* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Temporal and spatial alterations in GPi neuronal encoding might
Activity-dependent plasticity wikipedia , lookup
Brain–computer interface wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Response priming wikipedia , lookup
Time perception wikipedia , lookup
Cognitive neuroscience of music wikipedia , lookup
Multielectrode array wikipedia , lookup
Neuroplasticity wikipedia , lookup
Haemodynamic response wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Neuroeconomics wikipedia , lookup
Embodied language processing wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Neuroanatomy wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Development of the nervous system wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Mirror neuron wikipedia , lookup
Electromyography wikipedia , lookup
Neural oscillation wikipedia , lookup
Basal ganglia wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Neural coding wikipedia , lookup
Nervous system network models wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Metastability in the brain wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Pre-Bötzinger complex wikipedia , lookup
Central pattern generator wikipedia , lookup
Synaptic gating wikipedia , lookup
Optogenetics wikipedia , lookup
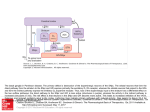
European Journal of Neuroscience, Vol. 24, pp. 1201–1208, 2006 doi:10.1111/j.1460-9568.2006.04984.x Temporal and spatial alterations in GPi neuronal encoding might contribute to slow down movement in Parkinsonian monkeys Arthur Leblois,1,2,4 Wassilios Meissner,1,3 Erwan Bezard,1 Bernard Bioulac,1,4 Christian E. Gross1,4 and Thomas Boraud1,4 1 Basal Gang, Laboratoire de Neurophysiologie, CNRS UMR 5543, Université Victor Segalen, 146 rue Léo Saignat, 33076 Bordeaux Cedex, France 2 Laboratoire de Neurophysique et Physiologie, CNRS UMR 8119, Université René Descartes, 45 rue des Saints Pères, 75270, Paris Cedex, France 3 Département de Neurologie, CHU Pellegrin, 1 place Amélie Raba-Léon, 33076 Bordeaux, France 4 Laboratoire franco-israelien de neurophysiologie et neurophysique des systèmes, Université Victor Ségalen, 146 rue Léo Saignat, 33076 Bordeaux Cedex, France Keywords: basal ganglia, globus pallidus, MPTP-monkey, Parkinson’s disease, single-unit electrophysiology Abstract Although widely investigated, the exact relationship between changes in basal ganglia neuronal activity and parkinsonian symptoms has not yet been deciphered. It has been proposed that bradykinesia (motor slowness) is related either to a modification of the activity of the globus pallidus internalis (GPi), the main output structure, or to a loss of spatial selectivity of the extrapyramidal motor system. Here we investigate the relationship between movement initiation and GPi activity in parkinsonian non-human primates. We compare neuronal encoding of movement in the normal and pathological conditions. After dopamine depletion, we observe an increased number of neurons responding to movement, with a less specific somato-sensory receptive field and a disruption of the selection mechanism. Moreover, the temporal order of the response of GPi neurons in parkinsonian animals is reversed. Indeed, whereas muscle activity and movement are delayed in parkinsonian animals, GPi neuronal responses to movement occur earlier and are prolonged, compared with normal conditions. Parkinsonian bradykinesia could thus result from an impairment of both temporal and spatial specificity of the GPi response to movement. Introduction Parkinsonian motor symptoms are classically described as (i) tremor, (ii) rigidity and (iii) akinesia (Agid, 1991). The term akinesia literally means ‘no movement’ and describes the impairment of voluntary motor performance. Akinesia encompasses a rich symptomatology ranging from true akinesia to, more often, several degrees of bradykinesia, e.g. the delay and slowing down in the execution of a voluntary movement. Whereas rigidity and tremor can be studied at rest, the analysis of voluntary movement impairment needs to be carried out during execution of a motor task. Basal ganglia (BG) activity is impaired by dopamine depletion in Parkinson’s disease. The exact role of this network in motor control remains unclear. As a delay in reaction time is part of parkinsonian bradykinesia (Benazzouz et al., 1992; Lalonde & Botez-Marquard, 1997), it was initially thought that the BG are involved in the initiation of movement (Chevalier & Deniau, 1990; Graybiel, 1990; Agid, 1991). This hypothesis is now generally rejected. First, in the normal animal, neurons of the globus pallidus internalis (GPi), the main output of the BG, only respond either once movement has begun or once electromyogram (EMG) activity starts to be modified (DeLong, Correspondence: Dr A. Leblois, 2CNRS UMR 8119, as above. E-mail: [email protected]. Received 5 April 2006, revised 30 May 2006, accepted 31 May 2006 1972; Brotchie et al., 1991; Mink & Thach, 1991a; Turner & Anderson, 1997). Second, the execution of movement is slowed by lesion and cooling of the GPi, while reaction time is not affected (Mink & Thach, 1991b; Kato & Kimura, 1992). Therefore, the most accepted current hypothesis considers the BG to act as an ‘action selection’ network under dopamine control by disinhibiting desired cortical motor program generators and by inhibiting other competitive inappropriate ones (Mink, 1996). According to this hypothesis, the ability to select one motor program relies on the precise pattern of response in the BG during the execution of a motor task. This pattern of response is impaired after dopamine depletion in Parkinson’s disease, hence the appearance of major motor symptoms, such as bradykinesia, due to an inability to select properly one motor program. However, this hypothesis does not take into account the time dimension underlying motor control (Roux et al., 2003) in spite of the possible contribution of dopamine in timing synaptic events through its modulation of GABAergic transmission (Plenz, 2003). Finally, another hypothesis is that modifications in the spontaneous firing pattern could be related to parkinsonian symptoms (Bergman et al., 1994; Wichmann & Delong, 1999). The present study addresses the issue of information timing in the BG by analysing extracellular neuronal activity of GPi sensory motor neurons in three macaque monkeys during voluntary movement before and after the induction of a moderate parkinsonian syndrome. Our aim ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd 1202 A. Leblois et al. Experiments were conducted on three female macaque monkeys (Macaca mulatta) weighing 3–4 kg (monkeys B, P and J). The animals were attended to by veterinarians skilled in healthcare and maintenance of non-human primates. Experiments were carried out in accordance with the NIH Guide for the care and Use of Laboratory Animals (1996) and the European Communities Council Directive of 24 November 1986 for care of laboratory animals (86 ⁄ 609 ⁄ EEC). reward of fruit juice. The recording protocol began once animals achieved a 90% success rate. During each recording session, at least 15 trials were carried out, during which muscle activity (EMG) was recorded using intramuscular electrodes acutely inserted into the deltoid, extensor and flexor carpi radialis, pectoralis, triceps and biceps muscles. The biceps muscle EMG recordings were chosen for analysis because this muscle was the first to react during movement (data not shown). The EMG was amplified and integrated with a time constant of 20 ms (Benazzouz et al., 1992). The onset of muscle activity is defined as the last bin in which the myogram increased by 10% for an epoch that reached a threshold of more than 2.5 times the standard deviation of the mean. For monkey J, we also recorded the onset of the movement. Response to passive movement was tested for monkeys B and P as previously described (Boraud et al., 2000). Surgical procedure and intoxication Data analysis A recording chamber was stereotactically installed under general anesthesia (ketamine hydrochloride 10–15 mg ⁄ kg i.m., Panpharma, France, and xylazine 1.5–2.5 mg ⁄ kg i.m., Sigma, St Louis, MO, USA) at an angle of 45 to the sagittal plane to facilitate the positioning of the microelectrodes. For the induction of the parkinsonian syndrome, the monkeys were treated daily (17:00 h) with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) hydrochloride (0.2 mg ⁄ kg, i.v., Sigma) dissolved in saline as previously described (Bezard et al., 2001). The monkeys were assessed daily with the Benazzouz rating scale (Benazzouz et al., 1995). Injections were ceased after the animal reached a severity of 5 ⁄ 25 on this scale (i.e. a cumulative dose of 2 mg ⁄ kg). Single unit activity was then analysed off-line in relation to passive (Boraud et al., 2000) or voluntary movement. Mean firing rate and pattern analysis at rest were performed as previously described (Boraud et al., 2001). For each trial (i), the neuron firing rate time course Fi(t) was first determined with a time bin of 10 ms by a kernel estimator (Silverman, 1986) in which the spike times itT itj iti were convolved with a kernel function K(t): j Fi(t) ¼ Rnj ¼ 1 K(t ) T i ). We used a gaussian kernel K(t) ¼ exp (–t2 ⁄ (2s2)) ⁄ (s2P)), where s determined the kernel width, controlling the degree of smoothing. We took s ¼ 0.25 ⁄ F, where F is the mean firing rate of the neuron over the recording period (Baker & Gerstein, 2001). The mean firing rate of the neuron across n trials (n > 15), and aligned on the corresponding stimulus, was then calculated, to give a smoothed version of the standard peri-event time histogram (PETH) (Baker & Gerstein, 2001). Smoothed PETHs were calculated for each recorded neuron, in relation to the four following events: the GO signal, the onset of muscle activity, the onset of movement (for monkey J) and, finally, the reward. The mean and standard deviation (SD) of the mean rate estimate were determined over a baseline region (during the 500 ms preceding the GO signal). A neuron was considered responsive if it modified its firing rate estimate by more than three times the SD of the mean. The onset time was defined as the first bin of an epoch where the estimate rate was modified by 10% from the mean in the same direction (i.e. activation or suppression). When a neuron was responsive to more than one stimulus, the slope of the first three bins deflecting from the mean by more than three SD was calculated for each stimulus. The steepest slope was considered to correspond to the stimulus to which the neuron was responsive. For this study we considered only neurons whose response was correlated best to the onset of the movement or to the EMG for monkey J and to the EMG for monkeys B and P. Offset time was defined as the last bin of this epoch in which the mean firing rate had deviated by more than 10%. Neurons were classified according to the polarity of their response: either an increase or a decrease in the firing rate. Occasionally a neuron responded to a single event with a sequence of two or even three modulations of its firing rate (11.7% in the normal condition and 10.1% in the MPTP condition). In this case, the response polarity was classified according to the first modulation. was to determine the relationship between sensorimotor and temporal encoding in GPi and parkinsonian motor slowness. Materials and methods Animals Recording procedure Recordings were performed on the left hemisphere of the three monkeys. During recording sessions the monkey’s head was immobilized, and 1–4 glass-coated tungsten microelectrodes (impedance 0.2–1 MW at 1000 Hz) confined within a cylindrical guide (2.2 mm diameter) were advanced into the GP. Each electrode was separately advanced and optimally placed in the vicinity of cells. GPi neurons were first identified by on-line analysis of the electrophysiological signal at rest (see Boraud et al., 1998). The output of each electrode was amplified with a gain of 5–20k and band-pass-filtered with a 300– 8000-Hz four-pole Butterworth filter. The electrical activity recorded from each electrode was sorted and classified on-line using a templatematching algorithm (Worgotter et al., 1986), implemented by a PCbased spike sorter (MSD, Alpha-Omega Engineering, Nazareth, Israel). The spike trains detected by this system, as well as the behavioral events and other measurements of the monkey’s behavior, were recorded for off-line analysis. Behavioral task In order to investigate neuronal activity related to movement and to limit responses of cognitive nature, we designed a very simple motor task. Monkeys were seated in a primate chair in front of a panel with a single key (Fig. 1). The left arm was immobilized with Velcro straps. Trials were initiated after the monkeys touched the handle fixed to their right armrest with their right hand. After a random period (1000– 1500 ms) a green LED turned on as a GO signal., The monkeys had 1000 ms in the normal condition (1500 ms in the parkinsonian condition) to press the key with a ballistic forward movement of the right hand. It had to hold this position for 500 ms in order to receive a Histology (for methodology see Bezard et al., 2001) All animals were killed by sodium pentobarbital overdose (150 mg/ kg, i.v.), and the brains were removed quickly after death. Each brain ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 Pallidal timing and Parkinsonian bradykinesia 1203 A B Hold Go 1000 to 1500 ms C D Movement Reward 500 ms Fig. 1. Behavioural task. (A) Monkeys were seated in a recording chair with their right arm on an armrest. A red LED indicated the hold condition. (B) A green LED turned on (GO signal) 1000–1500 ms after the hold position was reached. (C) The animal had to press the button within 1000 ms in normal condition (1500 ms in parkinsonian condition) and (D), to hold the position for 500 ms, in order to receive a reward of fruit juice. Each session consisted of at least 15 successful trials. was bisected along the midline, and the two hemispheres were immediately frozen by immersion in isopentane ()45 C) and then stored at )80 C. Tissue was sectioned coronally at 20 lm in a cryostat at )17 C, thaw-mounted onto gelatin-subbed slides, dried on a slide warmer, and stored at )80 C. Histochemical investigation showed a dramatic decrease in the number of tyrosine hydroxylaseimmunoreactive neurons in the substantia nigra of the three MPTPtreated monkeys (B, )63.7%; P, )69.1%; J, )84.4%), as compared to control values obtained in four normal animals, matched for age, sex and weight. reward delivery) were made using Student’s t-test (P < 0.05). Latencies for the three animals were pooled after a one-way anova test showed that they were comparable (P > 0.5). For each monkey, the coordinates (x, y, z) of all recorded neurons were compared between the two experimental conditions (normal and MPTP) for each monkey using a one-way multivariate analysis of variance. For all three monkeys, there were no significant differences between the two conditions (P > 0.05). Results Statistical analysis The effect of passive movement on firing rate (activation or suppression) was pooled between monkeys B and P after a v2 test showed that distributions were comparable (P > 0.5). The effect of voluntary movement on firing rate (activation or suppression) was also pooled between the three monkeys after a v2 test showed that distributions were comparable (P > 0.5). The normal and MPTP conditions were then compared for frequency of distribution (v2, d ¼ 1, P < 0.05 when v2 > 3.841) (Mushiake et al., 1991). Comparisons of the different latencies (latency between stimulus and onset of muscle activity, onset and offset of muscle activity, stimulus and onset of the movement, stimulus and onset of GPi activity, muscle activity and onset of GPi response, onset of movement and onset of GPi response, onset and offset of GPi response and onset of movement and Clinical examination After MPTP injection (see Methods), the three monkeys developed a moderate akineto-rigid parkinsonian syndrome, which still allowed them to perform the motor task. This syndrome was characterized by rigidity, slowness and decrease in the number of voluntary movements, and loss of dexterity, without tremor or postural disorders (clinical score ⁄ 25: monkey B ¼ 16.8 ± 1.4, P ¼ 15.3 ± 0.9, J ¼ 19.3 ± 1.3). This parkinsonian syndrome remained stable until the end of the experiment. Neuron database We recorded 144 GPi neurons in the normal condition, 61 in monkey B over 14 recording days, 50 in monkey P over 9 days and 33 in ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 1204 A. Leblois et al. Table 1. Summary of individual and mean data Individual results Parameter Monkey B (n) Monkey P (n) Monkey J (n) Mean results for monkeys B+P+J Firing rate at rest Normal MPTP 55.8 ± 16.6 76.5 ± 22.3* (61) (43) 62.1 ± 16.7 86.6 ± 26.3* (50) (55) 56.4 ± 17.9 57.9 ± 24.4 (33) (20) 57.3 ± 17.3 77.9 ± 26.4* (144) (118) Firing patterns (random ⁄ regular ⁄ bursting) Normal 29 ⁄ 16 ⁄ 16 MPTP 12 ⁄ 15 ⁄ 16 (61) (43) 15 ⁄ 14 ⁄ 21 10 ⁄ 16 ⁄ 29 (50) (55) 12 ⁄ 17 ⁄ 4 2 ⁄ 11 ⁄ 7* (33) (20) 56 ⁄ 47 ⁄ 41 24 ⁄ 42 ⁄ 52* (144) (118) Responses with passive movements Normal response (%) Response with MPTP (%) Normal I ⁄ A ratio I ⁄ A ratio with MPTP Normal multijoint response (%) Multijoint responses with MPTP (%) 45.9 69.8* 0.87 0.36* 7.1 56.7* (28 ⁄ 61) (30 ⁄ 43) (13 ⁄ 15) (8 ⁄ 22) (2 ⁄ 28) (17 ⁄ 30) 52.0 64.3 0.62 0* 11.5 30.0 (26 ⁄ 50) (27 ⁄ 42) (10 ⁄ 16) (0 ⁄ 27) (3 ⁄ 26) (9 ⁄ 27) n.a. n.a. n.a. n.a. n.a. n.a. – – – – – – 48.6 67.1* 0.74 0.16* 9.3 45.6* (54 ⁄ 111) (57 ⁄ 85) (23 ⁄ 31) (8 ⁄ 49) (5 ⁄ 54) (26 ⁄ 57) Responses with voluntary movements Normal response (%) Response with MPTP (%) Normal I ⁄ A ratio I ⁄ A ratio with MPTP Normal Go–Mv delay (ms) Go–Mv delay with MPTP (ms) Normal Go–EMG delay(ms) Go–EMG delay with MPTP (ms) Normal Mv duration (ms) Mv duration with MPTP (ms) Normal EMG duration (ms) EMG duration with MPTP (ms) Normal EMG–Mv delay (ms) EMG–Mv delay with MPTP (ms) Normal Go–GPi delay (ms) Go–GPi delay with MPTP (ms) Normal GPi–EMG delay (ms) GPi–EMG delay with MPTP (ms) Normal GPi–Mv delay (ms) GPi–Mv delay with MPTP (ms) Normal GPi duration (ms) GPi duration with MPTP (ms) 44.3 81.4* 0.29 0.06 n.a. n.a. 157 ± 16 241 ± 27* n.a. n.a. 865 ± 13 1654 ± 10* n.a. n.a. 148 ± 9 60 ± 12* )6 ± 18 )235 ± 22* n.a. n.a. 219 ± 15 387 ± 8* (27 ⁄ 61) (35 ⁄ 43) (6 ⁄ 21) (2 ⁄ 33) – – (27) (35) – – (27) (35) – – (27) (35) (27) (35) – – (27) (35) 46.0 58.2 0.53 0.1* n.a. n.a. 119 ± 12 320 ± 23* n.a. n.a. 831 ± 13 1639 ± 33* n.a. n.a. 119 ± 12 137 ± 52 2 ± 28 )173 ± 21* n.a. n.a. 220 ± 13 397 ± 29* (23 ⁄ 50) (32 ⁄ 55) (8 ⁄ 15) (3 ⁄ 29) – – (23) (32) – – (23) (32) – – (23) (32) (23) (32) – – (23) (32) 81.8 60.0 0.93 0.0* 281 ± 15 480 ± 35* 138 ± 15 284 ± 4* 557 ± 13 884 ± 27* 850 ± 30 1605 ± 21* 142 ± 18 202 ± 21* 140 ± 18 81 ± 4* )7 ± 26 )204 ± 4* )144 ± 18 )406 ± 27* 219 ± 11 371 ± 3* (27 ⁄ 33) (12 ⁄ 20) (13 ⁄ 14) (0 ⁄ 12) (27) (12) (27) (12) (27) (12) (27) (12) (27) (12) (27) (12) (27) (12) (27) (12) (27) (12) 53.5 66.9* 0.54 0.07* 281 ± 15 480 ± 35* 139 ± 21 280 ± 48* 557 ± 13 884 ± 27* 849 ± 24 1645 ± 23* 142 ± 18 202 ± 21* 136 ± 17 74 ± 23* )3 ± 28 )206 ± 34* )144 ± 18 )406 ± 27* 220 ± 14 384 ± 19* (77 ⁄ 144) (79 ⁄ 118) (27 ⁄ 50) (5 ⁄ 74) (27) (12) (77) (79) (27) (79) (77) (79) (27) (12) (77) (79) (77) (79) (27) (12) (77) (79) (n) Parameters are provided for each monkey (B, P or J) in each of the experimental conditions (normal and MPTP) where available. Mean values of pooled data are also provided. When appropriate, the number of neurons is provided in parentheses. From top to bottom: firing rate at rest (FR), firing pattern, parameters of the response to passive movement and of the response to the motor task (voluntary movements). For the response to passive movement, the percentage of neurons responding is provided along with the ‘inhibited to activated’ ratio (I ⁄ A) and the percentage of neurons responding to the manipulation of more than one joint. For the response to voluntary movement, the percentage of neurons responding, the I ⁄ A ratio and the latency between the several parameters of the task (mean ± SD) are provided. Mv, onset of movement; Mv-duration, duration of movement; EMG, onset of muscle response; EMG-duration, duration of this response; GPi, onset of GPi neurons response; GPi duration, duration of this response. *MPTP vs. normal, P < 0.05. monkey J over 8 days. One hundred and eighteen neurons were recorded in the MPTP-treated condition, 43 in monkey B over 17 days, 55 in monkey P over 15 days and 20 in monkey J over 10 days. In monkeys B and P the recordings in MPTP-treated condition were performed 48 and 42 days after MPTP intoxication, respectiveley, whereas in monkey J they were performed immediately after MPTP intoxication. The mean firing rate of GPi neurons was significantly increased after MPTP-induced dopaminergic depletion (77.9 ± 17.3 vs. 57.3 ± 17.3 spikes ⁄ s, P < 0.05). The number of bursting neurons also increased in the pathological condition (28.5%, 41 ⁄ 144 vs. 44.1%, 52 ⁄ 118). Neurons considered were restricted to a volume of 6 · 3 · 6 mm (sagittal · lateral · depth) for monkey B, 5.5 · 3.5 · 6 mm for monkey P and 7 · 6 · 6 mm for monkey J inside the GPi nucleus. There were no significant differences in the ratio of neurons responding to the task between the different locations of the recordings. Mean and individual numerical data are summarized in Table 1. Passive movements In monkeys B and P the number of GPi neurons responding to passive movement increased significantly after MPTP treatment (67.1 vs. 48.6%). The ratio of neurons responding to several joint movements increased also (45.6 vs. 9.3%). These data are consistent with previous results obtained in fully akinetic monkeys (Boraud et al., 2000). Motor task Voluntary movement consisted of the execution of a simple motor task (Fig. 1). Biceps EMG was monitored during the movement for the three monkeys. Onset of movement was recorded in monkey J. Modifications of the response to the movement task In the normal condition, 53.5% (77 ⁄ 144) of GPi neurons were responsive to either movement onset or EMG onset (Figs 2 and 3A). ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 Pallidal timing and Parkinsonian bradykinesia 1205 A sp/s Normal 100 221102/2_1 50 0 sp/s -1000 100 Mv Onset 1000 ms Mv Onset 1000 ms 281102/2_4 50 0 -1000 B sp/s MPTP 040203/2_13 100 50 0 -1000 C 100 EMG signal (mV) 1000 ms Mv Onset 50 MPTP Normal 0 0 Go signal 400 800 1200 1600 2000 Time (ms) Fig. 2. PETH of GPi neuronal activity linked to voluntary movement onset (Mv Onset) in monkey J. (A) Example of two GPi neurons in normal condition. Above, neuron 221102 ⁄ 2–1 responded with an increase (50 trials) and below, neuron 281102 ⁄ 2–4 responded with a decrease in firing rate (62 trials). (B) Example of GPi neuron activity in MPTP condition (neuron 040203 ⁄ 2–13, 45 trials). For each trial, a green circle on the raster line shows the GO signal, a red circle the onset of the EMG and a blue one the reward. Note that the reward often occurred after the end of the time window chosen for display. (C) example of biceps EMG aligned on the GO signal (t ¼ 0) in the normal (black line) and MPTP (grey line) conditions. ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 1206 A. Leblois et al. A 40 Normal MPTP number of neurons Only 11.7% of the responding neurons (10.1% in the parkinsonian condition) displayed multiple responses to one of these stimuli. Thus, all neurons were classified according to the polarity of their earliest response (see Methods). The mean firing frequency decreased significantly in 35.1% (27 ⁄ 77) and increased significantly in 64.9% (50 ⁄ 77) of GPi neurons, resulting in an inhibited ⁄ activated (I ⁄ A) ratio of 0.54. After MPTP intoxication, the number of GPi neurons responding to voluntary movement increased significantly to 66.9% (79 ⁄ 118, Fig. 3A). In addition the I ⁄ A ratio decreased significantly to 0.07 as 6.3% (5 ⁄ 79) of GPi neurons were inhibited whereas 93.7% (74 ⁄ 79) were activated. 20 10 0 Impairment of temporal encoding Go Signal 100 200 300 B number of neurons 30 20 10 0 -400 -300 -200 -100 EMG Onset 100 -400 -300 -200 -100 Mv Onset C 16 14 number of neurons In the normal condition the onset of biceps muscle activity (mean ± SD; Fig. 3C) occurred 139 ± 21 ms after the GO signal and lasted 849 ± 24 ms. The mean delay between the onset of muscle activity and the onset of movement was 142 ± 18 ms. The mean GPi response occurred 136 ± 17 ms after the GO signal and lasted 220 ± 14 ms. In monkey J, the onset of movement occurred 281 ± 15 ms after the GO signal and the movement lasted 557 ± 13 ms. GPi activity began 3 ± 28 ms before the onset of muscle activity and 144 ± 18 ms before the onset of actual movement. After MPTP intoxication (Fig. 3B), both the onset of biceps EMG activity and movement recorded in monkey J were significantly delayed to 280 ± 48 ms and 480 ± 35 ms after the GO signal, respectively, and lasted longer with 1645 ± 23 ms and 884 ± 27 ms duration, respectively. The mean delay between onset of muscle activity and movement was significantly increased to 202 ± 21 ms. Whereas these data confirm previous reports (Benazzouz et al., 1992), analysis of the response distribution of GPi neurons in relation to the onset of movement provided unexpected insights. The activity of GPi neurons was modified significantly earlier after the GO signal (74 ± 23 ms) and lasted significantly longer (384 ± 19 ms). The modification of firing activity of GPi neurons thus preceded the onset of muscle activity and movement onset by 206 ± 34 and 406 ± 27 ms, respectively. The timing of the GPi response to movement is therefore altered to a significant extent in the pathological situation. 30 12 10 8 6 4 2 Discussion It has been proposed that an abnormal firing pattern in the BG could be the cause of the motor symptoms observed in Parkinson’s disease (Bergman et al., 1994; Wichmann & DeLong, 1999). In the present work, we confirmed that the firing pattern of GPi neurons is modified in the pathological condition. However, the present study is the first to show that parkinsonian motor slowness is accompanied by modifications in both spatial selectivity of movement-related activity in the GPi as well as the timing of motor-related responsiveness, which must contribute to parkinsonian akinesia because of their concomitance. In the normal animal, the I ⁄ A ratio during voluntary movement was below 1 (0.54), as observed in other studies (Turner & Anderson, 1997). The GPi is a GABAergic inhibitory output structure of the basal ganglia that projects to the motor cortex through the motor thalamic nuclei. An I ⁄ A ratio below 1 has therefore been associated with the hypothesis that the function of the BG is to select a desired movement among cortically stored motor programs and to inhibit other competing programs (Mink, 1996; Boraud et al., 2000, 2002). The I ⁄ A ratio decreased dramatically to 0.07 following MPTP intoxication. Although not significant in one animal, the decrease in the I ⁄ A ratio was strong and consistent among the three animals. It can be interpreted as the inability for the GPi to disinhibit its thalamo- 0 -500 Time (ms) Fig. 3. (A) Distribution histogram of time lapse between Go signal and onset of GPi neuronal response, in normal and MPTP conditions. (B) Distribution histogram of time lapse between onset of the biceps response (EMG) and onset of GPi neuronal response, in normal and MPTP conditions. (C) Distribution histogram of time lapse between onset of movement (Mv Onset) and onset of GPi neuronal response, in normal and MPTP conditions. Note the smaller number of neurons because the onset of movement was recorded only in monkey J. cortical targets, instead exerting widespread inhibition. The BG are thus unable to perform the selection of motor programs correctly. This hypothesis has already been proposed by our group with regard to the analysis of passive movement (Boraud et al., 2000). In severely impaired parkinsonian animals, the number of GPi neurons responsive to movement increased, as did the number of neurons responding to the manipulation of several joints, indicating a concomitant loss in somato-sensory selectivity and the probable recruitment of other GPi neurons previously involved in different tasks (Filion et al., 1988; Boraud et al., 2000). In the present study, although our animals were ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 Pallidal timing and Parkinsonian bradykinesia 1207 only moderately parkinsonian, we made very similar observations. After MPTP-induced dopaminergic depletion, the number of neurons that responded to the manipulation of more than one joint was significantly increased from 9.3 to 45.6%. This increase was consistent in both animals considered but failed to be significant in one of them, probably owing to a too small sample size. The ratio of neurons involved in the motor task was significantly increased from 53.3 to 66.9%. This increase was observed in two of the three animals, but significant only in one of them. Note, however, that the total number of recorded neurons was quite low in the only animal displaying a decreased number of neurons involved in the task. This apparent inconsistency should thus be interpreted with caution. The present study may include neuronal activities related to the activation of various muscle groups and ⁄ or cognitive processes during the execution of the task. It is thus possible that this global increase in the number of neurons responding to the task is due to a change in the motor strategy adopted by the monkey. Finally, the GPi shifted from a state in which information provided by the structure was selective, i.e. (i) disinhibition of specific pathway against a background of tonic inhibition, (ii) selective somato-sensory encoding and (iii) selective neuronal territory involved in the task to a physiopathological state with (i) non specific hyper-inhibition during the movement, (ii) less selective somato-sensory encoding and (iii) more neuronal territory involved in the task. The pathological disruption of the temporal pattern of activity in the GPi during the task constitutes the main interest of the present work. In the normal animal, GPi neurons modified their firing activity around the onset of muscular activity. After MPTP intoxication, the overwhelming majority of GPi neurons began to modify their firing considerably earlier than the onset of muscle activity. Because the latter has been shown to begin later in MPTP-treated monkeys than in normal animals during a motor task (Benazzouz et al., 1992), this apparent change in the temporal relationship between GPi neurons and motor outputs may be corollary to the enhancement of inhibition exerted by the GPi neurons on their target neurons, with a consequent delay and slowness in motor output. More surprisingly, after dopamine depletion, GPi neurons began to modify their activity significantly closer to the GO signal (74 ± 23 vs. 136 ± 17 ms) in two of the three monkeys, and the duration of the GPi response was longer in the three monkeys. An earlier response is thus associated with motor slowness, which appears to be counter-intuitive. However, we have previously shown similar modifications in the timing of the task-related activity in the motor cortex, where neurons discharge earlier and longer after MPTP intoxication than in the normal animal (Doudet et al., 1990). In the present study it is worth noting that monkey P, in which the GPi response did not occur significantly closer to the GO signal after MPTP treatment, had the longest reaction times (time interval between the GO signal and the EMG onset). By contrast, the delay between GPi response and EMG onset was similar in the three monkeys in each condition, and if reaction time may be partially related to other disruption, movement slowness is related to this premature response. Parkinsonian bradykinesia may thus be related to a premature activation of the cortex–BG loop during movement. In parkinsonian patients or MPTP-treated monkeys, the GPi returns neural information prematurely to cortical motor areas via thalamic motor nuclei, which might subsequently delay movement initiation (Benazzouz et al., 1992). Until now, the most accepted hypothesis considers the BG selectively to facilitate or inhibit actions generated in the cortex (Mink, 1996). The late latency of the neuronal response in the GPi with respect to movement initiation seems to argue against this hypothesis. However, we showed in a recent theoretical study that action selection may follow the early non-selective cortical activation (Leblois et al., 2006). In this framework, the latency of the response with respect to the onset of the non-selective cortical input is longer in the GPi than in the cortex, although the BG network plays a crucial role in action selection. Moreover, we showed that the set of GPi neurons involved in the disinhibition of the desired motor program shrinks after dopamine depletion whereas the inhibition of the remaining of the thalamo-cortical circuits is spread. This spread inhibition occurs earlier before movement and lasts longer than in the normal condition, leading to a broad inhibition of cortical motor generators and to slowing of the intended movements. Bar-Gad & Bergman (2001) proposed that the role of BG is to reduce the dimensionality of sparse cortical information. In the specific case of motor control, the BG would act more as an information selection structure rather than one involved in action selection. Our findings show that temporal and spatial alterations in these information selection processes could be responsible for movement slowness in MPTP-treated animals. Acknowledgements We thank S. Dovero and C. Imbert for their technical assistance. We are indebted to P. Brown for his helpful comments on the manuscript. J. Simmers kindly edited the English text. W.M. was a Marie Curie Fellow of the European Community (HPMF-2001–01300). This work was supported by the CNRS, Université Victor Segalen Bordeaux 2, the IFR de Neurosciences (INSERM N8 – CNRS N13) and by CNRS grant CTI01-01. Abbreviations BG, basal ganglia; EMG, electromyogram; GPi, globus pallidus pars interna; I ⁄ A, inhibited to activated ratio; MPTP, 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine; PETH, peri-event time histogram. References Agid, Y. (1991) Parkinson’s disease: pathophysiology. Lancet, 337, 1321– 1323. Baker, S.N. & Gerstein, G.L. (2001) Determination of response latency and its application to normalization of cross-correlation measures. Neural Comput., 13, 1351–1377. Bar-Gad, I. & Bergman, H. (2001) Stepping out of the box: information processing in the neural networks of the basal ganglia. Curr. Opin. Neurobiol., 11, 689–695. Benazzouz, A., Boraud, T., Dubédat, P., Boireau, A., Stutzmann, J.M. & Gross, C. (1995) Riluzole prevents MPTP-induced parkinsonism in the rhesus monkey: a preliminary study. Eur. J. Pharmacol., 284, 299–307. Benazzouz, A., Gross, C., Dupont, J. & Bioulac, B. (1992) MPTP induced hemiparkinsonism in monkeys: behavioral, mechanographic, electromyographic and immunohistochemical studies. Exp. Brain Res., 90, 116–120. Bergman, H., Wichmann, T., Karmon, B. & DeLong, M.R. (1994) The primate subthalamic nucleus. II. Neuronal activity in the MPTP model of parkinsonism. J. Neurophysiol., 72, 507–520. Bezard, E., Dovero, S., Prunier, C., Ravenscroft, P., Chalon, S., Guilloteau, D., Crossman, A.R., Bioulac, B., Brotchie, J.M. & Gross, C.E. (2001) Relationship between the appearance of symptoms and the level of nigrostriatal degeneration in a progressive 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine-lesioned macaque model of Parkinson’s disease. J. Neurosci., 21, 6853–6861. Boraud, T., Bezard, E., Bioulac, B. & Gross, C. (2000) Ratio of inhibited-toactivated pallidal neurons decreases dramatically during passive limb movement in the MPTP-treated monkey. J. Neurophysiol., 83, 1760–1763. Boraud, T., Bezard, E., Bioulac, B. & Gross, C.E. (2001) Dopamine agonistinduced dyskinesias are correlated to both firing pattern and frequency alteration of pallidal neurons in the MPTP-treated monkey. Brain, 124, 546– 557. Boraud, T., Bezard, E., Bioulac, B. & Gross, C.E. (2002) From single extracellular unit recording in experimental and human parkinsonism to the development of a functional concept of the role played by the basal ganglia in motor control. Prog. Neurobiol., 66, 205–283. ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208 1208 A. Leblois et al. Boraud, T., Bezard, E., Guehl, D., Bioulac, B. & Gross, C. (1998) Effects of LDOPA on neuronal activity of the Globus Pallidus externalis (GPe) and Globus Pallidus internalis (GPi) in the MPTP-treated monkey. Brain Res., 787, 157–160. Brotchie, P., Iansek, R. & Horne, M.K. (1991) Motor function of the monkey globus pallidus. I. Neuronal discharge and parameters of movement. Brain, 114, 1667–1683. Chevalier, G. & Deniau, J.M. (1990) Disinhibition as a basic process in the expression of striatal functions. Trends Neurosci., 13, 277–280. DeLong, M.R. (1972) Activity of basal ganglia neurons during movement. Brain Res., 40, 127–135. Doudet, D.J., Gross, C., Arluison, M. & Bioulac, B. (1990) Modifications of precentral cortex discharges and EMG activity in monkey with MPTP-induced lesions of DA nigral neurons. Exp. Brain Res., 80, 177–188. Filion, M., Tremblay, L. & Bedard, P.J. (1988) Abnormal influences of passive limb movement on the activity of globus pallidus neurons in Parkinson’s disease. Brain Res., 444, 165–176. Graybiel, A.M. (1990) Neurotransmitters and neuromodulators in the basal ganglia. Trends Neurosci., 13, 244–254. Kato, M. & Kimura, M. (1992) Effects of reversible blockade of basal ganglia on a voluntary arm movement. J. Neurophysiol., 68, 1516–1534. Lalonde, R. & Botez-Marquard, T. (1997) The neurobiological basis of movement initiation. Rev. Neurosci., 8, 35–54. Leblois, A., Boraud, T., Meissner, W., Bergman, H. & Hansel, D. (2006) Competition between feedback loops underlies normal and pathological dynamics in the basal ganglia. J.Neurosci., 26, 3567–3583. Mink, J.W. (1996) The basal ganglia: focused selection and inhibition of competing motor programs. Prog. Neurobiol., 50, 381–425. Mink, J.W. & Thach, W.T. (1991a) Basal ganglia motor control. II. Late pallidal timing relative to movement onset and inconsistent pallidal coding of movement parameters. J. Neurophysiol., 65, 301–329. Mink, J.W. & Thach, W.T. (1991b) Basal ganglia motor control. III. Pallidal ablation: normal reaction time, muscle cocontraction, and slow movement. J. Neurophysiol., 65, 330–351. Mushiake, H., Inase, M. & Tanji, J. (1991) Neuronal activity in the primate premotor, supplementary, and precentral motor cortex during visually guided and internally determined sequential movements. J. Neurophysiol., 66, 705– 718. Plenz, D. (2003) When inhibition goes incognito: feedback interaction between spiny projection neurons in striatal function. Trends Neurosci., 26, 436– 443. Roux, S., Coulmance, M. & Riehle, A. (2003) Context-related representation of timing processes in monkey motor cortex. Eur J. Neurosci., 18, 1011–1016. Silverman, B.W. (1986) Density Estimation for Statistic and Data Analysis. Chapman & Hall, London. Turner, R.S. & Anderson, M.E. (1997) Pallidal discharge related to the kinematics of reaching movements in two dimensions. J. Neurophysiol., 77, 1051–1074. Wichmann, T. & DeLong, M.R. (1999) Oscillations in the basal ganglia. Nature 12, 621–622. Worgotter, F., Daunicht, W.J. & Eckmiller, R. (1986) An on-line spike form discriminator for extracellular recordings based on an analog correlation technique. J. Neurosci. Methods, 17, 141–151. ª The Authors (2006). Journal Compilation ª Federation of European Neuroscience Societies and Blackwell Publishing Ltd European Journal of Neuroscience, 24, 1201–1208