* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Caudal Topographic Nucleus Isthmi and the Rostra1
Nonsynaptic plasticity wikipedia , lookup
Multielectrode array wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Node of Ranvier wikipedia , lookup
Central pattern generator wikipedia , lookup
Electrophysiology wikipedia , lookup
Single-unit recording wikipedia , lookup
Holonomic brain theory wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Biological neuron model wikipedia , lookup
Apical dendrite wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Anatomy of the cerebellum wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Synaptogenesis wikipedia , lookup
Development of the nervous system wikipedia , lookup
Circumventricular organs wikipedia , lookup
Neuroanatomy wikipedia , lookup
Sexually dimorphic nucleus wikipedia , lookup
Nervous system network models wikipedia , lookup
Optogenetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Axon guidance wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
THE JOURNAL OF COMPARATIVE NEUROLOGY 261:319-346 (1987)
Caudal Topographic Nucleus Isthmi and
the Rostra1 Nontopographic Nucleus Isthmi
in the Turtle, Pseudemys scriptu
MARTIN I. SERENO AND PHILIP S. ULINSKI
Committees on Neurobiology (M.I.S., P.S.U.) and on the Conceptual Foundations of
Science (M.I.S.), and Department of Anatomy (P.S.U.),University of Chicago,
Chicago, Illinois 60637
ABSTRACT
Isthmotectal projections i n turtles were examined by making serial
section reconstructions of axonal and dendritic arborizations that were anterogradely or retrogradely filled with HRP. Two prominent tectal-recipient
isthmic nuclei-the caudal magnocellular nucleus isthmi (Irnc) and the rostral magnocellular nucleus isthmi (Imrbexhibited strikingly different patterns of organization. Zmc cells have flattened, bipolar dendritic fields that
cover a few percent of the area of the cell plate constituting the nucleus and
they project topographically to the ipsilateral tectum without local axon
branches. The topography was examined explicitly a t the single-cell level by
using cases with two injections at widely separated tectal loci. Each Imc
axon terminates as a compact swarm of several thousand boutons placed
mainly in the upper central gray and superficial gray layers. One Imc
terminal spans less that 1%of the tectal surface. Zmr cells, by contrast, have
large, sparsely branched dendritic fields overlapped by local axon collaterals
while distally, their axons nontopographically innervate not only the deeper
layers of the ipsilateral tectum but also ipsilateral Imc. Imr receives a
nontopographic tectal input that contrasts with the topographic tectal input
to Imc.
Previous work on nucleus isthmi emphasized the role of the contralatera1 isthmotectal projection (which originates from a third isthmic nucleus
in turtles) in mediating binocular interactions in the tectum. The present
results on the two different but overlapping ipsilateral tecto-isthmo-tectal
circuits set up by Imc and Imr are discussed in the light of physiological
evidence for selective attention effects and local-global interactions in the
tectum.
Key words: tectum, superior colliculus, parabigeminal nucleus, models of
attention, stimulus-specificsurrounds
All vertebrates have a complex of nuclei in the isthmus
at the pontine-midbrain junction that is intimately interconnected with the optic tectum or superior colliculus. The
isthmic complex always receives a topographically organized projection from the ipsilateral tectal lobe, and in
amphibians, reptiles, and mammals, it projects topographically to the contralateral tectal lobe. Glasser and Ingle (‘78)
and Grobstein et al. (‘78) showed that this two-stage pathway supports the binocular receptive fields observed in the
rostromedial tectum of frogs. But the isthmic complex also
projects topographically back to the ipsilateral tectum in all
0 1987 ALAN R. LISS, INC.
vertebrates. The general function of this ubiquitous return
pathway has so far been enigmatic.
The three topographic pathways described above were
identified in the course of an earlier HRP study of tectoreticular axons in turtles (Sereno, ’85). However, nontopo-
Accepted January 21,1987.
Address reprint requests to Martin Sereno, Division of Biology
216-76, California Institute of Technology, Pasadena, CA 91125.
320
M.I. SERENO AND P.S. ULINSKI
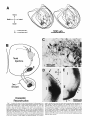
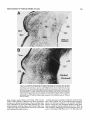
Fig. 1. A Transverse section through the caudal magnocellular nucleus
isthmi (Irnc). Note that both the rostral and caudal neuropile layers of Imc
(see also Fig. 2) are visible in this section through the middle of the nucleus.
The parvocellular nucleus isthmi (Ipj, which projects mainly to the contralateral tectal hemisphere, lies just ventromedial to Imc. B: Same-magnification view of Imc after a large but subtotal HRP injection into the ipsilateral
tectum. The dark topographic hand i n Imc consists of a mat of anterogradely
labeled fine-caliber tecto-isthmic terminals superimposed on a cluster of
retrogradely labeled Imc cells and their dendritic arborizations. In addition,
there is a less dense meshwork of very-largecaliber terminals homogeneously filling the entire nucleus. Ventral to Imc, horizontally directed collaterals arise and terminate in the dorsal part of the small-celled nucleus
(SCd); this nucleus projects to the contralateral retina.
graphic labeling also appeared in the topographically
organized caudal magnocellular nucleus isthmi (Irnc) and
in a nearby tectal-recipient nucleus identified as the rostral
magnocellular nucelus isthmi (Imr), suggesting that ipsilatera1 isthomotectal circuits had additional, as yet unrecognized, components. This paper examines the organization
of the isthmic compIex in turtles, with an emphasis on its
ipsilateral relationships. The topographic caudal isthmotectal projection is treated first. Second, the nontopographic
rostral isthmotectal projection is considered. The possible
involvement of these ipsilateral pathways in spatial selective attention and local-global computations is then explored.
MATERIALS AND METHODS
Twenty-five pond turtles (Pseudemys scriptu) weighing
0.5-1.5 kg were used. Animals were anesthetized with a
small (0.3 mlkg) dose of Brevital (Wang et al., '77) and then
placed in ice for surgery. A craniotomy was performed and
a micropipette was introduced into the tectum. In six ani-
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
321
A 6hrrl;iations
cEnt
CG
dLFB
ICO
i-IT
Imc
Imlf
Imr
IP
IPd
IPV
Mes V
MFB
Caudal entupeduncular nucleus
Central gray
Dorsal peduncle of the lateral forebrain bundle
Intercollicular nucleus
Ipsilateral isthmotectal tract (from Imc)
Caudal magnocellular nucleus isthmi
Interstitial nucleus of t h e medial longitudinal fasciculus
Rostra1 magnocellular nucleus isthmi
Parvocellular nucleus isthmi
Dorsal interpeduncular nucleus
Ventral interpeduncular nucleus
Mesencephalic trigeminal nucleus
Medial forebrain bundle
Medial longitudinal fasciculus
MLF
Nucleus of the medial longitudinal fasciculus
Nmlf
Trochlear nerve
N. N
Optic tract
OT
Pa
Paraventricular hypothalamic nucleus
Posterior commissure
PC
PMc
Profundus mesencephali caudalis
Profundus mesencephali rostralis
PMr
Profundus mesencephali ventralis
PMv
Prerubral area
PR
Nucleus reuniens
He
Red nucleus
RN
RSUdm) Reticularis superioris lateralis, dorsomedial segment
WIXvl) Reticularis superioris lateralis, ventrolateral segment
Ipsilateral
0I
/
\
Tect-Imc
1
Rostra1
in tectum
Imc
in tectum
neuropile
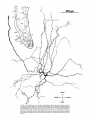
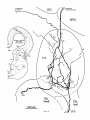
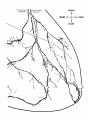
Fig. 2. Schematic diagram of the organization of caudal magnocellular
nucleus isthmi Umc). The nucleus consists of'a thick, tilted cell plate faced
by rostral and caudal neuropilc laycrs into which the dendrites of Imc cells
extend. Spatially restricted rodlike tectoisthmic afferents CI'ect-Imcl penetrate the nucleus perpendicular to the cell plate. The reciprocal point-topoint mapping between the tectum and Imc is arranged so that ventral in
the cell plate corresponds to rostral in the tectum while lateral in the cell
plate equals lateral in the tectum.
Ksl
Lateral superior raphe nucleus
Reticularis superioris medius
Medial superior raphe nucleus
SAC
Stratum album centrale
Scd
Smallcelled nucleus, dorsal segment
scv
Small-celled nucleus, ventral segment
SFGS
Stratum fibrosum e t griseum superficiale (superficial gray)
SGC
Stratum griseum centrale (central gray)
SGP
Stratum p i s e u m pcriventriculare (periventricular gray)
SN
Substantia nigra
SO
Stratum opticum
SP
Suprapeduncular nucleus
TBd(lg) Dorsal tectobulbar pathway, large-caliber component
TBdtsm) Dorsal tcctobulbar pathway, small-caliber component
TBi
Intermediate tectobulbar Dathwav
TBv(rned) Ventral tectobulbar pathway, medium-caliber component
TBvkn) Ventral tectobulbar pathway, small-caliber component
Tmt-Imc Tecto-isthmic tract (to Imc)
Torc
Torus semicircularis, central nucleus
Tor1
Torus semicircularis, laminar nucleus
'lTh
Tectothalamic tract
vLFB
Ventral peduncle of the lateral forebrain bundle
VTA
Ventral tegmental area
x-IT
Crossed isthmotectal tract (from Ip)
111
Oculomotor nucleus
V mr
Mesencephalic root of the trigeminal nerve
KSM
Rsm
L
~~
_I
mals, multiple iontophoretic injections (2-3 pA for 20 minutes at each site) of concentrated Sigma type VI HRP in pH
8.6 Tris buffer were made a t 2 to 4 sites in one tectal
hemisphere. Thirteen other animals received small iontophoretic injections (1 pA pulsed for 20-100 seconds with a
5-30 pm I.D. tip) at a single site or at two widely separated
sites. Six animals received large pressure injections (0.5 pl)
in the medial or lateral half of the pontine tegmentum.
Animals survived for 3 days at 20°C before intracardial
perfusion with phosphate-buffered saline followed by a buffered solution containing 15%paraformaldehyde and 3% glutaraldehyde. Gelatin-imbedded brains were soaked in 30%
sucrose and sectioned the next day on a freezing microtome
at 110 or 120 pm. Transverse or horizontal serial sections
were processed as described in Adams ('77) and counterstained with cresyl violet.
Low-power reconstructions of injection sites, labeled somata, and axon terminals were made from serial sections
with a drawing tube. A stereogram of labeled somata was
made by hand (Glenn and Burke, '81; Sereno, '85) for one
of the horizontally sectioned double injection cases. It can
be viewed by ocular divergence or by using a standard
stereo viewer (note: fusion attained by crossed-eye viewing
will result in inverse depth). In cases with small injections,
single HRP-filled axonal and dendritic arborizations were
reconstructed with a drawing tube from a number of adjacent sections under a 1 0 0 ~oil objective to illustrate their
detailed morphology (see Sereno, '85, for details). Nomenclature of cell groups and fiber tracts is the same as that
used in Sereno ('85).
RESULTS
Cytoarchitecture of the isthmic complex
The isthmic complex in turtles contains three cytoarchitectonically distinct nuclei-caudal and rostral magnocellular nucleus isthmi, and parvocellular nucleus isthmi. The
caudal magnocellular nucleus isthmi (Imc) is a prominent
nucleus at the caudal border of the midbrain with a reniform profile in transverse sections (Fig. lA,B). It consists of
a thick plate of cells oriented approximately perpendicular
M.I. SERENO AND P.S. ULINSKI
322
and ending about midway through the tectum (Fig. 3A). In
transverse sections, Imr appears as a loose grouping of
neurons with robust, elongated somata (20-30 pm wide,
30-40 Fm long) that is completely surrounded by a cell-free
neuropile layer (Fig. 13A). The medium-caliber component
of the ventral tectobulbar pathway (TBv[med])and the ipsilateral Imc-tectal pathway (i-IT)pass just medial to Imr
while the small-caliber component of the ventral tectobulbar pathway (TBv[sm])and the tectoisthmic pathway (TectImc) pass through it (Fig. 13A, B; see also Sereno, ’85).
Imrhas been given other names since it was identified by
Cruce and Nieuwenhuys (“74).Foster and Hall (‘75), ten
Donkelaar and Nieuwenhuys (‘79), and Wang et al. (‘83),
for example, labeled it “nucleus profundus mesencephali,”
while Brauth et al. (’83) and Kiinzle and Schnyder (‘84)
label Imr together with Imc as a unitary “nucleus isthmi
magnocellularis.”
The parvocellular nucleus isthmi (Ip) lies at the medial
edge of the neuropile-somata-neuropile sandwich composing Imc. Because of the tilt, Ip is placed a bit rostral to Imc
(Fig. 1A; insets in Figs. 8-10). It consists of a close-packed
group of smaller elongated somata (5-8 pm wide, 8-12 pm
long) that are most densely packed near the border with
Imc. Kiinzle and Schnyder (‘84) showed that Ip projects to
the contralateral tectal hemisphere. The following sections
concentrate on the magnocellular nuclei.
Imm
B
Caudal magnocellular nucleus isthmi (Imc)
1
c*
Figure 1B is a transverse view of Imc after a large tectal
injection. Such injections resulted in Golgi-like filling of
neurons with somata, terminal arbors, or just fibers of
passage a t the injection site. The dark band in Imc consists
of anterogradely labeled fine-caliber tectoisthmic terminals
superimposed on retrogradely labeled Imc somata and dendrites. The location of the dense band depended on the locus
of the tectal injection. In addition, a lighter meshwork of
large-caliber axons and terminal boutons homogeneously
fills the entire nucleus. This background labeling of Imc
appeared in other cases regardless of what tectal locus was
iniected. A few labeled cells and terminals were occasionally seen in Ip after large tectal injections.
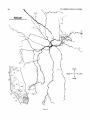
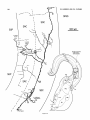
Figure 3A shows a horizontal reconstruction of a typical
small tectal HRP injection that labeled cells in Imc, Imr,
and other nuclei not shown, and several different types of
terminals in the tectum. One terminal type was morphologically very distinct and always appeared rostromedial t o an
injection site (e.g., in Fig. 3A, asterisks represent all such
terminals found in this case). Each terminal arose from a
robust parent axon (3 pm diameter) coursing through the
stratum griseum centrale and consisted of a dense, localized, cylindrical thicket of several thousand boutons spanning the stratum grieseum centrale (SGC), the stratum
fibrosum et griseum supeficiale (SFGS), and the stratum
opticum (SO).
These terminals were identified as terminals of ipsilatera1 Imc neurons by several pieces of evidence. First, they
.-;ID
&n
Imc
Imr
Fie.
- 3. A Horizontal reconstruction of a tectal injection. The tectum. the
caudal magnocellular nucleus isthmi (Irnc), and the rostral magnocellular
nucleus isthmi (Imrj were reconstructed from serial transverse sections.
This is a stereotaxic view with no correction for the curvature of the tectum.
A small HRP injection retrogradely filled 14 cells in a small region of Imc
and nine cells scattered throughout Imr. B: Imc and Imr redrawn at the
same scale to show the location of the labeled cells. In addition, the injection
anterogradely labeled a total of six dense, localized terminal arbors in the
tectum (asterisks in A-drawn to scale) rostromedial t o the injection site.
Their parent axons were about 3 pm in diameter and coursed through the
SGC from the injection site without branching. These terminals most likely
arise from Imc neurons (see text). Two of the arbors (at X and Y) are
reconstructed a t high magnification in Figures 5 and 6.
to the rostrocaudal axis of the brainstem. The dorsal and
lateral edges of the plate tilt caudally (Figs. 2, 7A, ll),so
that a single section containing all parts of the cell plate is
difficult to obtain in either the horizontal or the transverse
planes. Neuropile layers adjoin the rostral and caudal faces
of the cell plate (Fig. 2). The tilt just noted makes both the
caudal and rostral neuropile layers visible in some transverse sections (Fig. 18; dotted-lines in Fig. 11). The cell
plate contains compact~y arranged, medium-sized, elongated somata (9-15 pm wide, 18-25 pm long).
The rostral magnocellular nucleus isthmi (Cruce and
Nieuwenhuys, ’74)is a rostrocaudally oriented tube of large
cells that borders directly on the lateral edge of the superficial layers Of the tectum. It is 13000-19500
pm long, beginning near the caudal face of the tectum just dorsal to Imc
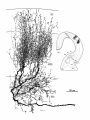
Fig. 4. Transverse view of caudal magnocellular nucleus isthmi (Imc)
cell. This neuron was labeled by a small ipsilateral tectal injection and then
reconstructed from transverse sections. Its medium-caliber (3 pmj myelinated axon arises from the thickest primary dendrite (at open arrow) and
passes rostrally into the isthmotectal tract without emitting local collaterals. Dendrites are covered with irregular spicules and arborize mostly in a
plane almost perpendicular to the viewer. A perspective box has been drawn
around the arborization (see also inset) to make the resultant foreshortening
more apparent: the dendritic field is actually a little longer rostrocaudally
than mediolaterally.
I
SGC
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
are unlikely to be retinal terminals (e.g., Fig 7E), which
avoid the SGC and have parent axons in the stratum opticum. Second, they are not contralateral isthomotectal terminals, which arise from thin (1pm diameter) axons that
cross in the supraoptic decussation, approach the tectum
from the front, and then turn caudomedially to run over
the tectal surface in the stratum opticum (Fig. 12A,B;insets
in Figs. 8, 16). Third, these terminals do not arise from
neurons labeled in Imr, nucleus lentiformis mesencephali,
or profundus mesencephali rostralis (PMr). The axons of
neurons in those nuclei were reconstructed from serial sections and found to branch widely, sparsely innervating large
areas of the tectum. Fourth, the dense thickets are not
terminals of thalamic neurons or neurons in the dorsal
nucleus of the posterior commissure (dNPC) since parent
axons of these terminals enter the tectum via the tectothalamic tract ( n h ) . Finally, the parent trunks of these terminals match the diameter and laminar position of the
axons arising from Imc neurons. Kunzle and Schnyder (‘84)
injected Imc with HRP (tetramethyl benzidine development) or a radioactive tracer. In both instances, the laminar
distribution of mass label precisely matched that of the
dense terminals in the present material. Thus, tectal injections permitted analyses of both the dendritic and axonal
arbors of Imc neurons.
Large control injections of HRP into the medial or lateral
half of the pontine tegmentum left Imc as a label-freeisland
surrounded by densely labeled reticular structures. Notably, Ip in all these cases was filled with fine-caliber terminals that were particularly dense in the medial third of Ip
along its border with Imc; this label was much denser than
the terminal label seen in Ip after large, equivalently distant tectal injections.
Dendritic morphology of Irnc neurons. Sixteen Imc cells
labeled by small tectal injections were completely reconstructed from serial sections. Many more were examined
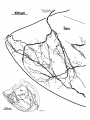
locally or partially reconstructed. Figure 4 is a drawing of
an Imc neuron made from transverse sections while Figures 8-10 are drawings of horizontally sectioned Imc neurons located at dorsal, ventral, and intermediate levels in
the nucleus. Figure 7C is a photomicrograph of labeled Imc
cells.
Four to six primary dendrites radiate from the soma in
all directions in the horizontal plane. Secondary and tertiary dendrites turn rostrally or caudally, generating a
flattened, elongated dendritic field that extends 500-600
pm rostrocaudally, 150-300 pm mediolaterally, and 75-150
pm dorsoventrally. Consequently, Imc dendritic fields are
greatly foreshortened in transverse view (Fig. 4) and are
seen almost en face in horizontal reconstructions (Figs. 7C,
8-10). Viewed on end, a single dendritic field spans roughly
1/50 of the area of the cell plate. One of the primary dendrites is typically more robust than the others (stippled
dendrite in Figs. 4, 8, 9). Secondary and tertiary dendrites
bear short, filamentous appendages with en passant varicosities that grow more numerous in the rostra1 and caudal
Fig. 5. Axon terminal arbor in the tectum originating from a caudal
magnocellular nucleus isthmi (Imc) cell. This terminal was labeled by a
small tectal injection (see inset) and then reconstructed from several sections. It corresponds to arbor X in Figure 3. The 3 pm diameter myelinated
parent trunk emitted no other collaterals before turning upward to give off
a radially oriented spray of about 2,700 boutons that were most densely
packed in the upper two-thirds of the retinal-recipient SFGS. The boutons
in the SGC were somewhat larger (about 2.5 pm diameter) than those in
the SFGS (about 1.5 pm diameter).
325
neuropile layers. The great majority of the varicosities are
smaller, more transparent, and have less-regular shapes
than putative synaptic boutons arising from HRP-filled
axon collaterals in the same material; however, the ends of
some dendrites occasionally bear larger, smoother, darkerstaining varicosities that are indistinguishable from synaptic boutons (e.g., swellings near asterisk at upper right
corner of Fig. 9).
A medium-caliber axon (2.5-3.5 pm) originates usually
from the thick primary dendrite about 50 pm from the
soma. After an initial constricted segment that is 20-40 wm
long, the axons stain less darkly, except at short, periodically appearing constrictions, suggesting myelination. Local collaterals were never observed in Imc. The axons gather
into a loose bundle-the ipsilateral isthmotectal tract (iIT)-and exit the dorsomedial corner of the nucleus. The iIT fans out as it approaches the caudolateral face of the
tectum, appearing Y-shaped in horizontal sections (inset in
Fig. 16). It passes just medial to Imr (Fig. 13B) and enters
the tectum to run in the upper layers of the SGC. Over 60
Imc axons were traced through serial sections until they
entered a tectal injection site. In not one instance was an
axon collateral observed in the tectum.
Axon terminal arbors of Irnc neurons. The terminal arbors of Imc neurons each consist of a conspicuous, dense
vertical array of several thousand boutons and are similar
at all tectal loci. The myelinated parent axons of these
terminals were never observed to emit collaterals before
the main trunk turned upward to form the thicketlike
arbor. The first vertical branches are myelinated for 100200 pm. The arbors occupy cylinders approximately 150 pm
in diameter and 400 pm tall. A single arbor thus covers
about 1/200 of the surface area of one tectal lobe. An average of about 3,000 boutons are packed into each cylinder.
The terminal in Figure 5, for example, contained a little
more than 2,700 boutons, while the one in Figure 6 contained about 3,600 boutons. There is a characteristic sublaminar pattern of bouton distribution and size. About 10%
of the boutons are located in the SO, about 60% are in the
upper two-thirds and 5% in the lower third of the SFGS,
and about 25% are in the upper half of the SGC. The
boutons in the SGC tend to be larger than those in the
SFGS (about 2.5 pm vs. 1.5 pm diameter). Bouton size is
quite variable in the upper half of the SO, where a few very
large boutons (up to 7 pm in diameter) are mixed in with
medium-sized ones. There was little variation in bouton
density perpendicular to the main radial axis of an arbor.
(The appearance of there being two columns of boutons in
the SFGS in the terminal of Figure 6 is the result of a
vertical dent in the arbor made by a blood vessel.)
Topography of the Imc-tectal projection. The topographic organization of the projection from Imc to the tectum was assayed at the single-cell level by comparing the
dendritic field locations of neurons labeled by two disjunct
injections in the same animal. Across-case comparisons
based on single injections in different animals were also
made. Figure 7A and B show a horizontal reconstruction of
a case with two small tectal injections. Photomicrographs
of labeled Imc cells and sections through each injection site
are shown in Figure 7C-E. Individual axons of labeled Imc
neurons were traced through serial sections to determine
which injection site they entered. The labeled neurons form
two nonoverlapping clusters in the tilted cell plate of Imc.
Most of the neurons are located in the lateral third of the
cell plate with the dark-outline cluster (injection11)situated
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
dorsal to (i.e., closer to the viewer than) the less-compact
thin-outline cluster (injection I). Several densely filled neurons were reconstructed from serial sections to determine
the morphology and location of their dendritic fields. Three
of these-labeled 2, 3, and 1 in Figure 7A-are illustrated
at high magnification in Figures 8-10. Neuron 2 (Fig. 8)
was situated at the edge of the main injection I1 cluster
while neuron 3 (Fig. 9) was at the edge of the main injection
I cluster (it was not possible to unambiguously reconstruct
neurons in the very center of each cluster). The flattened,
elongated dendritic fields of these two neurons are nonoverlapping, though each overlaps with the dendritic fields of
many of the neurons in the cluster to which it belongs. An
analysis of this and other double injection cases is consistent with a scheme in which rostral in the tectum corresponds to ventral in the Imc cell plate, while lateral in the
tectum corresponds to lateral in the cell plate (Fig. 2).
The Imc neurons labeled by injection I are less compactly
arranged than the tight cluster of injection I1 neurons. One
of the outlying injection I neurons-neuron 1in Figure 7A
(cell marked with asterisk in Fig. 7C)-was serially reconstructed (Fig. 10).Its dendritic field not only avoids those of
injection I1 neurons but also those of most of its fellow
injection I neurons. Two features of injection I are that it
invaded the SGC (Fig. 7D) and that it anterogradely labeled
ten “ectopic” Imc terminals (asterisks in Fig. 7B and large
arrow in Fig. 7D). Injection 11, by contrast, was mainly
restricted to the SFGS (Fig. 7E) and labeled only one “ectopic” Imc terminal. Thus, it seems likely that some of the
ventromedial scatter in the injection I cluster is due to HRP
uptake by Imc axons that terminate rostromedial to the
injection site.
Comparisons of the loci of labeled Imc cells among cases
with single injections confirmed the proposed topography.
Figure 11shows three transversely sectioned cases. Taking
into consideration the tilt of the cell plate, increasingly
rostral injection sites (B then A then C) result in successively more ventral main clusters while increasingly lateral injection sites (A then B then C) result in successively
more lateral clusters. The ventromedial scatter sometimes
observed (e.g., in B) is consistent with a fibers-of-passage
interpretation. In summary, the Imc-tectal projection appears to be a topological, point-to-point mapping in which
the dendritic and axon terminal fields of single Imc neurons
occupy less than a few percent of the surface of the source
and target maps.
327
trast to the restricted topographic band of labeled cells and
terminals observed in Imc, a dense mat of labeled cells and
terminals is spread throughout Imr. A similar pattern was
observed with injections occupying less than 1%of the
tectal surface. The nine Imr cells labeled by the small
injection illustrated in Figure 3, for example, were evenly
scattered throughout the entire extent of the nucleus. Since
scatter occurred no matter what tectal locus was injected,
each small patch of the tectum must contain axons from
neurons in many parts of Imr.
The dense mat of axons in Imr contained three components: fine-caliber fibers passing through without branching, many fine-caliber preterminal branches bearing smalldiameter boutons that often contacted Imr cells (Fig. 12C),
and large-caliber preterminal branches bearing robust boutons (Fig. 12D,E). Single neuron reconstructions showed
that the fine-caliber fibers consist partly of TBv(sm) and
Tect-Imc axons (Sereno, ’85) while the large boutons arise
from the local collaterals of Imr axons (see below). The
presence of dense, fine-caliberterminal degeneration in Imr
after tectal lesions (Sereno, ’85; Foster and Hall, ’75) and
the labeling of only fine fibers after large tegmental injections suggest that the small boutons represent a tectal
input to Imr and not a retrogradely labeled branched input
to Imr and the tectum originating from somewhere else. By
contrast, the nontopographic background label in Imc described initially (Fig. 1B) turned out to be a retrogradely
labeled branched input to Imc and the tectum originating
from Imr.
Dendritic and local axonal morphology of Irnr neuron.~. Thirteen Imr cells labeled by small tectal injections
were completely reconstructed from serial sections. A much
larger number were examined in single sections or partially reconstructed. Figure 14 is a reconstruction of an Imr
neuron and its local collaterals made from transverse sections, while Figures 16 and 18 are reconstructions of Imr
neurons made from horizontal sections. Imr neurons have
robust rostral and caudal primary dendrites that arise from
an elongated soma and branch only once or twice, giving
rise to a large, sparse dendritic field 750-1,000 pm long,
200-300 pm wide, and 200-300 pm deep that approaches
the size of the entire nucleus (note the lower magnification
of the Imr reconstructions compared to those of Imc cells).
Distal dendrites often extend medially through the Imr
neuropile to reach profundus mesencephali caudalis. All
secondary and tertiary dendrites were covered with short,
uniform hairlike spicules quite unlike the longer, irregular
Rostra1 magnocellular nucleus isthmi (Imr)
appendages seen on distal Imc neuron dendrites.
Figure 13B is a transverse view of Imr from the same
A robust axon (4-6 p m in diameter) originates most often
large tectal injection case illustrated in Figure 1B. In con- from the rostral primary dendrite a short distance from the
soma. The initial branching of the axon appears random
(compare Figs. 14, 16, 18),but a pattern emerges when the
branches are followed through serial sections. Each Imr
neuron gives rise to one or more myelinated trunks to the
tectum and one or more myelinated trunks to Imc, which
will be described in turn. In addition, a sparse field of local
axon collaterals bearing large boutons usually appeared
within Imr (e.g., Figs. 14, 18).
Fig. 6. Axon terminal arbor in the tectum originating from caudal magBranches of Irnr axom in the tectum. Figure 15 is a
nocellular nucleus isthmi (Imc) cell. This terminal was also labeled by a
tectal injection (see inset) and recovered from several sections. It corre- reconstruction of part of one of the branches that reaches
sponds t o arbor Y in Figure 3. The 3,600 boutons in this arbor appear to the tectum from the Imr neuron illustrated in Figure 14.
form two columns, especially in the SFGS. This is the result of a large The axon leaves Imr to enter the SGC (see inset to Fig. 15)
vertical blood vessel taking a chunk out of a bouton distribution that is
about one-third of the way forward from the caudal face of
otherwise radially homogeneous when viewed from the tectal surface. All
nodes on the 3 pm diameter parent trunk situated outside the dense arbor- the tectal hemisphere. Preterminal branches (open arrows)
ization (for example, node a t paired triangles) were unbranched.
are emitted at intervals of about 200 pm. (By contrast, an
A
Rostra1
Medial +*LOh?*l
Caudal
a- to tech1injection site I
[$ - to tecta~injection site n
500 urn
Tectal
Injections
Horizontal
Reconstruction
Fig. 7. Analysis of a case with two tectal injections. A Stereoscopic view
of retrogradely labeled cells i n caudal magnocellular isthmi (Imc). Injection
I1 cells (bold outlines) form a tight clump situated dorsal (i.e., near the
viewer) to the more diffuse injection I cell cluster (thin outlines). The
dendritic arbors of the cells numbered 2, 3, and 1 are reconstructed at
higher magnification in Figures 8-10,The tilt of the cell plate (boundaries
indicated by dashed lines) is especially obvious when the figure is viewed in
stereo hy ocular divergence or by using a standard stereo viewer (fusion
obtained by cross-eyed viewing will result in inverse depth). B: Horizontal
reconstruction of the tectal injections and the corresponding labeled clusters
of Imc cells. Injection I (Iight stipple and photomicrograph in D) antero-
gradely labeled many more Imc axon arbors (asterisks) than did injection I1
(dark stipple and photomicrograph in El, probably because injection I invaded the SGC, where the myelinated parent trunks of these arbors run. C:
Photomicrograph of labeled Imc cells in the injection I clump. The cell with
a n asterisk is cell number 1 in A and is reconstructed in Figure 10. The
arrows indicate axon origins. D: Injection site I. An anterogradely labeled
Imc terminal (large arrow) is visible rostra1 to the injection. The retinalrecipient SFGS and SO, however, are unlabeled rostrally (small arrows). E:
Injection site 11. The retinal terminal layers are heavily labeled around this
injection (small arrows).
100 urn
Fig. 8. High-magnification view of caudal magnocellular nucleus isthmi (Imc) neuron number 2
from Figure 7A. This neuron was labeled by tectal injection I1 (Fig. 7B,E) and reconstructed from
horizontal sections, resulting in an en face view of the flattened, rostrocaudally elongated dendritic
field. The dendrites bear irregular spicules that grow more numerous on the secondary and tertiary
branches that penetrate the rostra1 and caudal neuropile layers. The 3 pm diameter axon (origin at
open arrow; unbranched nodes at paired triangles) arose from the thickest primary dendrite (stippled)
and emitted no collaterals until it entered the tectal injection site. The inset shows the location of the
neuron.
330
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
entire Imc terminal arbor is only about 150 pm wide.) These
branches course toward the ventricular surface, giving rise
to radially oriented strings of 5 to 30 large boutons situated
in the lower half of the SGC and in the stratum album
centrale (SAC). The main trunk continued to branch until
it was lost without any sign of thinning at the injection
site. Thicker, apparently myelinated collaterals are given
off at some branch points (one at A and two at C). They are
almost as thick as the main trunk and course rostrally
through the SGC and SAC (3-D arrows) giving off radially
oriented strings of boutons into the SGC and SAC at regular intervals. The collateral at A innervated the middle and
near rostral tectum while the collaterals at C innervated
the rostral tectum, one reaching the extreme rostromedial
edge of the tectum. The second branch to the tectum (at
upper left in Fig. 14) was similar in morphology to the
trunk just described, except that it distributed strings of
boutons to the caudal face of the tectum. It was lost near
the caudomedial edge of the tectal hemisphere, at which
point it had thinned considerably. The tectal branches of
other Imr neurons were similar in overall form to the one
illustrated in Figures 14 and 15 though sometimes they
were a bit less fastidious in covering all parts of the tectum.
Branches of Irnr axom in Imc. The other target of Imr
axons besides the tectum was, unexpectedly, Imc. Figure 17
is a horizontal reconstruction of a single axon terminal
arbor that arose from the Imr neuron shown in Figure 16
and terminated in Imc. Typically, Imr neurons give off two
or three thick myelinated trunks to Imc that enter either
the front face or the medial edge of the cell plate, often a t
more than one dorsoventral level (for example, neuron in
Fig. 18).Once in Imc, each trunk splits into several myelinated branches that wander back and forth, giving off strings
of very large (2-3.5 pm diameter) en passant boutons that
stand out amongst the much smaller (1pm diameter) boutons found in the terminal arbors of tectoisthmic axons.
Terminal boutons of Imr axons are often capped with a tiny
varicosity (Fig. 17). A single Imr axon thus sparsely distributes 1,000-2,500 boutons to many mediolateral and dorsoventral levels in the cell plate and the caudal neuropile of
Imc. The arbor in Figure 17 contained about 1,840 boutons.
The terminal field is rigorously confined to Imc; when a
branch comes to the edge of the nucleus, it rarely continues
for more than a few microns outside of it before reentering (for example, branch at extreme lower right of arbor in
Fig. 17).
The nontopographic nature of a single Imr axon in Imc is
brought out by comparing its spatial distribution to that of
the restricted clusters of retrogradely labeled Imc cells and
anterogradely labeled tecto-isthmic terminals produced by
the same injection. In Figure 17, for example, small clusters
Fig. 9. High-magnification view of caudal magnocellular nucleus isthmi
(Imc) neuron number 3 from Figure 7A. This neuron was labeled by tectal
injection I (Fig. 7B,D). Its flattened, elongated dendritic field volume is
entirely ventral to that of the injection I1 cell illustrated in Figure 8. The 3
pm diameter myelinated axon (origin a t open arrow) arises from the thickest
primary dendrite (stippled) and emits no local collaterals. Most dendrites
bear filamentous spicules; the tip of one dendrite (asterisk), however, gives
rise to varicosities morphologically indistinguishable from synaptic boutons
as it ends within the ventral tectobulbar tract (TBv-see inset).
331
of Imc cells and Tect-Imc terminals overlapped in the lateral third of Imc (not illustrated). The Imr axon, by contrast, not only distributed boutons in and around these
clusters, but actually somewhat more densely innervated
the topographically inappropriate medial half of Imc. Other
reconstructions of Imr axons in Imc revealed equally nontopographic bouton distributions.
The uniform background labeling of Imc seen after large
tectal injections (Fig. 1B) thus represents a number of Imr
axon arbors filled through the tectal branches of their bifurcating axons. Figure 19 is a high-magnification photomicrograph of the upper left-hand corner of Imc from the
transverse section shown in Figure 1B. The background
label consists of a meshwork of robust, myelinated branches
bearing very large boutons that are precisely confined to
Imc, clearly marking its boundaries. The morphology and
organization of the background terminals labeled by large
tectal injections closely matches that of single identified
Imr axon arbors in bouton size, lack of topography, and
complete restriction to Imc. The lack of nontopographic
large-caliber terminal degeneration in Imc after tectal lesions (Sereno, '85) is consistent with this conclusion.
DISCUSSION
Neurons in the tectal-receiving rostal magnocellular nucleus isthmi (Irnr) of the turtle give rise to a strikingly
nontopographic output that is superimposed on the topographically organized circuit between the ipsilateral tecturn and the caudal magnocellular nucleus isthmi (Irnc),
not only in the tectum but also in Imc itself. Figure 20
schematically illustrates the morphology of single Imc and
Imr neurons. In this section, the organization of Imc and
Imr is summarized and compared with findings on similar
nuclei in other vertebrates. Some functional implications of
these two ipsilateral tecto-isthmo-tectal circuits are then
considered.
Anatomy of isthmotectal neurons
The caudal topographic nucleus isthmi. A component of
the nucleus isthmi complex that receives a tectal input and
projects back to the ipsilateral tectum appears to be present
in all vertebrate classes. This nucleus always has a topographic connection with the ipsilateral tectum and often
consists of a platelike array of neurons with elongated
dendritic fields that project both to the superficial gray
layers and to a directly underlying non-retinal-recipient
layer in the central gray. In each instance, however, there
are differences in detail. In turtles, Imc somata are arranged into a thick cell plate faced with rostra1 and caudal
neuropile layers. The neurons have flattened bipolar dendritic fields covering a few percent of the cell plate area as
well as spatially restricted axon arbors covering less than
1% of the ipsilateral tectal hemisphere. The swarm of several thousand boutons typically given off by a single Imc
axon occupies not only the superficial retinal-recipient laminae (SO and SFGS) but also part of the underlying central
gray (upper half of the SGC). Imc neurons appear to be
cholinergic (Desan et al., '84).
In bony fish (Sakamoto et al., '81; Ito et al., '82), nucleus
isthmi contains a sharply bounded cell plate (shell) but then
only a single, caudal neuropile layer (core) into which the
unipolar dendritic arbors of the cell plate neurons extend.
Their axon terminals in the tectum innervate the superficial gray and a non-retinal-recipient layer at the top of the
central gray. However, many fish species have one or two
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
333
B
-
-
01
Lateral
Lateml
Rc
-
C
a1
Lateral
Q
Q
Q
Q
Q
1 mm
._--._
Q
Tectum
(horizontal
reconstruction)
Ipsilateral Imc. (transverse)
1 mm
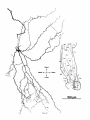
Fig. 11. Labeled caudal magnocellular nucleus isthmi (Imc) neurons in three transversely sectioned
cases each with a single tectal injection. Taking account of the tilt of the Imc cell plate (dorsal and
lateral edges caudal to its ventral and medial edges), increasingly rostral tectal injections (B then A
then C) are seen to result in more and more ventral clusters in the cell plate, while increasingly
lateral injections (A then B then C) give more and more lateral clusters. The uentromedial scatter of
labeled Imc cells in case B is consistent with a fibers-of-passage interpretation using that scheme;
some of the Imc axon trunks, which stream rostromedially through the central gray of the tectum,
must have been filled a t the injection site before they terminated. The dashed lines in Imc indicate
the boundaries of the cell plate with rostral and neuropile layers. In case C, the dendritic arbors of the
labeled cells were also drawn. No correction was made for the curvature of the tectum in the horizontal
reconstructions of the injection sites.
additional even deeper layers of retinal terminals in the
middle and lower parts of the central gray (Vanegas et al.,
’84). Some of the contacts made by isthmotectal terminals
Fig. 10. High-magnification view of “ectopic” caudal magnocellular nucleus isthmi (Imc) neuron number 1 from Figure 7A. This neuron was
labeled by tectal injection (Fig. 7B,D). Unlike many other injection I neurons whose dendritic field volumes overlap each other ventrally and laterally in the nucleus (see, for example, neuron 3 illustrated in Fig. 91, this
neuron’s dendritic field occupies the most medial part of the nucleus (Fig.
7C), overlapping few other labeled dendritic fields to a significant degree. It
was probably injected as a fiber of passage whose terminal arborization lies
rostral and medial to the injection site (possibly one of the ten “ectopic”
arbors shown beyond injection I in Fig. 7B,D). A 3 pm diameter myelinated
axon arises (open arrow) from the base of a dendrite and makes a semicircular detour around a blood vessel soon after becoming myelinated. The
inset shows the location of the neuron.
may be axoaxonic synapses on retinal terminals (Henley et
al., ’86). A contralateral isthomotectal projection is apparently lacking.
In frogs, a loosely packed cell plate and its associated
neuropile layers (medulla) are almost completely enclosed
by a unique, tightly-packed sheet of cells (cortex)resulting
in a mediolaterally oriented tacolike structure (Gruberg
and Udin, ’78; Grobstein and Comer, ’83).Part of the medulla and part of the anterior limb of the cortex project to
the ipsilateral tectum. Ipsilateral isthmotectal s o n s terminate throughout the superficial retinal-recipient zone
(layers A-F), but also just below it (layer 8) (Gruberg and
Udin, ’78; Gruberg and Lettvin, ’80). As with bony fish,
some frog species (for example, Rana pippiens)have an even
M.I. SERENO AND P.S. ULINSKI
334
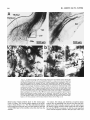
Fig. 12. A Photomicrograph of the lateral border of the tectum in transverse section showing the
crossed isthmotectal tract (x-IT). I t was retrogradely labeled by a large tectal injection (not shown)
that also heavily labeled other tectal afferents (for example, from profundus mesencephali-PMc) and
tectal efferent pathways (for example, the large- and small-caliber dorsal tectobulbar pathwaysTBd[lg] and TBd[sml. The x-IT arises from parvocellular nucleus isthmi (Ip in Fig. lA), passes rostrally
to cross in the supraoptic decussation, and then caudally to approach the rostrolateral edge of the
contralateral tectum. When a fascicle of x-IT fibers reaches the appropriate rostrocaudal point along
the edge of the tectum, it turns upward abruptly (shown here) to run over the tectal surface in the
stratum opticum (SO) until it reaches the mediolateral locus where it terminates. B: High-magnification view of x-IT (boxed region in A) showing fascicles of fine-caliber axons (about 1pm diameter). C:
High-magnification view of Imr (see Fig. 13B for low-power view) showing putative contacts (arrows)
between small-diameter synaptic boutons and a n unlabeled, NissI-stained Imr cell soma. These
boutons were labeled after a large tectal injection and probably represent a tectal input to Imr (see
text). D,E: Smaller numbers of very-large-caliber boutons (arrows) also appeared in Imr (same magnification as C). These probably arise from local collaterals of Imr cells (see Figs. 14,16).
deeper lying retinal terminal layer in the central gray
(layer G) (Lazar, '84). Nucleus isthmi appears t o be cholinergic in frogs (Desan et al., '84). The contralateral isthmotectal projection arises not only from the posterior limb and
ventral folded part of the tacolike cortex but also from the
medulla.
In snakes, the somata and dendrites of nucleus isthmi
neurons are not organized into obvious cell plate and neuropile layers. More notable, however, are the small terminal arbors of these neurons, which bypass not only the SGC
but also the SFGS t o terminate in only the most superfical
of the retinal-recipient laminae-the SO-and in the over-
ORGANIZATION OF TURTLE ISTHMTC NUCLEI
335
Fig. 13. A Transverse section through the rostra1 magnocellular nucleus isthmi (Imr). The large
somata in Irnr form an oval cluster that is completely surrounded by a neuropile layer. B. Samemagnification view of Imr after a large but subtotal HRP injection into the ipsilateral tectum (same
case a s in Fig. 1B). The dark blob that completely engulfs Imr in this and most other sections through
the nucleus consists of a mass of fine-caliber terminals probably from the tectum (see text and Fig.
12C)superimposed on a cluster of labeled Imr cells, their dendrites, and the robust local collaterals
arising from their large-caliber axons. In addition, the small-caliber component of the ventral tectobulbar pathway, TBv(sm),passes directly through Imr without branching (Sereno, '85). The ipsilateral
isthmotectal tract &IT) and the medium-caliber component of the tectobulbar pathway, TBv(med),
pass Imr medially.
lying stratum zonale (Dacey and Ulinski, '86b). In this
respect, snakes appear to differ from all other vertebrates
(as they do in having the dendrites of their principal deeplayer tectobulbar cell types end before entering any of the
retinal-recipient laminae-Dacey and Ulinski, '86a). Contralaterally projecting isthmotectal neurons are intermixed
with ipsilateralIy projecting ones.
In birds, the nucleus similar t o turtle Imc is the parvicellular nucleus isthmi, Ipc. It lacks differentiated neuropile
layers but otherwise conforms to the general vertebrate
pattern in that cells with elongated dendritic fields innervate the superficial retinal-recipient layers 2-5 (=ITb-d) as
well as directly underlying non-retinal-recipient layers 10
and 11(=IIi) (Cajal, 1899; Hunt et al., '77). The dense nest
100 urn
Figure 14
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
of boutons in each Ipc terminal is confined to a narrower
cylinder than turtles. Hunt et al. (’77) showed that Ipc
terminal boutons contain round vesicles and apparently
synapse only on dendrites.
In several marsupial and placental mammals (oppossum:
Mendez-Otero et al., ’80; tree shrew: Harting et al., ’73; cat:
Graybiel, ’78; Sherk, ’79; monkey: Harting et al. ’80), the
parabigeminal nucleus has a reciprocal topographic connection with the ipsilateral superior colliculus. The main difference between mammals and nonmammals is that the
ipsilateral parabigeminotectal projection avoids terminating in the rostralmost portion of the colliculus where ipsilateral visual fields are represented (except in primates).
As in nonmammals, the ipsilateral isthmic afferents terminate extensively throughout the superficial retinal recipient zone and less densely below it (in the intermediate
gray) (Graybiel, ’79). Rodents are apparently unique in that
ipsilaterally projecting parabigeminal neurons are separated into noncontiguous dorsal and ventral cell groups by
an intermediate group of contralaterally projecting cells
Watanabe and Kawana, ’79; Linden and Perry, ’83; Kiinzle
and Schnyder, ’84). In other mammals, contralaterally projecting cells do not appear to be segregated.
The rostral nontopographic nucleus isthmi. The evidence for a nucleus similar to the rostral magnocellular
nucleus isthmi (Irnr) of turtles across the vertebrates is less
clear-cut, but suggestive data are available for a number of
classes. In turtles, Imr receives a nontopographic tectal
projection and then projects nontopographically to both Imc
and the tectum via bifurcating axons. The very large synaptic boutons given off by Imr axons are rigorously confined
to Imc and to the central and periventricular layers of the
tectum.
In bony fish, a tectal-receiving nucleus-the nucleus pretectalis-projects strongly to the topographic nucleus isthmi
(Ito et al., ’81; Sakamoto et al., ’81). Nucleus pretectalis
terminal boutons in nucleus isthmi are very large and
contain flat vesicles (Ito et al., ’82). Like turtle Imr, nucleus
pretectalis also projects to the tectum (Grover and Sharma,
’81).
In the frogs Rana and Acris, Udin (’87) recently demonstrated a projection to both the contralateral and ipsilateral
nucleus isthmi from a scattered group of large cells-the
anterodorsal nucleus-located in the mesencephalic tegmentum just rostral to nucleus isthmi. This region receives
a tectal input (Masino and Grobstein, ’85) and projects as
Fig. 14. Transverse view of a rostral magnocellular nucleus isthmi (Imr)
neuron. This cell was labeled by a small tedal injection (see inset). The
dendrites (stippled) extend rostrally and caudally and therefore appear
quite foreshortened in this transverse reconstruction. The robust, complexly
branched axon (origin a t open arrow) has three main parts. First, it gives
off several local collaterals within Imr that support about 200 large boutons.
Second, it sends two main myelinated trunks to the tectum; the thinner
branch innervates the caudal face of the tectum while the thicker terminates in the middle and rostral tectum. A portion of the tectal course of the
thick trunk (through which the cell was filled) is reconstructed in Figure
15. Finally, the neuron gives off three ventrally and caudally directed
trunks that emit large boutons at several different dorsoventral and mediolateral levels within the caudal magnocellular nucleus isthmi (Imc, not
shown). The inset shows the location of the neuron.
337
well to the tectum (Wilczynski and Northcutt, ’77). The
tegmento-isthmic projection seems rather nontopographic
at the single axon level (for example, Fig. 1A in Udin, ’87).
Its bilateral distribution may be related to the bilateral
projection of the medulla of frog nucleus isthmi.
In snakes, nucleus isthmi contains a mixture of small and
large neurons, both of which are labeled by ipsilateral tectal injections (Dacey and Ulinski, ’86b). Nucleus isthmi
injections, in turn, label thin and thick axons in the ipsilatera1 tectum. The spatially restricted terminal arbors of the
thin axons were already mentioned. The thick axons, by
contrast, each give rise to a widely spaced series of vertical
collaterals that nontopographically innervate the superficial gray layers and especially, the stratum zonale. This
contrasts with the deeper layer targets of turtle Imr. Snake
nucleus isthmi thus combines features of turtle Imc and
Imr.
In lizards, there is a distinct nucleus rostral to the topographic nucleus isthmi that closely resembles turtle Imr in
location, morphology, and connections (it receives tectal
afferents and projects back to the tectum and to the topographic nucleus) (Wang et al., ’83).The topography (if any)
of these projections is unknown.
In birds, the magnocellular nucleus isthmi is the probable
homologue of the turtle Imr. The avian nucleus receives a
nontopographic tectal input via the tectopontine pathway
(Hunt and Kiinzle, ’76; Hunt and Brecha, ’84) and may
project heavily to the topographic nucleus isthmi, Ipc, since
injections encroaching on the magnocellular nucleus result
in “heavy fibrous labeling” of Ipc (Hunt et al., ’77).Antibodies to glutamic acid decarboxylase (GAD) densely stain the
somata of magnocellular nucleus isthmi neurons but not
Ipc neurons; Ipc is instead filled with large-caliber GADstained axon terminals (personal observation, material
courtesy Catherine Carr) probably arising from neurons in
the magnocellular nucleus. The magnocellular nucleus also
projects to the deep layers of the tectum (Hunt and Brecha,
’84). Reubi and Cuenod (‘76) showed that Ipc stimulation
causes GABA release in the pigeon tectum; this may have
resulted from the antidromic activation of magnocellular
nucleus isthmi axons in Ipc that also project to the tectum.
(Abbreviations are confusing here because the avian “magnocellular nucleus isthmi” is usually written as “Imc” while
in turtles, “Irnc” stands for caudal magnocellular nucleus
isthmi, the equivalent of avian Ipc.)
In mammals, there are several tectal-recipientcell groups
just medial and rostral to the parabigeminal nucleus that
project back to the tectum (Graybiel, ’78; Kiinzle and
Schnyder, ’84). Cells in this region also project to the parabigeminal nucleus (Edwards, ’75; Sherk, ’79) and may constitute a mammalian counterpart to Imr, though none of
the cell groups in mammals are as architectonically distinct
as turtle Imr or bird magnocellular nucleus isthmi. Roldan
et al. (’83) have reported, in addition, a light, nontopographic projection to the tectum originating from within
the parabigeminal nucleus, which recalls the situation in
snakes.
Functional implications
A key feature of the circuitry interconnecting Imr, Imc,
and the tectum is that topographic and nontopographic
visual information is superimposed. Although the nontopographic Imr projection essentially ignores the retinotopic
organization of Imc and the tectum, it is strictly confined to
these two intimately related structures. Taken together
338
Figure 15
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
with behavioral, physiological, and histochemical data, the
present results suggest several testable hypotheses about
the function of these two ipsilateral loops.
Spatial selective attention. Ingle (‘75) demonstrated in
frogs that when a wormlike stimulus is moved briefly
enough to avoid eliciting orienting and snapping responses,
the stimulated region of the visual field becomes sensitized
for several seconds so that a second, similarly brief stimulus, at the same location can often release prey-catching.
Recordings made in the superficial laminae of the tectum
of immobilized frogs reveal “attention units” with small
receptive fields that give a slow, steady discharge for 3-6
seconds after a 1-or 2-second delay. These units are abolished by knife cuts at the caudolateral edge of the tectum
that interrupt the ipsilateral but not the contralateral isthmotectal pathway (Ingle, personal communication), suggesting that they are (or depend strongly on input from)
nucleus isthmi terminals. Interestingly, a similar pattern
of delayed, prolonged excitation has been recorded in the
nucleus isthmi of a lizard (Wang et al., ’83) and a teleost
fish Williams et al., ’83).
These results suggests that the topographically organized
ipsilateral isthmotectal pathway may provide punctate positive feedback to the tectum, with nucleus isthmi acting as
a sort of scratchpad on which interesting target locations
cab be temporarily written. Subtotal lesions of nucleus
isthmi in frogs result in a permanent contralateral visual
scotoma inside which prey and threatening stimuli are
ineffective in eliciting snapping and avoidance responses
(Caine and Gruberg, ’85).Outside the scotoma, prey-catching and threat-avoidance are normal. The scotoma appears
very similar to that produced by tectal lesions (Ingle, ’73;
see also parallels between the effects of tectal and isthmic
lesions in pigeons-Hodos and Karten, ’74; Jarvis, ’74),
suggesting that isthmic feedback may not only highlight a
339
+
Rostra1
Medial
Lateral
Caudal
Imr
Fig. 15. Continuation of the thicker tectal branch of the rostal magnocellular nucleus isthmi (Irnr) neuron shown in Figure 14.The main myelinated
trunk courses through the stratum griseum centrale (SGC) emitting thin,
vertically oriented strings of large boutons at about 200 pm intervals (branch
points A-E). For comparison, an entire Imc terminal arbor would fit between just two of these collaterals. In addition to the thin branches, thicker
myelinated collaterals arise at some branch points (one at A and two at C)
and course rostrally (3-D arrows) through the SGC and the stratum album
centrale (SAC) to reach the middle and far rostral tectum, emitting vertically oriented strings of boutons a t regular intervals the whole way (not
illustrated). A single Imr axon, thus, sparsely innervates a large percentage
of the tectal hemisphere. The inset shows the location of the high-power
view.
Fig. 16. Horizontal view of a rostral magnocellular nucleus isthmi (Imr)
neuron. This cell was labeled by a small tectal injection. Its large, rostrocaudally elongated, sparsely branched dendritic field is typical for Imr neurons,
but its soma is more eccentrically placed than most. The robust (6 pm
diameter) axon (origin at open arrow) trifurcated almost immediately into a
thick tectal trunk and two other myelinated trunks, labeled A and B, that
descend to caudal magnocellular nucleus isthmi (Imc). The terminal arborization in Imc arising from trunks A and B is illustrated in Figure 17 (branch
B itself bifurcates on the way to Imc). This neuron apparently lacked local
collaterals in Imr, which were usually found on such neurons. The inset
shows the location of the high-power view.
100 urn
Figure 16
M.I. SERENO AND P.S. ULINSKI
342
certain visual field location but may be involved in initiating the tectoreticular-mediated orienting response toward it.
The role of the ipsilateral isthomotectal projection in spatial selective attention could be investigated by locally
stimulating the topographic nucleus isthmi and determining the effects on tectal output cells and orienting movements and by recording from that nucleus during awake
orienting. In vitro slice experiments would help to determine if the delayed, prolonged excitation attributed to ipsilaterally projecting nucleus isthmi neurons is generated
intrinsically or by positive feedback. Tectally mediated visuomotor responses are influenced by other cell groups (for
example, pretectum) and it would be interesting to compare
nucleus-isthmi-mediated effects to those mediated by other
structures.
Local-global interactions. Frost et al. ('81, '84) showed
that direction-selective neurons in all but the most superficial layers of the pigeon tectum typically have large stimulus-specific inhibitory surrounds extending far beyond the
edges of their excitatory receptive fields and sometimes
including the entire visual field. Moving a random dot
pattern through the surround completely suppresses the
response to an optimal receptive field stimulus (moving bar
or random dot patch) if the surround moves "in phase" with
it, but enhances the response if the surround moves in the
opposite direction. (By themselves, the different surround
stimuli produce no effect as long as they remain outside the
excitatory receptive field.) In addition, moving the surround
in a given direction changes the best direction of the receptive field center to the opposing direction for many of these
cells (Frost and Nakayama, '83). This effect is so strong
that the peak in the direction tuning curve of the receptive
field center can be shifted 180"C. Stimulus-specific inhibitory surrounds have also been observed in the tectum of
Rostml
part of
Fig. 17. (See the two previous pages for figure.) Termination of rostral
magnocellular nucleus isthmi (lmr) axon (branches A and B from Imr cell
shown in Fig. 16) in caudal magnocellular nucleus isthmi (Imc). This Imr
cell terminal arbor was labeled by a small tectal injection (via the axon's
tectal branch) and reconstructed from seven horizontal sections. Three thick,
myelinated trunks (one is branch A while two are from branch B, which
bifurcated on its way to Imc) enter Imc from the front. These each subdivide
into several myelinated branches that begin wandering to and fro, emitting
strings of large 12-3.5 pm) boutons. The boutons (1,840 total) are distributed
across many mediolateral and dorsoventral levels of the Imc cell plate but
remain strictly confined to the nucleus. Small, overlapping clusters of tectoisthmic terminals and Imc neurons also labeled by the tectal injection were
located in the lateral third of the nucleus, emphasizing the nontopographic
nature of this terminal arbor. Three asterisks indicate where small trunks
were lost before they ended. The inset shows the location of the terminal
arbor and the outlines of Imc in some of the sections used in the
reconstruction.
Fig. 18. Horizontal view of a rostral magnocellular nucleus isthmi (Imr)
neuron. This cell was labeled by a small tectal injection. The large, rostrocaudally elongated dendritic field is seen almost en face in this reconstruction. As with the Imr neuron in Figure 14, the robust axon of this neuron
(origin at open arrow) emits a few local collaterals in Imr (supporting about
150 boutons), a thick tectal branch that innervates a large portion of the
tectum, and several branches to different parts of the map in caudal magnocellular nucleus isthmi (Imc). The dendrites (stippled) of this and other
Imr neurons are covered with fine spicules like those seen on dorsal and
intermediate tectobulbar neurons (Sereno and Ulinski, '85).The inset shows
the location of the neuron.
CG
TB
Tor'
TBv
Figure 18
ORGANIZATION OF TURTLE PSTHMIC NUCLEI
Fig. 19. High-magnification view (upper left-hand corner) of the nontopographic background labeling of Imc shown in the low-power photomicrograph i n Figure 1B. The axon terminals here were labeled by a large tectal
injection. They appear not to be tectal afferents, but rather, branched
afferents from Imr that innervate both Imc and the tectum (see text and
Figs. 14-18). The robust (2-3.5 fim diameter) boutons are distributed quite
uniformly throughout the entire volume of Imc in marked contrast to the
restricted band of tectoisthmic terminals and Imc cells also labeled by the
injection. The nontopographic label is, however, rigorously confined to Imc,
effectively outlining the nucleus.
343
Arbitration of competing stimuli. An alternate function
of the nontopographic nucleus isthmi may be to participate
closely with topographic nucleus isthmi in the construction
of a “winner-take-all” network Feldman, ’82; Koch and
Ullman, ’85) in the tectum. An obvious problem in generating tectoreticular-mediated orienting responses is that
there may be more than one possible interesting new target
simultaneously present in the pattern of activity sent to
the superficial layers of the tectum by the retina; but only
one locus in the deeper-lying motor map (see Sereno and
Ulinski, ’85, for discussion) must be activated to avoid orienting t o the “average” of the stimuli, Koch and Ullman
(‘85) present two model networks that perform a maximumfinding computation. The two tecto-isthmo-tectal loops described here appear to form a network like their first one.
They set up a “saliency map” (cf. the superficial tectum)
that directly drives a “winner-take-all” map (cf. the intermediate and deep tectum); in the latter, the most active
locus suppresses all other activated loci while itself being
driven to saturation. The update rule for the “winner-take
all” network is: if a unit’s input is greater than the average
input for the whole map, increase that unit’s output in
proportion to the difference, while if its input is below
average, decrease its output in the same manner. Something similar to this in principle may describe the action of
the ipsilateral isthmic nuclei on the motor map in the
tectum. The positive feedback loop between the tectum and
topographic nucleus isthmi (or the intrinsic properties of
topographic nucleus isthmi neurons) could provide a multiplicative or exponential augmentation of activated loci necessary to always give the most active locus the advantage.
The nontopographic nucleus isthmi is in a position to calculate the average input to tectoreticular neurons, and its
direct and indirect nontopographic outputs to the tectum
could uniformly inhibit all tectal loci, eventually suppressing all but the most active locus. The local collaterals of the
nontopographic nucleus isthmi neurons could control the
gain of the inhibition. The main difference from Koch and
Ullman’s presentation is that excitatory and inhibitory influences are carried by separate pathways that differ in
more than their sign of action.
Inactivation of nontopographic nucleus isthmi should in
this case result in disinhibition of tectal and topographic
nucleus isthmi neurons to receptive field stimuli along with
a loss of nonspecific surround inhibition (direction-selective
inhibition, for instance, might remain if it is mediated
through pretectal or intrinsic tectal circuits). In addition,
this scenario predicts no direction-selective neurons in nontopographic nucleus isthmi. At the behavioral level, ibotenic acid lesions in that nucleus should produce deficits in
orienting when several salient stimuli are present.
frogs (Burghagen and Ewert, ’83)and in the superior collicACKNOWLEDGMENTS
ulus of cats (Sterling and Wickelgren, ’69).
This work was supported by PHS grant NS 12518 and an
These mechanisms could mediate the discrimination of
object motion from self-induced motion (Ewert, ’84; Allman NSF predoctoral fellowship.
et al., ’85). To find out whether the nontopographic nucleus
LITERATURE CITED
isthmi is involved in generating these effects, one could
record from a tectal cell with a surround and then inacti- Adams, J.C. (1977) Technical considerations on the use of horseradish peroxidase as a neuronal marker. Neuroscience 2:141-145.
vate the nontopographic nucleus isthmi with lidocaine to
J.,F. Miezin, and E. McGuinness (1985)Stimulus specific responses
see if the stimulus-specific surround inhibition is made Allman,
from far beyond the classical receptive field Neurophysiological mechnonselective or abolished. One would also expect to find
anisms for local-global comparisons in visual neurons. Annu. Rev. Neurosci. 8:407-429.
wide-field direction-selective cells in nontopographic nucleus isthmi and small-field cells with large surrounds in Brauth, S.E., A. Reiner, C.A. Kitt, and H.J. Karten (1983) The substance-Pcontaining striatotegmental path in reptiles: An immunohistochemical
topographic nucleus isthmi.
study. J. Comp. Neurol. 219:305-327.
M.I. SERENO AND P.S. ULINSKI
344
Imr neuron
Imc neuron
I
I
I
I
I
I
JJ,
I
I
I
I
I
I
I
I
Figure 20
ORGANIZATION OF TURTLE ISTHMIC NUCLEI
Burghagen, H., and J.-P. Ewert (1983) Influence of the background for
discriminating object motion from self-induced motion in toads Bufo
bufo
J. Comp. Physiol. 152t241-249.
Caine, H.S., anc! E.R. Gruberg (1985) Ablation of nucleus isthmi leads to
loss of specific visually elicited behaviors in the frog Rana pipiens.
Neurosci. Lett. 54t307-312.
Cajal, S. Ra.mon y (1899) Adiciones a nuestros trabojos sobre 10s centros
opticos de las aves. Rev. Trimest. Microgr. 4t77-86.
Cruce, W.L.R., and R. Nieuwenhuys (1974)The cell masses i n the brainstem
of the turtle Testudo hermanni. J. Comp. Neurol. 156t277-306.
Dacey, D.M., and P.S. Ulinski (1986a)The optic tectum of the eastern garter
snake, Tharnnophis sirtalis: 11. Morphology of efferent cells. J. Comp.
Neurol. 245:198-327.
Dacey, D.M., and P.S. Ulinski (1986b)The optic tectum of the eastern garter
snake, Thamnophis sirtailis: V. Morphology of brainstem afferents and
general discussion. J. Comp. Neurol. 245423-453.
Desan, P.H., E.R. Gruberg, and F. Eckenstein (1984)A cholinergic projection
from the nucleus isthmi to the optic tectum in turtle and frog. Proc. SOC.
Neurosci. 10:575 (Abstract).
Edwards, S.B. (1975) Autoradiographic studies of the projections of the
midbrain reticular formation: Descending projection of the nucleus cuneiformis. J. Comp. Neurol. 161t341-358.
Ewert, J.-P. (1984) Tectal mechanisms that underlie prey catching and
avoidance behaviors in toads. In H. Vanegas (ed): Comparative Neurology of the Optic Tectum. New York: Plenum Press, pp. 247-416.
Feldman, J.A. (1982) Dynamic connections in neural networks. Biol. Cybern. 46:27-39.
Foster, R.E. and W.C. Hall (1975) The connections and laminar organization
of the optic tectum in a reptile (Iguana iguana). J. Comp. Neurol.
163:397-426.
Frost, B.J., P. Cavanagh, and B. Morgan (1984) Kinetograms drive deep
tectal cells in pigeons. Proc. Soc. Neurosci. lot574 (Abstract).
Frost, B.J., and K. Nakayama (1983) Single visual neurons code opposing
motion independent of direction. Science 220:744-745.
Frost, B.J., P.L. Scilley, and S.C.P. Wong (1981) Moving background patterns reveal double-opponency of directionally specific pigeon tectal
neurons. Exp. Brain Res. 4.2173-185.
Glasser, S., and D. Ingle (1978)The nucleus isthmi as a relay station in the
ipsilateral visual projection to the frog’s optic tectum. Brain Res.
159:214-2 18.
Glenn, L.L., and R.E. Burke (1981)A simple and inexpensive method for 3dimensional visualization of neurons reconstructed from serial sections.
J. Neurosci. Methods. 4t127-134.
Graybiel, A.M. (1978) A satellite system of the superior colliculus: The
parabigeminal nucleus and its projection to the superficial collicular
layers. Brain Res. 145,365-374.
Grobstein, P., C. Comer, M. Hollyday, and S.M. Archer (1978) A crossed
isthmo-tectal projection and its movement in the ipsilateral visuo-tectal
projection. Brain Res. 156t117-123.
Grobstein, P., and C. Comer (1983) The nucleus isthmi as an intertectal
relay for the ipsilateral oculotectal projection in the frog Rana pipiens.
J. Comp. Neurol. 21754-74.
Grover, B.G., and S.C. Sharma (1981) Organization of extrinsic tectal connections in goldfish (Carussiusauratus). J. Comp. Neurol. 196:471-488.
Gruberg, E.R., and J.Y. Lettvin (1980) Anatomy and physiology of a binocular system in the frog, Ranapipiens. Brain Res. 192:313-325.
Gruberg, E.R., and S.B. Udin (1978) Topographic projections between the
nucleus isthmi and the tectum of the frog, Rana pipiens. J. Comp.
Neurol. 179:487-500.
Harting, J.K., W.C. Hall, I.T. Diamond, and G.F. Martin (1973)Anterograde
degeneration study of the superior colliculus i n Tupaia glis: Evidence
for a subdivision between superficial and deep layers. J. Comp. Neurol.
148:361-386.
a,).
Fig. 20. Schematic diagram of the typical axonal and dendritic morphology of single caudal magnocellular nucleus isthmi (Imc) and rostra1 magnocellular nucleus isthmi (Imr) neurons, drawn to the same scale. The two
isthmic nuclei have been enlarged relative to the tectum for clarity. The
restricted dendritic and axonal arbors of Imc neurons contrast with the
strikingly nontopographic projection of the larger Imr neurons not only to
the tectum but also to Imc.
345
Harting, J.K., M.F. Huerta, A.J. Frankfurter, N.L. Strominger, and G.L.
Royce (1980) Ascending pathways from the monkey superior colliculus:
An autoradiographic analysis. J. Comp. Neurol. 192852-882.
Henley, J.M., J.M. Lindstrom, and R.E. Oswald (1986)Acetylcholine receptor synthesis in retina and transport to optic tectum in goldfish. Science
232:1627-1629.
Hodos, W., and H.J. Karten (1974)Visual intensity and pattern discrimination deficits after lesions of the optic lobe in pigeons. Brain Behav. Evol.
9t165-194.
Hunt, S.P., and N. Brecha (1984) The avian optic tectum: A synthesis of
morphology and biochemistry. In H. Vanegas (ed): Comparative Neurology of the Optic Tectum. New York: Plenum Press, pp. 619-648.
Hunt, S.P., and H. Kiinzle (1976)Observations on the projections and intrinsic organization of the pigeon optic tectum: An autoradiographic study
based on anterograde and retrograde, axonal and dendritic flow. J.
Comp. Neurol. 170:153-172.
Hunt, S.P., P. Streit, H. Kunzle, and M. Cuenod (1977) Characterization of
the pigeon isthmotectal pathway by selective uptake and retrograde
movement of radioactive compounds and by Golgi-like horseradish peroxidase labelling. Brain Res.. 129t197-212.
Ingle, D. (1973)Two visual systems in the frog. Science 18It1053-1055.
Ingle, D. (1975)Selective visual attention in frogs. Science 188t1033-1035.
Ito, H., N. Sakamoto, and K. Takatsuji (1982) Cytoarchitecture, fiber connections and ultrastructure of nucleus isthmi in a teleost (Nuuodon
modestus) with a special reference to degenerating isthmic afferents
from optic tectum and nucleus pretectalis. J. Comp. Neurol. 205t299311.
Ito, H., H. Tanaka, N. Sakamoto, and Y. Morita (1981) Isthmic afferent
neurons identified by retrograde HRP method in a telost, Nauodon
modestus. Brain Res. 207t163-169.
Jarvis, C.D. (1974) Visual discrimination and spatial localization deficits
after lesions of the tectofugal pathways in pigeons. Brain Behav. Evol.
9t195-228.
Koch, C., and S. Ullman (1985) Shifts in selective attention: Toward the
underlying neural circuitry. Hum. Neurobiol. 4:219-227.
Kiinzle, H., and H. Scbnyder (1984)The isthmus-tegmentum complex in the
turtle and rat: A comparative analysis of its interconnections with the
optic tectum. Exp. Brain Res. 56:509-522.
Lazar, G. (1984) Structure and connections of the frog tectum. In H. Vanegas
(ed): Comparative Neurology of the Optic Tectum. New York Plenum
Press, pp. 185-210.
Linden, R., and V.H. Perry (1983) Retrograde and anterograde-transneuronal degeneration in the parabigeminal nucleus following tectal lesions in developing rat. J. Comp. Neurol. 218t270-281.
Masino, T., and P. Grobstein (1985)The organization of tectal projections to
the ventral midbrain in Rana pipiens. Proc. SOC.Neurosci. 11:289
(Abstract).
Mendez-Otero, R., C.E. Rocha-Miranda, and V.H. Perry (1980) The organization of the parabigemino-tectal projections in the opossum. Brain Res.
198; 183-189.
Reubi, J.X., and M. Cuenod (1976) Release of exogenous glycine in the
pigeon optic tectum during stimulation of a midbrain nucleus. Brain
Res. 112t346-361.
Roldan, M., F. Reinoso-Suarez, and A. Tortelly (1983)Parabigeminal projections to the superior colliculus. Brain Res. 28O:l-13.
Sakamoto, N., H. Ito, and S. Ueda (1981) Topographic projection between
the nucleus isthmi and the optic tectum in a teleost, Nauodon modestus.
Brain Res. 224r225-234.
Sereno, M.I. (1985) Tectoreticular pathways in the turtle, Pseudemy.s scripta.
I. Morphology of tectoreticular axons. J. Comp. Neurol. 233t48-90.
Sereno, M.I., and P.S. Ulinski (1985)Tectoreticular pathways in the turtle,
Pseudemys scripta. 11. Morphology of tectoreticular cells. J. Comp. Neurol. 233:91-114.
Sherk, H. (1979) Connections and visual-field mapping in cat’s tectoparabigeminal circuit. J. Neurophysiol. 42t1656-1668.
Sterling, P., and B.G. Wickelgren (1969) Visual receptive fields in the
superior colliculus of the cat. J. Neuroghysiol. 321-15.
ten Donkelaar, H.J., and R. Nieuwenhuks (1979) The brain stem. In C.
Gans, R.G. Northcutt, and P.S. Ulinski (eds): Biology of the Reptilia,
Vol. 10. Neurology B. London: Academic Press, pp. 133-200.
Udin, S.B. (1986) A projection from the mesencephalic tegmentum to the
nucleus isthmi i n the frogs, Rana pipiens and Acris crepitans. Neuroscience (in press).
346
Wang, R., J.L. Kubie, and M. Halpern (1977) Brevital Sodium: An effective
anesthetic agent for performing surgery on small reptiles. Copeia
1977:739-743.
Wang, S.-R., K. Yan, Y..-T Wang, S.J. Jiang, and X.-S Wang (1983) Neuroanatomy and electrophysiology of the lacertilian nucleus isthmi. Brain
Res. 275355360,
Watanabe, K., and E. Kawana (1979) Efferent projections of the parabigeminal nucleus in rats: A horseradish peroxidase (HFtP)study. Brain Res.
168:1-11.
M.I. SERENO AND P.S. ULINSKI
Wilczynski, W., and R.G. Northcutt (1977) Afferents to the optic tectum of
the leopard frog: An HRP study. J. Comp. Neurol. 173.219-230.
Williams, B., N. Hernandez, and H. Vanegas (1983) Electrophysiological
analysis of the telostean nucleus isthmi and its relationship with the
optic tectum. J. Comp. Physiol. A 152.545-554.
Vanegas, H., S.O.E. Ebbesson, and M. Laufer (1984) Morphological aspects
of the telostean optic tectum. In H. Vanegas (ed): Comparative Neurology of the Optic Tectum. New York: PIenum Press, pp. 93-120.