and the regulation of striatal dopamine levels through mono-amine oxidase A (MAOA), the enzyme responsible for the degradation of dopamine. Using per2 expression analysis we were able to demonstrate that per2 regulates MAOA expression, leading to increased dopamine release in the mesolimbic reward system of per2 mutant mice. We used behavioral tests and in vivo electrophysiological recordings in the striatum to explore the consequences of the altered dopamine levels in these mice and observed behavioral changes and altered electrophysiological responses to antidepressant drugs. There is evidence that circadian rhythm disturbances are related to metabolic disorders such as type 2 diabetes. Several studies investigating this link have shown metabolic abnormalities in mice carrying mutations in clock genes (105-107). Furthermore, a recent study found that a specific destruction of SCN tissue causes significant metabolic abnormalities and hepatic insulin resistance (108). Importantly, several studies show that SCN function deteriorates during aging (109, 110), leading to a decrease in amplitude of the rhythmic timing signal generated by the SCN. In 25 1 General introduction exposed animals to short (22 hours) or long (26 hour) light-dark cycles. Using implanted microelectrodes, we observed that such changes in the light-dark cycle resulted in considerable changes in the waveform of the SCN firing rate rhythm. If SCN neuronal activity levels are in fact involved in regulating the level of behavioral activity, the observed changes in the circadian waveform of SCN neuronal activity should result in changes in the behavioral activity distribution within the active period, which we tested in our dataset. 1 General introduction chapter 7, we investigated whether a decrease in SCN amplitude affects metabolism and may contribute to the increased prevalence of type 2 diabetes mellitus in elderly people. To test the effects of SCN amplitude on metabolism, we disrupted the circadian rhythm of adult mice by exposing them to constant light. Using implanted micro-electrodes in the SCN, we observed that this treatment led to a reduction of amplitude of the SCN neuronal activity rhythm comparable to the amplitude reduction observed during aging. The metabolic effects of constant light treatment were investigated using metabolic cage assays and hyperinsulinemic-euglycemic clamp analysis. An additional metabolic challenge was provided to part of the animals by feeding them with a high-fat diet. The results showed that disrupting SCN amplitude leads to body weight gain and a loss of circadian rhythms in metabolism and insulin sensitivity. Whereas chapters 6 and 7 investigated the adverse effects of genetic and environmental disruption of circadian rhythms on disease susceptibility, it may also be that disease-related physiological changes adversely affect the function of the circadian system. For instance in patients with renal failure, sleep disorders are frequently observed. In chapter 8 we collaborated with the department of nephrology in the LUMC to investigate whether the circadian rhythm of behavioral activity is affected in a mouse model for chronic renal failure (CRF). In these mice, a surgical procedure was used to disrupt kidney function, causing significantly increased serum urea levels and anemia compared to sham-operated mice. Both mice with disrupted kidney function as well as sham-operated mice were housed under constant darkness while their behavioral activity was monitored in order to test the capacity of the circadian system to maintain a circadian cycle of activity and rest under physiological conditions mimicking chronic renal failure in patients. The results showed that the circadian rest-activity cycle was not impaired in the CRF animals compared to shamoperated mice, indicating that uremic toxins do not impair the function of the circadian pacemaker in the SCN. 26 References 1. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. 18. 19. 20. 21. 22. 23. 24. 25. 26. 27. 28. 29. 27 1 General introduction 2. 3. 4. 5. Bromundt V et al. (2011) Sleep-wake cycles and cognitive functioning in schizophrenia. Br J Psychiatry 198:269– 276. McClung CA (2007) Clock genes and bipolar disorder: implications for therapy. Pharmacogenomics 8:1097–1100. Germain A, Kupfer DJ (2008) Circadian rhythm disturbances in depression. Hum Psychopharmacol 23:571–585. de D'Ortous de Mairan J-J (1729) Observation Botanique. Histoire de l'Académie royale des sciences:35–36. Aschoff J (1955) Tagesperiodik bei Mäusestämmen unter konstanten Umgebungsbedingungen. Pflugers Arch:51– 59. Pittendrigh CS (1960) Circadian Rhythms and the Circadian Organization of Living Systems. Cold Spring Harb Symp Quant Biol:159–184. Aschoff J (1960) Exogenous and Endogenous Components in Circadian Rhythms. Cold Spring Harbor Symposia on Quantitative Biology 25:11–28. Pittendrigh CS, Minis DH (1964) The entrainment of circadian oscillations by light and their role as photoperiodic clocks. The American Naturalist 98:261–294. DeCoursey PJ (1964) Function of a light response rhythm in hamsters. J Cell Physiol 63:189–196. DeCoursey P (1960) Daily Light Sensitivity Rhythm in a Rodent. Science 131:33–35. DeCoursey P (1961) Effect of light on the circadian activity rhythm of the flying squirrel, Glaucomis volans. Journal of Comparative Physiology A: Neuroethology 44:331–354. Watson JD, Crick FH (1953) Molecular structure of nucleic acids; a structure for deoxyribose nucleic acid. Nature 171:737–738. Konopka RJ, Benzer S (1971) Clock mutants of Drosophila melanogaster. Proc Natl Acad Sci USA 68:2112–2116. Stephan FK, Zucker I (1972) Circadian rhythms in drinking behavior and locomotor activity of rats are eliminated by hypothalamic lesions. Proc Natl Acad Sci USA 69:1583–1586. Moore RY, Eichler VB (1972) Loss of a circadian adrenal corticosterone rhythm following suprachiasmatic lesions in the rat. Brain Res 42:201–206. Inouye ST, Kawamura H (1979) Persistence of circadian rhythmicity in a mammalian hypothalamic “island” containing the suprachiasmatic nucleus. Proc Natl Acad Sci USA 76:5962–5966. Green DJ, Gillette R (1982) Circadian rhythm of firing rate recorded from single cells in the rat suprachiasmatic brain slice. Brain Res 245:198–200. Shibata S, Oomura Y, Kita H, Hattori K (1982) Circadian rhythmic changes of neuronal activity in the suprachiasmatic nucleus of the rat hypothalamic slice. Brain Res 247:154–158. Groos G, Hendriks J (1982) Circadian rhythms in electrical discharge of rat suprachiasmatic neurones recorded in vitro. Neurosci Lett 34:283–288. Ralph MR, Foster RG, Davis FC, Menaker M (1990) Transplanted suprachiasmatic nucleus determines circadian period. Science 247:975–978. Ralph MR, Menaker M (1988) A mutation of the circadian system in golden hamsters. Science 241:1225–1227. Paxinos G, Franklin KBJ (2001) The Mouse Brain in Stereotaxic Coordinates (Academic Press, San Diego). 2nd Ed. Abrahamson EE, Moore RY (2001) Suprachiasmatic nucleus in the mouse: retinal innervation, intrinsic organization and efferent projections. Brain Res 916:172–191. Jarvis ED et al. (2005) Avian brains and a new understanding of vertebrate brain evolution. Nat Rev Neurosci 6:151–159. Kalsbeek A et al. (2006) SCN outputs and the hypothalamic balance of life. J Biol Rhythms 21:458–469. Hattar S, Liao HW, Takao M, Berson DM, Yau K-W (2002) Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295:1065–1070. Hattar S et al. (2003) Melanopsin and rod-cone photoreceptive systems account for all major accessory visual functions in mice. Nature 424:76–81. Hankins MW, Peirson SN, Foster RG (2008) Melanopsin: an exciting photopigment. Trends Neurosci 31:27–36. Lucas RJ, Foster RG (1999) Neither functional rod photoreceptors nor rod or cone outer segments are required for the photic inhibition of pineal melatonin. Endocrinology 140:1520–1524. 1 General introduction 30. Freedman MS et al. (1999) Regulation of mammalian circadian behavior by non-rod, non-cone, ocular photoreceptors. Science 284:502–504. 31. Lucas RJ, Freedman MS, Muñoz M, Garcia-Fernández JM, Foster RG (1999) Regulation of the mammalian pineal by non-rod, non-cone, ocular photoreceptors. Science 284:505–507. 32. Lucas RJ et al. (2003) Diminished pupillary light reflex at high irradiances in melanopsin-knockout mice. Science 299:245–247. 33. Morin LP, Allen CN (2006) The circadian visual system, 2005. Brain Res Brain Res Rev 51:1–60. 34. Chen SK, Badea TC, Hattar S (2011) Photoentrainment and pupillary light reflex are mediated by distinct populations of ipRGCs. Nature 476:92–95. 35. Meijer JH, van der Zee EA, Dietz M (1988) Glutamate phase shifts circadian activity rhythms in hamsters. Neurosci Lett 86:177–183. 36. Meijer JH, Albus H, Weidema F, Ravesloot JH (1993) The effects of glutamate on membrane potential and discharge rate of suprachiasmatic neurons. Brain Res 603:284–288. 37. Meijer JH, Watanabe K, Détàri L, Schaap J (1996) Circadian rhythm in light response in suprachiasmatic nucleus neurons of freely moving rats. Brain Res 741:352–355. 38. Colwell CS, Foster RG, Menaker M (1991) NMDA receptor antagonists block the effects of light on circadian behavior in the mouse. Brain Res 554:105–110. 39. de Vries MJ, Treep JA, de Pauw ES, Meijer JH (1994) The effects of electrical stimulation of the optic nerves and anterior optic chiasm on the circadian activity rhythm of the Syrian hamster: involvement of excitatory amino acids. Brain Res 642:206–212. 40. Michel S, Itri J, Han JH, Gniotczynski K, Colwell CS (2006) Regulation of glutamatergic signalling by PACAP in the mammalian suprachiasmatic nucleus. BMC neuroscience 7:15. 41. Beaule C et al. (2009) Temporally Restricted Role of Retinal PACAP: Integration of the Phase-Advancing Light Signal to the SCN. J Biol Rhythms 24:126. 42. Dragich JM et al. (2010) The role of the neuropeptides PACAP and VIP in the photic regulation of gene expression in the suprachiasmatic nucleus. Eur J Neurosci 31:864–875. 43. Mrosovsky N (1996) Locomotor activity and non-photic influences on circadian clocks. Biol Rev Camb Philos Soc 71:343–372. 44. Mistlberger R (2009) Food-anticipatory circadian rhythms: concepts and methods. Eur J Neurosci. 45. Antle MC, Mistlberger RE (2000) Circadian clock resetting by sleep deprivation without exercise in the Syrian hamster. J Neurosci 20:9326–9332. 46. Ciarleglio CM, Resuehr HES, McMahon DG (2011) Interactions of the serotonin and circadian systems: nature and nurture in rhythms and blues. Neuroscience 197:8–16. 47. Pickard GE et al. (1999) 5-HT1B receptor-mediated presynaptic inhibition of retinal input to the suprachiasmatic nucleus. J Neurosci 19:4034–4045. 48. Rea M, Pickard G (2000) Serotonergic modulation of photic entrainment in the syrian hamster. Biological Rhythm Research 31:284–314. 49. Biello SM, Janik D, Mrosovsky N (1994) Neuropeptide Y and behaviorally induced phase shifts. Neuroscience 62:273–279. 50. Agez L, Laurent V, Pévet P, Masson-Pévet M, Gauer F (2007) Melatonin affects nuclear orphan receptors mRNA in the rat suprachiasmatic nuclei. Neuroscience 144:522–530. 51. Sumová A, Illnerová H (1996) Melatonin instantaneously resets intrinsic circadian rhythmicity in the rat suprachiasmatic nucleus. Neurosci Lett 218:181–184. 52. Vida B et al. (2008) Oestrogen receptor alpha and beta immunoreactive cells in the suprachiasmatic nucleus of mice: distribution, sex differences and regulation by gonadal hormones. J Neuroendocrinol 20:1270–1277. 53. Karatsoreos IN, Butler MP, LeSauter J, Silver R (2011) Androgens Modulate Structure and Function of the Suprachiasmatic Nucleus Brain Clock. Endocrinology. 54. Yamazaki S, Kerbeshian MC, Hocker CG, Block GD, Menaker M (1998) Rhythmic properties of the hamster suprachiasmatic nucleus in vivo. J Neurosci 18:10709–10723. 55. Schaap J, Meijer JH (2001) Opposing effects of behavioural activity and light on neurons of the suprachiasmatic nucleus. Eur J Neurosci 13:1955–1962. 56. Deboer T, Vansteensel MJ, Détàri L, Meijer JH (2003) Sleep states alter activity of suprachiasmatic nucleus neurons. Nat Neurosci 6:1086–1090. 28 29 1 General introduction 57. Morin LP, Shivers K-Y, Blanchard JH, Muscat L (2006) Complex organization of mouse and rat suprachiasmatic nucleus. Neuroscience 137:1285–1297. 58. Colwell CS et al. (2003) Disrupted circadian rhythms in VIP- and PHI-deficient mice. Am J Physiol-Reg I 285:R939–49. 59. Hughes ATL, Piggins HD (2008) Behavioral responses of vipr2-/- mice to light. J Biol Rhythms 23:211–219. 60. Kalsbeek A, Fliers E, Hofman MA, Swaab DF, Buijs RM (2010) Vasopressin and the output of the hypothalamic biological clock. J Neuroendocrinol 22:362–372. 61. Albus H, Vansteensel MJ, Michel S, Block GD, Meijer JH (2005) A GABAergic mechanism is necessary for coupling dissociable ventral and dorsal regional oscillators within the circadian clock. Curr Biol 15:886–893. 62. Choi HJ et al. (2008) Excitatory actions of GABA in the suprachiasmatic nucleus. J Neurosci 28:5450–5459. 63. Schaap J, Pennartz CMA, Meijer JH (2003) Electrophysiology of the circadian pacemaker in mammals. Chronobiol Int 20:171–188. 64. Kuhlman SJ, McMahon DG (2006) Encoding the ins and outs of circadian pacemaking. J Biol Rhythms 21:470– 481. 65. Schaap J et al. (2003) Heterogeneity of rhythmic suprachiasmatic nucleus neurons: Implications for circadian waveform and photoperiodic encoding. Proc Natl Acad Sci USA 100:15994–15999. 66. VanderLeest HT et al. (2007) Seasonal encoding by the circadian pacemaker of the SCN. Curr Biol 17:468–473. 67. Brown TM, Banks JR, Piggins HD (2006) A novel suction electrode recording technique for monitoring circadian rhythms in single and multiunit discharge from brain slices. J Neurosci Methods 156:173–181. 68. Welsh DK, Logothetis DE, Meister M, Reppert SM (1995) Individual neurons dissociated from rat suprachiasmatic nucleus express independently phased circadian firing rhythms. Neuron 14:697–706. 69. Groos GA, Meijer JH (1985) Effects of illumination on suprachiasmatic nucleus electrical discharge. Ann N Y Acad Sci 453:134–146. 70. Meijer JH, Groos GA, Rusak B (1986) Luminance coding in a circadian pacemaker: the suprachiasmatic nucleus of the rat and the hamster. Brain Res 382:109–118. 71. Colwell CS (2011) Linking neural activity and molecular oscillations in the SCN. Nat Rev Neurosci 12:553–569. 72. Lundkvist GB, Kwak Y, Davis EK, Tei H, Block GD (2005) A calcium flux is required for circadian rhythm generation in mammalian pacemaker neurons. J Neurosci 25:7682–7686. 73. Maywood ES et al. (2006) Synchronization and maintenance of timekeeping in suprachiasmatic circadian clock cells by neuropeptidergic signaling. Curr Biol 16:599–605. 74. Welsh DK, Takahashi JS, Kay SA (2010) Suprachiasmatic nucleus: cell autonomy and network properties. Annu Rev Physiol 72:551–577. 75. Liu AC et al. (2007) Intercellular coupling confers robustness against mutations in the SCN circadian clock network. Cell 129:605–616. 76. Rohling J, Wolters L, Meijer JH (2006) Simulation of day-length encoding in the SCN: from single-cell to tissuelevel organization. J Biol Rhythms 21:301–313. 77. Chee CA, Roozendaal B, Swaab DF, Goudsmit E, Mirmiran M (1988) Vasoactive intestinal polypeptide neuron changes in the senile rat suprachiasmatic nucleus. Neurobiol Aging 9:307–312. 78. Kalló I, Kalamatianos T, Piggins HD, Coen CW (2004) Ageing and the diurnal expression of mRNAs for vasoactive intestinal peptide and for the VPAC2 and PAC1 receptors in the suprachiasmatic nucleus of male rats. J Neuroendocrinol 16:758–766. 79. Ohta H, Yamazaki S, McMahon DG (2005) Constant light desynchronizes mammalian clock neurons. Nat Neurosci 8:267–269. 80. Ohta H, Mitchell AC, McMahon DG (2006) Constant light disrupts the developing mouse biological clock. Pediatr Res 60:304–308. 81. Silver, Lesauter J, Tresco PA, Lehman MN (1996) A diffusible coupling signal from the transplanted suprachiasmatic nucleus controlling circadian locomotor rhythms. Nature 382:810–813. 82. Meyer-Bernstein EL et al. (1999) Effects of suprachiasmatic transplants on circadian rhythms of neuroendocrine function in golden hamsters. Endocrinology 140:207–218. 83. Kramer et al. (2001) Regulation of daily locomotor activity and sleep by hypothalamic EGF receptor signaling. Science 294:2511–2515. 1 General introduction 84. Abrahamson EE, Moore RY (2006) Lesions of suprachiasmatic nucleus efferents selectively affect rest-activity rhythm. Mol Cell Endocrinol 252:46–56. 85. Cheng MY et al. (2002) Prokineticin 2 transmits the behavioural circadian rhythm of the suprachiasmatic nucleus. Nature 417:405–410. 86. Zhou Q-Y, Cheng MY (2005) Prokineticin 2 and circadian clock output. FEBS J 272:5703–5709. 87. Masumoto K-H et al. (2006) Distinct localization of prokineticin 2 and prokineticin receptor 2 mRNAs in the rat suprachiasmatic nucleus. Eur J Neurosci 23:2959–2970. 88. Ren P et al. (2011) Prokineticin 2 regulates the electrical activity of rat suprachiasmatic nuclei neurons. PLoS ONE 6:e20263. 89. Prosser HM et al. (2007) Prokineticin receptor 2 (Prokr2) is essential for the regulation of circadian behavior by the suprachiasmatic nuclei. Proc Natl Acad Sci USA 104:648–653. 90. Li J-D et al. (2006) Attenuated circadian rhythms in mice lacking the prokineticin 2 gene. J Neurosci 26:11615– 11623. 91. Kraves S, Weitz CJ (2006) A role for cardiotrophin-like cytokine in the circadian control of mammalian locomotor activity. Nat Neurosci 9:212–219. 92. Reppert SM, Artman HG, Swaminathan S, Fisher DA (1981) Vasopressin exhibits a rhythmic daily pattern in cerebrospinal fluid but not in blood. Science 213:1256–1257. 93. Groblewski TA, Nunez AA, Gold RM (1981) Circadian rhythms in vasopressin deficient rats. Brain Res Bull 6:125– 130. 94. Kalsbeek A, Buijs RM, van Heerikhuize JJ, Arts M, van der Woude TP (1992) Vasopressin-containing neurons of the suprachiasmatic nuclei inhibit corticosterone release. Brain Res 580:62–67. 95. Chou TC et al. (2002) Afferents to the ventrolateral preoptic nucleus. J Neurosci 22:977–990. 96. Moore RY, Danchenko RL (2002) Paraventricular-subparaventricular hypothalamic lesions selectively affect circadian function. Chronobiol Int 19:345–360. 97. Kalsbeek A, Yi C-X, La Fleur SE, Buijs RM, Fliers E (2010) Suprachiasmatic nucleus and autonomic nervous system influences on awakening from sleep. Int Rev Neurobiol 93:91–107. 98. Kalsbeek et al. (2010) Hypothalamic control of energy metabolism via the autonomic nervous system. Ann N Y Acad Sci. 99. Kalsbeek A et al. (2008) Circadian control of the daily plasma glucose rhythm: an interplay of GABA and glutamate. PLoS ONE 3:e3194. 100. Yi C-X et al. (2009) A major role for perifornical orexin neurons in the control of glucose metabolism in rats. Diabetes 58:1998–2005. 101. Kalsbeek A et al. (2011) Circadian disruption and SCN control of energy metabolism. FEBS Lett. 102. Wirz-Justice A (2006) Biological rhythm disturbances in mood disorders. International clinical psychopharmacology 21 Suppl 1:S11–5. 103. Wirz-Justice A (2007) Chronobiology and psychiatry. Sleep Med Rev 11:423–427. 104. Abarca C, Albrecht U, Spanagel R (2002) Cocaine sensitization and reward are under the influence of circadian genes and rhythm. Proc Natl Acad Sci USA 99:9026–9030. 105. Rudic RD et al. (2004) BMAL1 and CLOCK, two essential components of the circadian clock, are involved in glucose homeostasis. PLoS Biol 2:e377. 106. Turek FW et al. (2005) Obesity and metabolic syndrome in circadian Clock mutant mice. Science 308:1043–1045. 107. Dallmann R, Touma C, Palme R, Albrecht U, Steinlechner S (2006) Impaired daily glucocorticoid rhythm in Per1 ( Brd ) mice. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 192:769–775. 108. Coomans CP et al. (2012) The Suprachiasmatic Nucleus Controls Circadian Energy Metabolism and Hepatic Insulin Sensitivity. Diabetes. 109. Nakamura TJ et al. (2011) Age-related decline in circadian output. J Neurosci 31:10201–10205. 110. Farajnia S et al. (2012) Evidence for neuronal desynchrony in the aged suprachiasmatic nucleus clock. J Neurosci 32:5891–5899. 30