* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
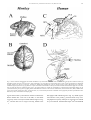
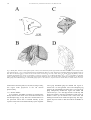
Download Eye fields in the frontal lobes of primates
Holonomic brain theory wikipedia , lookup
Neuroscience in space wikipedia , lookup
Brain–computer interface wikipedia , lookup
Brain Rules wikipedia , lookup
Synaptic gating wikipedia , lookup
Neurophilosophy wikipedia , lookup
Affective neuroscience wikipedia , lookup
Neurolinguistics wikipedia , lookup
Neuropsychology wikipedia , lookup
Optogenetics wikipedia , lookup
Cortical cooling wikipedia , lookup
Human multitasking wikipedia , lookup
History of neuroimaging wikipedia , lookup
Time perception wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
Environmental enrichment wikipedia , lookup
Neuroanatomy wikipedia , lookup
Emotional lateralization wikipedia , lookup
Neuroesthetics wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Neurostimulation wikipedia , lookup
Neuroanatomy of memory wikipedia , lookup
Human brain wikipedia , lookup
Evoked potential wikipedia , lookup
Metastability in the brain wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Neuroeconomics wikipedia , lookup
Neuroplasticity wikipedia , lookup
Transsaccadic memory wikipedia , lookup
Embodied language processing wikipedia , lookup
Aging brain wikipedia , lookup
Cognitive neuroscience of music wikipedia , lookup
Process tracing wikipedia , lookup
Motor cortex wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Brain Research Reviews 32 Ž2000. 413–448 www.elsevier.comrlocaterbres Full-length review Eye fields in the frontal lobes of primates Edward J. Tehovnik ) , Marc A. Sommer, I-Han Chou, Warren M. Slocum, Peter H. Schiller Department of Brain and CognitiÕe Sciences, Massachusetts Institute of Technology, E25-634, Cambridge, MA 02139, USA Accepted 19 October 1999 Abstract Two eye fields have been identified in the frontal lobes of primates: one is situated dorsomedially within the frontal cortex and will be referred to as the eye field within the dorsomedial frontal cortex ŽDMFC.; the other resides dorsolaterally within the frontal cortex and is commonly referred to as the frontal eye field ŽFEF.. This review documents the similarities and differences between these eye fields. Although the DMFC and FEF are both active during the execution of saccadic and smooth pursuit eye movements, the FEF is more dedicated to these functions. Lesions of DMFC minimally affect the production of most types of saccadic eye movements and have no effect on the execution of smooth pursuit eye movements. In contrast, lesions of the FEF produce deficits in generating saccades to briefly presented targets, in the production of saccades to two or more sequentially presented targets, in the selection of simultaneously presented targets, and in the execution of smooth pursuit eye movements. For the most part, these deficits are prevalent in both monkeys and humans. Single-unit recording experiments have shown that the DMFC contains neurons that mediate both limb and eye movements, whereas the FEF seems to be involved in the execution of eye movements only. Imaging experiments conducted on humans have corroborated these findings. A feature that distinguishes the DMFC from the FEF is that the DMFC contains a somatotopic map with eyes represented rostrally and hindlimbs represented caudally; the FEF has no such topography. Furthermore, experiments have revealed that the DMFC tends to contain a craniotopic Ži.e., head-centered. code for the execution of saccadic eye movements, whereas the FEF contains a retinotopic Ži.e., eye-centered. code for the elicitation of saccades. Imaging and unit recording data suggest that the DMFC is more involved in the learning of new tasks than is the FEF. Also with continued training on behavioural tasks the responsivity of the DMFC tends to drop. Accordingly, the DMFC is more involved in learning operations whereas the FEF is more specialized for the execution of saccadic and smooth pursuit eye movements. q 2000 Elsevier Science B.V. All rights reserved. Keywords: Dorsomedial frontal cortex; Supplementary eye field; Frontal eye field; Oculomotor behaviour; Skeletomotor behaviour; Learning; Monkey; Human Contents 1. Introduction . ....................................................................... 2. The anatomy of the DMFC and the FEF . 2.1. The anatomy of the DMFC . . . . . 2.2. The anatomy of the FEF . . . . . . . 2.3. Summary . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 416 416 419 420 3. Electrical stimulation of the DMFC and the FEF . . . . . . . . . . . . . . . . . . . . . . . . . . 3.1. Maps and coding schemes in monkeys . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3.2. Electrical stimulation and the behavioural state of monkeys . . . . . . . . . . . . . . . . . . 3.3. Skeletomotor responses elicited by electrical stimulation of the DMFC and FEF in monkeys 3.4. DMFC and FEF stimulation in humans . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 3.5. Summary . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 420 420 421 423 423 423 4. Lesions and reversible inactivation of the DMFC and the FEF . . . . . . . . . . . . . . . . . . . . . . 4.1. The effects of lesions and reversible inactivation of the DMFC in monkeys . . . . . . . . . . . . 4.2. The effects of lesions and reversible inactivation of the FEF in monkeys . . . . . . . . . . . . . . 4.3. Direct comparison of DMFC and FEF lesions and inactivation on visually guided eye movements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 423 423 424 426 ) . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . Corresponding author. Fax: q1-617-253-8943; e-mail: [email protected] 0165-0173r00r$ - see front matter q 2000 Elsevier Science B.V. All rights reserved. PII: S 0 1 6 5 - 0 1 7 3 Ž 9 9 . 0 0 0 9 2 - 2 . . . . . . . . . . . . . . . . . . . . . . . . 414 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 414 4.4. The effects of DMFC and FEF damage in humans . 4.5. Summary . . . . . . . . . . . . . . . . . . . . . . . ................................................ ................................................ 427 428 . . . . . . . . 429 429 430 431 ........................................................... 432 5. Single-cell recordings in the DMFC and FEF . . . . . . 5.1. The characteristics of single neurons in the DMFC 5.2. The characteristics of single neurons in the FEF . . 5.3. Summary . . . . . . . . . . . . . . . . . . . . . . . 6. Event-related potentials in humans . 7. Metabolic imaging of the DMFC and FEF in humans . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 435 ......................................................... 436 ................................................................. 437 ........................................................................ 439 9. Misplaced human FEF? . Acknowledgements . References . . . . ................................................. 8. Motor learning and the DMFC and FEF 10. Discussion . . . . ..................................................................... 441 .......................................................................... 441 1. Introduction Two areas within the frontal lobes play important roles in eye movement control. One area resides laterally and is known as the frontal eye field ŽFEF.. The other resides medially and dorsally and is often termed the supplementary eye field Žsee Schall w230x.. As will become clear from this review, the size, exact location, and precise function of this second eye field are still under debate; therefore, we refer to it using the anatomical designation, the dorsomedial frontal cortex ŽDMFC., or more specifically ‘‘the eye field within the DMFC’’. Our examination of the literature suggests that the differences between the two frontal lobe eye fields, the FEF and DMFC, are profound. The FEF plays an important role in generating eye movements, utilizing an eye-centered code, whereas the DMFC integrates oculomotor and skeletomotor behaviour, utilizing predominantly a head-centered code, and is involved in visuo-motor learning. One of the first investigators to examine the role of the cortex in eye-movement control was Hitzig w106x. Studying patients unable to move their eyes voluntarily, Hitzig found that he could induce eye movements by delivering pulses of direct current through electrodes attached to the skull. Hitzig believed that the eye movements produced were due to excitation of brain tissue. However, the resulting eye movements could have been due to surface conduction to the eye muscles; to resolve this ambiguity, Hitzig needed to stimulate the brain directly. In collaboration with Fritsch, he performed these experiments on dogs. In 1870 they established that neocortical stimulation elicited movement of the extremities, but they failed to produce eye movements w68,307x. Soon thereafter, Ferrier w59x discovered that eye movements could be evoked in monkeys from parts of the frontal, parietal, and temporal lobes, the superior colliculi, and the cerebellum. The frontal lobe eye field that Ferrier described extends from the arcuate sulcus to the midline ŽFig. 1A,B.. It is remarkable that the only major modification to this eye field since Ferrier’s original publication has been a split into two subfields, one wholly within the curve of the arcuate sulcus and the other about a centimeter away, near the midline. At the end of the 19th century, tissue removal and electrical stimulation were the main techniques used to examine brain function in animals. Interpretation of the former was difficult due to qualitative, irreproducible behavioural testing and post-operative infections w77,107x. Electrical stimulation results, on the other hand, were more unequivocal and enlightening. Beevor and Horsley w10x confirmed Ferrier’s findings showing that eye movements could be elicited from both the prearcuate gyrus and the peri-midline cortex in the frontal lobe of monkeys. They further showed that eye movements could be evoked from the lateral frontal convexity of an ape as well w11x, and this was confirmed by Grunbaum and Sherrington w92x and ¨ w x Leyton and Sherrington 144 after the turn of the century in orangutans, chimpanzees, and gorillas. However, neither of these groups uncovered the existence of an eye field near the midline of their apes. Subsequently, investigators focused on the prearcuate eye field, which eventually became known as the FEF; the medial eye field representation was essentially ignored for decades. Neurosurgeons were, in large part, responsible for reviving scientific curiosity about the eye fields in the DMFC. Foerster w60,61x reported that there were two main E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 415 Fig. 1. Classic results in examining the neocortical contribution to eye movements. Anterior is to the left and dorsal to the top unless otherwise noted. ŽA. Stimulation regions of Ferrier w59x, lateral view. In the frontal lobe, eye movements were evoked from region 12 only Žborder surrounding it is emphasized in bold.. Species was Macaca cynomolgus, according to Walker w302x, although this was not noted in Ferrier’s paper. ŽB. Dorsal view of Ferrier’s stimulation regions Žanterior at top.. ŽC. Foerster’s map of movements and sensations evoked by electrical stimulation of human brain w60x. The two frontal lobe eye fields he found are outlined in bold. Note, in an apparent attempt to simplify his presentation, he superimposed his functional findings on the cytoarchitectural map of Vogt and Vogt w296x. Human brain landmarks relevant to this review are labeled: IFS, inferior frontal sulcus; MFG, middle frontal gyrus; SFS, superior frontal sulcus; PCS, precentral sulcus; CS, central sulcus Žalso known as Rolandic sulcus.. ŽD. Map of human peri-Rolandic motor and sensory areas, from Penfield and Jasper w194x. regions from which eye movements could be evoked in the human frontal lobe. One was in the FEF, on the lateral convexity of the cortex Žlower bold-outlined region in Fig. 1C., and the other was in Vogts’ area 6ab, medial to the FEF Župper bold-outlined region in Fig. 1C.. Both regions were anterior to the primary, precentral motor strip. Foerster w60x did not specify what part of Area 6ab was related to eye movements. Penfield and Jasper w194x and Penfield 416 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 and Rasmussen w195x clarified the location of the 6ab eye field in their subsequent studies of the human brain, localizing it to the rostral part of the supplementary motor area ŽFig. 1D.. Penfield also confirmed Foerster’s finding that the human FEF was on the lateral convexity, anterior to the precentral motor strip ŽFig. 1D, lower set of eyes.. At about the same time, Woolsey w309x and Woolsey et al. w311x showed in an assortment of animals that the representation of the body was oriented eyes-rostrally and tailcaudally in the DMFC of every animal studied. Since the 1950s, research directed toward the neural control of eye movements has profited from five major methodological advances: Ž1. the development of a variety of microelectrodes suitable for single-cell recordings and for microstimulation; Ž2. the emergence of instrumentation that allowed investigators to accurately measure eye movements; Ž3. the introduction of behavioral methods that made it possible to train animals to move their eyes in response to sensory stimuli in predictable, reproducible ways; Ž4. the development of techniques that allowed for making focal brain lesions or for reversibly inactivating selected brain areas using a variety of pharmacological agents; and Ž5. the application of various imaging procedures to localize areas involved in eye-movement control. This review focuses extensively on research conducted using these methods. We will compare and contrast the function and organization of the two eye fields in the frontal lobes based on research carried out in primates, including humans. 2. The anatomy of the DMFC and the FEF 2.1. The anatomy of the DMFC 2.1.1. Monkey DMFC In monkeys, the DMFC is situated anterior to the motor cortex. It occupies a region between the superior limb of the arcuate sulcus and the cerebral midline dorsal to the cingulate cortex. Brodmann w29x defined this area as the mesial portion of area 6. Subsequently, this region was partitioned. Vogt and Vogt w295x divided the DMFC into rostral and caudal zones: 6ab and 6aa , respectively ŽFig. 2A.. von Bonin and Bailey w297x also partitioned the DMFC into rostral and caudal zones, but the rostral zone was appreciably smaller. Barbas and Pandya w8x divided the DMFC into three regions, a medial region they called MII, a rostrolateral region they called 6DR, and a caudal region they called 6DC. Some have also suggested that the DMFC includes area 8b w147x, which is situated immediately adjacent to the anterior border of area 6 w20x. Woolsey et al. w311x observed that the DMFC contains a somatotopic map with the head represented rostrally and the tail represented caudally. Although some have disputed this general topography w305x, there is now overwhelming evidence corroborating the original observations of Woolsey w27,64,66,86,110,149,166,228,243,275,308x. Single-unit recording experiments have shown that neurons modulated by eye movements are located rostrally, that neurons modulated by hindleg movements are located Fig. 2. Monkey DMFC. Each panel shows a top view of one hemisphere of the frontal cortex of a macaque monkey. ŽA. The DMFC is divided according to the scheme of Vogt and Vogt w295x. ŽB. Each oval represents a region from the DMFC that contained cells modulated by eye, forelimb, and hindleg movements in the same monkey ŽBrinkman and Porter w27x, monkey SMA-3.. ŽC. Each oval represents a region from the DMFC from which movements could be evoked using electrical stimulation ŽLuppino et al. w149x, monkey MK-7r.. ŽD. The DMFC is divided according to the scheme of Matelli et al. w159x. A scale bar is shown at the bottom. The central sulcus ŽCs., arcuate sulcus ŽAs., and principal sulcus ŽPs. are indicated in ŽA.. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 caudally, and that neurons modulated by forelimb movement are located in between the rostral and caudal extremes of the DMFC ŽRef. w27x; see Fig. 2B.. Neurons with responses modulated by distal forelimb movement are found anterior to those modulated by proximal forelimb movements. These findings concur with results obtained using microstimulation ŽRefs. w149,166x; see Fig. 2C.. 2.1.2. Multiple sub-areas of monkey DMFC Based on cytoarchitectonics, Matelli et al. w159x partitioned the DMFC into four regions: F2, F3, F6, and F7 ŽFig. 2D.. More recently, these regions have been given functional labels now adopted by Tanji and colleagues under the premise that these regions are functionally distinct w69,161,162,173,254,255,277,279x. These labels have also been used to organize data from functional imaging experiments carried out on humans w204x. In this nomenclature, F2 corresponds with the premotor area, F3 with the supplementary motor area, F6 with the pre-supplementary motor area, and F7 with the supplementary eye fields. The subdivisions, F2, F3, F6, and F7, as established by Matelli et al. w159x, were based on six macaque monkeys. As sections were taken more rostrally in the DMFC, proceeding from F3 to F6 or from F2 to F7, the cells in layer V exhibited a noticeable decrease in size. 1 Layer V was thicker in F3 than in F6 and layer V was thicker in F2 than in F7. Progressing from caudal to rostral regions of F3, a decrease in the frequency of the giant pyramidal cells was observed. Although Matelli et al. w159x suggest that F2 differs from F3 and that F6 differs from F7, this is not apparent from their figures. 2 Based on their illustrations, one would be hard put to establish a difference between F2 and F3 and a difference between F6 and F7. Furthermore, a discrete border between F3 and F6 and between F2 and F7 is hard to discern in their Fig. 3, which depicts horizontal sections including F3 and F6, and in their Fig. 7 which depicts parasagittal sections including F2 and F7. Current anatomical techniques do not convincingly denote marked differences between these areas other than cell size and laminar thickness. Even though the cytoarchitectonics of the DMFC do not support the subdivisions of Matelli et al. w159x, the projections from these regions might favor a segregation such that eye-movement areas only innervate F7 Žthe supplementary eye fields.. The FEF and superior colliculus are connected with area F7 w5,67,108,132,231,257x, yet they are also connected with areas F2, F3 and F6 w5,128, 132,231,257x. Studies examining the projections between the DMFC and motor cortex show that the entire DMFC Ži.e., F2, F3, F6, and F7. sends projections to the forelimb area of the 1 Matelli et al. w159x: their Fig. 6, cf. F3 and F6 and their Fig. 9, cf. F2 and F7. 2 Compare Fig. 5 with Fig. 8 of Matelli et al. w159x. 417 motor cortex w56,79,129,140,146,160x. Motor cortex, however, does not project to F6 and F7 w150x, although it does innervate F2 and F3 w140,150x. Thus the cytoarchitectonics and the efferent and the afferent connections of the DMFC do not provide an unambiguous way of subdividing the DMFC according to the scheme of Matelli et al. w159x. 2.1.3. Neuron size and somatotopy of monkey DMFC Matelli et al. w159x observed that the thickness of cortex decreases from caudal to rostral DMFC. 3 The number of cells within a cylinder of constant cross-sectional area of 1 mm2 of cerebral cortex is constant w16,23,217x, which makes it understandable why cortical thickness from caudal to rostral portions of the DMFC decreases concurrently with decreases in cell size as illustrated by Matelli et al. w159x. Mitz and Wise w166x show that the number of cells with large diameters Žover 600 mm2 . also decreases systematically from caudal to rostral DMFC. It is well-known that there is a direct correspondence between cell size and axon diameter w259x and that the larger the cell, the further the axon tends to project from the cell body w114,172x. That cells decrease in size going from caudal to rostral DMFC concurs with the somatotopy of the DMFC. Caudal DMFC, which has been associated with the hindlimbs, projects as far as the lumbarrthoracic cord w99,110x; intermediate DMFC, which has been associated with forelimbs, projects as far as the cervical cord w56,99,110,123,132,151,221x; and rostral DMFC, which has been associated with the eyes and head, projects as far as the brainstem w108,132,142,187,256,257x. Hence, the difference in cell size across the DMFC accords with its general somatotopy. 2.1.4. ConnectiÕity of monkey DMFC As already alluded by the foregoing, the DMFC is innervated by both oculomotor as well as skeletomotor regions of the brain. It innervates the FEF and the superior colliculus w5,67,108,128,132,231,257x, both of which are major channels to the saccade generator in the brainstem. Also, it receives robust efferent and afferent projections from the motor cortex w56,79,129,140,146,150,160x and sends fibres to a myriad of brainstem w108,132,142,187, 256,257x and spinal cord w56,99,110,123,132,151,221x nuclei involved in the execution of eye, head, and limb movements. In addition, visual centers, such as the lateral intraparietal area and the medial superior temporal area, innervate the DMFC w108,232x, and somatosensory areas, such as the superior parietal lobule, send robust projections to it w113,202x. Moreover, DMFC is reciprocally connected to prefrontal cortex w108x and innervates various thalamic and striatal sites that have been implicated in oculomotor and 3 Caudal: F2 and F3s 2162 and 2157 mm, respectively. Rostral: F6 and F7s1990 and 1992 mm, respectively. 418 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Fig. 3. Monkey FEF. Anterior is to the right for panels A and C, and to the left for panel B. ŽA. Location of the FEF on a lateral view of monkey frontal lobe, defined using the - 50 mA current-threshold criterion Žmodified from Bruce et al. w31x.. Small arrows and shading delimit the mediolateral range of FEF. Most of the FEF, however, extends into the arcuate sulcus and cannot be seen. CS, central sulcus; AS, arcuate sulcus. ŽB. Monkey cortex as parcellated by Brodmann w29x. Species is presumably Cercopithecus campbelli Žsee von Bonin and Bailey w297x, p. 64.. ŽC. Parcellation of prefrontal cortex by Walker w301x. Species is M. mulatta. ŽD. Coronal section through the genu of the arcuate sulcus, showing the parcellation of von Bonin and Bailey w297x from mediodorsal Žtop. to ventrolateral Žright.. Species is M. mulatta; as, superior branch of arcuate sulcus; ai, inferior branch of arcuate. skeletomotor functions w108,125,128,158,187,258x. Finally, this region sends projections to the red nucleus w108,257,289x. 2.1.5. Human DMFC As in monkeys, the DMFC in humans is located immediately anterior to the motor cortex on the dorsal surface of the hemisphere ŽFig. 1C,D.. The area is situated anterior to the precentral sulcus and is bounded laterally by the superior frontal sulcus and medioventrally by the cingulate cortex w32x. Brodmann w28x has defined this region as mesial area 6 of the agranular cortex and Campbell w32x called it the intermediate precentral area. Vogt and Vogt w295x subsequently divided the region into caudal and rostral zones Ži.e., 6aa and 6ab . similar to what they had done in the monkey. Electrical stimulation of this region in humans evokes eye movements from rostral sites and hindleg movements from caudal sites w194,310x. This topographic layout is similar to that observed for the DMFC of monkeys. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 2.2. The anatomy of the FEF 2.2.1. Monkey FEF There has been general agreement among electrophysiological studies regarding the gross location of the FEF in the monkey. Ferrier w59x showed that electrical stimulation of the prearcuate gyrus causes contraversive eye movements ŽFig. 1A.. This central observation has been repeatedly confirmed w262x. The subsequent use of microelectrodes facilitated the establishment of the exact location and extent of the FEF. Bruce and Goldberg w30x functionally defined the FEF as the region from which saccadic eye movements can be evoked with currents of less than 50 mA. This region lies within the rostral bank of the posterior curve of the arcuate, extending anteriorly over the lip of the arcuate ‘‘ . . . a few millimeters onto the surface of the prearcuate gyrus’’ ŽRef. w31x, p. 723., and posteriorly encroaching a few millimeters into the caudal bank w84,85,248x. Mediolaterally, it extends about 10 mm, centered approximately on the arcuate’s midpoint as defined by the intersection of the straight extension of the principal sulcus with the arcuate ŽFig. 3A.. The overall area of the FEF, approximately 100 mm2 , is about equal to the area of the DMFC. The functionally defined FEF does not correspond to any of the classical cytoarchitectural regions defined by anatomists of the 20th century. Brodmann’s area 8 w29x is close with respect to its medial and lateral extent ŽFig. 3B., but is situated too far rostrally, extending anteriorly to the caudal tip of the principal sulcus and posteriorly only to the lip of the arcuate sulcus Žas described by Walker w301x.. The functional FEF therefore overlaps with both areas 8 and 6 of Brodmann. Walker’s area 8A ŽFig. 3C. extends 1r2 to 2r3 of the way down from the lip of the arcuate and hence, like Brodmann’s area 8, also fails to cover the full caudal extent of the FEF. Laterally, Walker picked out another region in the arcuate’s rostral bank and called it area 45 ŽFig. 3C., but how far this extends into the sulcus was not specified. Thus, in terms of Walker’s nomenclature, the functional FEF overlaps with areas 8A and 45 Žand encroaches caudally into Brodmann’s area 6.. von Bonin and Bailey w297x included the medial half of the arcuate’s rostral bank in their frontal granular region ŽFD., but they agreed with Walker that the lateral part differed and called it FD gamma Žafter the notation of von Economo and Koskinas w299x; Fig. 3D.. Other investigators have split the cortex comprising the FEF into even more subfields, upon which we will not elaborate w203,211,295x. Although the functionally defined FEF does not coincide neatly with traditionally defined cytoarchitectonic regions, it does have a distinct, constant anatomical feature. Stanton et al. w267x reported that the functionally defined FEF coincides with a relatively high concentration of large layer V pyramidal cells in the arcuate’s rostral bank. This feature is so prominent that Stanton et al. w267x claimed it to be macroscopically evident in tangential sections through 419 the prearcuate gyrus. The layer V pyramidal cells are thought to be the major conveyors of oculomotor signals from the FEF to the superior colliculus and the pons w247,248,267x. Does any anatomical characteristic of tissue change systematically across the FEF? Successive generations of anatomists have disagreed about which microstructural changes in the FEF are reliable and important. The following changes are worth noting because they have been independently confirmed by the majority of anatomists; 4 how they relate to function, however, is unknown. From rostral FEF Žnear the arcuate lip. to caudal Žnear the fundus., a moderately thick and granular layer IV tapers off, becoming almost invisible at the fundus and into the posterior bank, while radial fascicles become thicker. From medial FEF to lateral, layer III pyramidal cells get bigger and the distinct radial and horizontal myelination patterns that exist in medial FEF become more diffuse. The FEF makes profuse reciprocal connections w109x with the lateral intraparietal area, with area 46, with the contralateral FEF, and with the eye field in the DMFC. Subcortically, the FEF is reciprocally and heavily connected with the lateral sector of the mediodorsal nucleus of the thalamus and, to a lesser extent, with thalamic regions lateral and anterior to this w76,268x. The FEF projects directly to the superior colliculus and to brainstem regions fundamental to saccade generation w141x, and the FEF receives reciprocal input from the superior colliculus via a disynaptic pathway relayed by the thalamus w153x. The FEF also projects to the striatum w187,268x; this projection may be reciprocated through a polysynaptic loop to substantia nigra pars reticulata, thalamus, and back to FEF w3,153x. 2.2.2. Human FEF The anatomy of human FEF is an enigma. The most recent conclusions about the gross location of human FEF come from experiments using positron emission tomography ŽPET. imaging or functional magnetic resonance imaging ŽfMRI., and from these studies, it appears that the human FEF lies within the precentral sulcus w43,148,189x just caudal to the middle frontal gyrus. The human FEF seems to be placed surprisingly far back in the frontal lobe; it is within, or attached to, the precentral motor strip. In monkeys, by contrast, the FEF is distinctly rostral to the precentral motor strip. The human and monkey FEF are dissimilar in terms of their microstructures, as well. In monkeys, as already noted, a striking characteristic of the FEF is that, within it, layer IV changes rostrocaudally from being thick and granular to being nearly non-existent. In humans, however, the FEF lies entirely in agranular cortex ŽFig. 4A.. The major thalamic input to monkey FEF is the 4 To our knowledge, modern myelin staining has been performed in the FEF by only one laboratory w211x. Therefore, our myelination conclusions are based on a single report and one might consider them tentative. 420 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 offered a number of explanations for it w43,148,189x. We will return to this topic later in the review. 2.3. Summary Two distinct areas involved in eye-movement control have been discerned in the frontal lobe, the FEF and the eye fields in the DMFC. In the monkey, the FEF is located in a well-defined region of the arcuate sulcus. The FEF receives extensive projections from area 46, the intraparietal sulcus, the DMFC, and the contralateral FEF; layer V cells in the FEF project extensively to subcortical centers including the superior colliculus and the brainstem. The exact size and location of the eye field in the DMFC are not as clearly defined as in the FEF. However, it is evident that the size of cells increases antero-posteriorly in the DMFC and that the projections from the anterior regions terminate mostly in the brainstem while those from the posterior regions have projections that reach the spinal cord. The DMFC innervates regions that mediate both oculomotor and skeletomotor responses. The two eye fields have also been identified in the human. The FEF in the human is located more closely to the central sulcus than it is in the monkey. The inputs to these areas and their outputs are less well-known in humans than in monkeys. Fig. 4. Human FEF. Anterior is to the left and dorsal is to the top. Ellipses show the approximate locations of human FEF as determined by imaging studies. ŽA. Basic cytoarchitecture of human frontal lobe. Region 2 is the extent of granular frontal cortex; caudal to that, region 1, in white, is agranular frontal cortex Žthe granularity of regions caudal to the central sulcus ŽCS. is not considered.. Imaging studies place the human FEF in the precentral sulcus ŽPCS., totally in agranular cortex. Derived from Walker w301x, who based his figure on the human brain study of von Economo and Koskinas w299x. ŽB. Connections of the human brain with respect to thalamus. The ‘‘regio frontalis’’ is connected primarily with the mediodorsal ŽMD. nucleus of the thalamus; the region caudal to that is connected primarily with the ventrolateral ŽVL. thalamic nucleus. Modified from Bailey and von Bonin w7x. mediodorsal nucleus, as noted above. In humans, the general layout of thalamic projections to the cortex has been estimated w7x by analyzing retrograde degeneration subsequent to prefrontal leucotomy or lobectomy w65,165x and by extrapolating results from monkeys w300x. According to this mapping, it is doubtful that the human FEF receives substantial mediodorsal thalamus input ŽFig. 4B.. Rather, like its neighbor, the precentral motor strip, the human FEF appears to receive its primary afferents from more ventrolaterally located nuclei. The human FEF, as determined by imaging methods, therefore, is strikingly different from the monkey FEF. Imaging investigators recognize this conflict and have 3. Electrical stimulation of the DMFC and the FEF 3.1. Maps and coding schemes in monkeys In 1969, Robinson and Fuchs w216x showed that electrical stimulation of the FEF elicited fixed-vector saccadic eye movements such that the size and direction of the elicited saccades were largely invariant with the initial position of the eyes in orbit. They also noted an orderly arrangement of stimulation-elicited saccades in the FEF Žwhich was first alluded to by the work of Beevor and Horsley w10x.: in the ventrolateral regions, small-amplitude saccades were evoked, whereas stimulation of the dorsomedial regions yielded large-amplitude saccades ŽFig. 5A.. Robinson and Fuchs w216x also found that when long trains of stimulation were delivered to the FEF, a staircase of saccades was elicited with alternating saccades and fixations, so that each saccade had a similar vector ŽFig. 5B, right.. Bruce et al. w31x confirmed the FEF’s map of saccadic amplitudes, and also found that the direction of saccades varied as a function of the depth of stimulation in the arcuate sulcus. Numerous other studies have confirmed these basic findings w6,80,157,229,233,282,284x. Electrical stimulation of the DMFC yields notably different results from those obtained in the FEF. It has been known since 1987 w243x that saccades evoked by electrical stimulation from most sites within the DMFC tend to shift the center of gaze to a particular location of craniotopic E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Fig. 5. Coding schemes. ŽA. Cranio- and retinocentric coding schemes described for the DMFC and FEF, respectively, are shown. A side-view of the macaque monkey brain is depicted. Each box represents the visual space in front of a monkey with head fixed. The right side of a box represents contralateral craniotopic space and the left side represents ipsilateral craniotopic space Ži.e., with respect to the side of stimulation.. The arrows within each box represent saccades starting from different fixation positions. The top boxes show the types of saccades evoked from sites situated along the rostrocaudal axis of the DMFC. A shaded circle represents the termination zone of the saccades evoked from a given site. The boxes aligned at left show the types of saccades evoked from sites situated along the mediolateral axis of the FEF. ŽB. Horizontal eye traces are shown of saccades evoked from the DMFC and FEF. Below each eye trace is a black bar which represents the onset and duration of electrical stimulation. ŽTop. When a 400-ms train of stimulation was delivered to the DMFC, a single saccadic eye movement was evoked and when such a train of stimulation was delivered to the FEF, a series of saccadic eye movements was evoked. ŽBottom. When a 70-ms train of stimulation was delivered to the DMFC or FEF, a single saccadic eye movement was evoked. space. This finding has been confirmed by the present authors laboratory w139,156,264,282,283,285,286x as well as by others w19,229x. The orbital position reached by the eye upon stimulation has subsequently been named the termination zone w282x. The location of a termination zone corresponds to the location of the fixation fields of neurons in the stimulated area w19x. Further work revealed that the DMFC contains an orderly topographic map representing different termination zones w139,264,282,283,285,286x: stimulation of rostral sites in the DMFC elicits saccades that shift the direction of gaze into contralateral craniotopic space, whereas stimulation of caudal sites shifts gaze into central or ipsilateral craniotopic space ŽFig. 5A w139x.; stimulation of medial and lateral sites direct the eyes to lower and upper craniotopic space, respectively. When the head is displaced with respect to the trunk or 421 when the head is tilted with respect to the vertical axis, the location of a termination zone remains fixed to the head w285x. When long trains of stimulation are delivered to the DMFC, only one saccadic eye movement is typically produced ŽFig. 5B, left. w229,243,282,284x after which the eyes remain fixated in the termination zone for the duration of stimulation, thereby inhibiting any subsequent eye movements w282,283x. These results suggest that the DMFC, unlike the FEF, contains a craniocentric Žor headcentered. code for the execution of saccadic eye movements. In conflict with these reports is a study by Russo and Bruce w222x who found that stimulation in the DMFC produces saccadic eye movements similar to those found in the FEF: the size and direction of the saccadic eye movements did not vary remarkably with respect to the starting position of the eye. There could be several reasons for this difference. First, it is possible that area DMFC is not homogeneous; different subregions may carry different codes. Second, it is possible that stimulation at certain sites in the DMFC antidromically activated axon collaterals from the FEF. Consistent with this hypothesis, our experience has been ŽRef. w264x and unpublished observations. that fixed vectors can be evoked from the DMFC at about 4% of sites Ž N s 53 penetration., but only within narrowly defined depths; stimulation a few hundred microns above or below these sites again yields craniocentric saccades. Ablation or inactivation of the FEF during stimulation of the DMFC could test the hypothesis that fixed-vector saccades elicited from the DMFC occur by virtue of activating the collaterals of FEF neurons. The third possible reason that Russo and Bruce obtained what look like fixed-vector saccades is that they used only short-duration stimulation trains of 70 ms. Under such conditions, the saccades elicited from the DMFC often do not reach their full extent w284x. An example of this is shown in Fig. 6, demonstrating that the amplitude of stimulation-elicited saccades increases with increasing train duration. In the experiments of Russo and Bruce, low current levels were used which may have also contributed to the execution of truncated saccades. This, however, cannot be the entire explanation since others have managed to evoke convergent saccades from the DMFC using low currents w19,229,243x. For more details regarding these issues, see Schall w230x and Tehovnik w280,281x. 3.2. Electrical stimulation and the behaÕioural state of monkeys It has been shown in many experiments that the probability of evoking a saccadic eye movement with electrical brain stimulation is influenced by the behavioral state of an animal. Such factors as state of alertness and intent to fixate or to carry out a specific task can significantly alter the size, direction, or latency of saccadic eye movements or affect the current threshold at which saccades are 422 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 variables. These results, along with the finding that low currents Že.g. - 50 mA., short train durations Že.g., - 70 ms., and high pulse frequencies Že.g., ) 300 Hz. are more effective in evoking saccades from the FEF than from the DMFC, suggest that the FEF, compared to the DMFC, is more closely connected to the saccade generator in the brainstem w284,286x. Fig. 6. Effect of train duration on saccades evoked from the DMFC. The left hemifield, out to 108, is shown for a monkey. The animal fixated a target located in central craniotopic space. Electrical stimulation was delivered to one site in the DMFC to evoke saccades contralateral to the side of stimulation. The arrows point to the end points of the elicited saccades when using stimulation trains of 120, 160, and 200 ms. Notice that the amplitude of the saccades increased systematically with increases in train duration. produced w19,69,82,131,157,186,237,253,266x. A recent examination of the effectiveness of stimulation in the frontal lobes has shown that the behavioural state of animals has a much greater effect on saccadic eye movements evoked electrically from the DMFC than from the FEF w284x. Monkeys were required to fixate a visual target for 600 ms after which a juice reward was given. Electrical stimulation was delivered at various times during and after fixation. When stimulating the DMFC early during the fixation period, 16 times as much current ŽFig. 7A. was required to evoke saccades than when current was delivered after termination of the fixation spot ŽFig. 7C.. When the same manipulations were carried out in the FEF, only three times as much current ŽFig. 7B. was required to evoke saccades early during fixation as compared with stimulation after the fixation spot was turned off ŽFig. 7C.. This finding for the FEF concurs with the results of Goldberg et al. w82x. The probability of evoking saccades from the DMFC was also noticeably decreased when the duration of active fixation was decreased or when the delivery of juice reward was delayed; stimulation-elicited saccades from the FEF were much less affected by these Fig. 7. Current threshold for eliciting saccades on 70% of stimulation trials is plotted as a function of stimulation onset time for the ŽA. DMFC and ŽB. FEF. Curves ‘‘a’’ through ‘‘f’’ in ŽA. represent the current thresholds for evoking saccades from sites in the DMFC, and curves ‘‘a’’ through ‘‘h’’ in ŽB. represent the current thresholds for evoking saccades from sites in the FEF. Stimulation was delivered 200 ms after the termination of the fixation spot Ž200 ms., 0 ms after the termination of the fixation spot Ž0 ms., 200 ms before the termination of the fixation spot Žy200 ms., or 400 ms before the termination of the fixation spot Žy400 ms.. The left panel shows the method. The top grey bar represents the duration of fixation Žfix., which was 600 ms, and the black bars represent onset and duration of stimulation which was 200 ms. Juice was delivered immediately after the fixation spot was terminated as long as the animal remained fixated for the 600 ms. No juice was delivered if the animal broke fixation during this period, whether the break was self-initiated or stimulation-induced. ŽC. A normalized threshold ratio is plotted as a function of stimulation onset time for the DMFC and FEF. The ratio was computed by dividing the mean current required to evoke saccades at a given stimulation onset time by the mean current required to evoke saccades at a stimulation onset time of 200 ms. Standard errors are indicated. Pulse duration and pulse frequency were 0.2 ms and 200 Hz, respectively. Data from Tehovnik et al. w284x. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 3.3. Skeletomotor responses elicited by electrical stimulation of the DMFC and FEF in monkeys As indicated earlier, the DMFC, within which the supplementary motor area resides, contains a topographic map of the body. Stimulation of rostral sites tends to evoke eye movements, whereas stimulation of caudal sites tends to evoke hindlimb movements; stimulation of intermediate sites tends to evoke forelimb movements ŽFig. 2C.. Other responses that have been evoked from the DMFC include ear and neck movements, as well as eyelid blinking w20,243x. In contrast, skeletomotor responses are not readily triggered from the FEF w31,157,216,233,237,239x. 3.4. DMFC and FEF stimulation in humans It has been known for some time that electrical stimulation delivered to either the DMFC or the FEF in humans evokes eye movements w60x. Such stimulation of the human DMFC has disclosed a somatotopic map similar to that observed in the monkey w194,310x. These findings have recently been replicated w66,78x. Penfield and Welch w196x found that stimulation of some sites in the DMFC evoked a contralateral gaze shift that was accompanied by an arm movement coincident with the change in the direction of gaze Žrepresented by cartoon in supplementary motor area of Fig. 1D.. These findings suggest that the DMFC may be involved in hand–eye coordination and that forelimb and eye have a common termination position that is laid out in a topographic fashion in this structure w282x. Penfield and Welch w196x also found that stimulation delivered to the DMFC often inhibited the execution of movements. These findings are in consonance with the already noted finding that in the monkey, visually evoked saccades are inhibited when the eyes are already positioned in the termination zone w282,283x. 3.5. Summary Saccadic eye movements can be elicited with electrical stimulation from both the FEF and the DMFC. The nature of the saccadic eye movements produced, however, is quite different in the two areas. In the FEF, the saccadic eye movements elicited have constant vectors; the amplitude and direction of a saccade at each site stimulated are largely unaffected by initial eye position. Prolonged stimulation produces a staircase of equal-size saccades. There is a clear topographic order in the FEF, with large amplitude saccades represented in medial portions and small amplitude saccades represented in lateral portions of the anterior bank of the arcuate. In the eye fields of the DMFC, electrical stimulation at most sites elicits saccadic eye movements that bring the eyes to a specific orbital position. Prolonged stimulation holds the eye in place at that position. There is a topographic order in the DMFC eye field area representing 423 different terminal positions. At some sites in this region, one can also elicit eye movements with constant vectors. More posteriorly in the DMFC, electrical stimulation can produce skeletomotor responses. Such responses are somatotopically ordered. In general, eye movements can be produced more reliably and with lower currents in the FEF than in the DMFC. This is especially true when an animal is actively fixating. 4. Lesions and reversible inactivation of the DMFC and the FEF 4.1. The effects of lesions and reÕersible inactiÕation of the DMFC in monkeys Many groups have studied the effect of DMFC lesions on forelimb movement tasks Že.g., Refs. w25,26,35,147, 188,227,255,288,306x.. In 1984, Brinkman w26x made lesions of various portions of the DMFC confined to the mesial bank ŽFig. 8A.. The animals were tested on two tasks. In the first, a unimanual task, monkeys were required to remove small morsels of food embedded in slots cut into a board. The orientation of each slot was different, requiring the animal to orient his fingers appropriately to extract the food item. In the second task, a bimanual one, monkeys were required to use both hands to obtain the food morsels. One hand was needed to push the food item through a slot and the other to grab it. Shortly after unilateral DMFC lesions, monkeys were impaired on both of these tasks; they no longer adjusted the orientation of their hands to extract the food in the unimanual task. Both hands showed impairment. On the unimanual task, the deficit disappeared after 2 weeks. In contrast, the poor performance on the bimanual task persisted even 1 year after the lesion. Lucchetti et al. w147x found that following a lesion of the DMFC that included anterio-lateral regions ŽFig. 8B., a monkey was impaired at reaching toward visual objects presented in contralateral space. This deficit subsided after 1 month, however. In another set of experiments, Thaler et al. w288x and Chen et al. w35x made bilateral lesions of the DMFC that included both the mesial and lateral regions of the area ŽFig. 8C.. Monkeys were trained on several tasks. In the first, they had to displace one lever at the onset of a visual stimulus and then displace another lever upon the termination of the stimulus. In the second task, monkeys were trained to delay the movement of a joystick by having them activate the joystick 5–8 s after the onset of a visual stimulus. In the third task, the animals had to shift the joystick to the left and then forward after the visual stimulus appeared. In the fourth task, reversal learning was assessed by making reward contingent on repeatedly pressing the joystick to the left and then, after 27 trials, to the 424 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Fig. 8. DMFC lesions in monkeys. The method of Tehovnik w280x was used to determine the size and location of DMFC lesions across various studies. The rostrocaudal and mediolateral extents of a lesion were determined in millimeters and the location of a lesion was determined with respect to the cerebral midline ŽMl. and posterior tip of the arcuate sulcus ŽPa.. In each panel, the tick marks along the midline axis are spaced by 1 mm. ŽA. The overlapped rectangles show the location of the chronic lesions of monkeys used in the study of Brinkman w26x: monkeys SMX1, SMX2, M6, M7, M9. Lesions were all unilateral. ŽB. The rectangle shows the extent and location a chronic lesion of a monkey used by Lucchetti et al. w147x. The lesion was unilateral. ŽC. The overlapped rectangles show the location of the chronic lesions of monkeys used in the study of Thaler et al. w288x and Chen et al. w35x: monkeys 62, 63, 64, 65, 66, 67, 83, 84, 85. Lesions were bilateral. ŽD. The shaded rectangles represent the sites injected with muscimol in monkeys BT and PS of Shima and Tanji w255x. The dotted rectangle represents the maximal spread of the muscimol which was estimated to be 2.0 mm from the cannula tip w255x. All injections were bilateral. ŽE. The rectangle represents the location and size of a cooling probe used in the study of Sasaki and Gemba w227x. Cooling probes were situated bilaterally. ŽF. The shaded rectangles represent the sites injected with lidocaine in the study of Sommer and Tehovnik w264x. The anterior-most rectangle represents the injection sites of Monkey L and the posterior-most rectangle represents the injection sites of Monkey I. The dotted rectangle represents the maximal spread of the lidocaine. Each injection was estimated to spread 1.5 mm from the cannula tip w287x. All injections were unilateral. ŽG. The rectangle shows the extent and location of a typical chronic lesion in the study of Schiller and Chou w234x: monkey 3. Lesions were typically unilateral. right; the number of trials required for successful reversal was assessed. Damage to the DMFC had little effect on the performance of these tasks. The last two tasks did show a minor deficit, but continued training rapidly reinstated pre-operative performance. One additional task was used in which monkeys were required to break a photobeam with their forelimbs in order to obtain a reward. The animals were exposed to this task for 30 minrday. On this task, 3 weeks after a DMFC lesion, monkeys were still deficient. However, due to limited testing Žonly four occasions., these results are difficult to evaluate. Using reversible inactivation methods, Shima and Tanji w255x showed that the learning of a sequential task, which required animals to move a manipulandum to successive positions Žby a push, pull, or turn., was disrupted when muscimol was injected bilaterally into various regions of the DMFC ŽFig. 8D.. The severity of the deficit decreased with the repetition of a particular sequence, however. When the monkeys were required to execute the movements in the presence of visual cues, no deficit was observed following inactivation. In another reversible inactivation experiment in which a large portion of the DMFC was cooled both unilaterally and bilaterally ŽFig. 8E., Sasaki and Gemba w227x showed that performance was disrupted on a task that required the lifting of a lever when a visual stimulus appeared. They found that the latency with which the animals lifted the lever upon the appearance of the visual stimulus increased when tissue was cooled. The effect, however, was transitory; after 2 months of continued testing, the response latencies were no longer affected by cooling. When a similar experiment was performed on the motor cortex, the disruptive effect of cooling on lever lifting never faded w226x. Corroborating the amelioration of deficits with repeated cooling is a study by Miyashita et al. w168x, who have shown that after extensive training, inactivation of the DMFC becomes ineffective. Only a few studies have examined the effects of DMFC inactivation and lesions on oculomotor responses. Since these studies compared directly the effects of DMFC and FEF lesions or inactivation, they will be considered in a separate section below. 4.2. The effects of lesions and reÕersible inactiÕation of the FEF in monkeys The effects of FEF ablations and inactivation have been extensively studied. Investigators who have examined the execution of saccadic eye movements to singly appearing stationary visual targets report that after FEF ablation or inactivation, the deficits are moderate. There is a small decrease in saccadic velocities and an increase in saccadic latencies; this has been reported both with lesions and with reversible inactivation using lidocaine or muscimol w52, 54,234,238,240,263x. After FEF ablation, there is relatively rapid recovery in both saccadic velocities and saccadic latencies. Time for recovery is longer for saccades made to visual targets appearing at relatively large eccentricities Ž108–208. w235,236x. Reversible inactivation studies also E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 show that monkeys have difficulty in acquiring targets that appear beyond 158 of eccentricity from straight ahead and to then maintain fixation on them w263x. This deficit has its counterpart in the fact that there is a decrease in saccadic amplitude that is especially notable for large saccades following FEF ablations. These deficits recover at about the same rate as does the deficit in saccadic latency w235,236x. In contrast to the relatively mild and short-lasting deficits in the latency and amplitude of saccades made to visual targets and in maintained fixation, ablation or inactivation of the FEF ŽFigs. 9 and 10A,B,C,D. dramatically affects the execution of single saccades generated to remembered target positions. FEF lesions ŽFig. 10A,B,E. also interfere with two successive saccades made in response to two successively presented targets. Deficits in generating such ‘‘memory-guided saccades’’ persist even 4 months after an FEF ablation ŽFig. 10C.. The effects of FEF lesions ŽFig. 10F. on the execution of smooth pursuit eye movements elicited by moving targets have also been examined. Deficits in pursuit have been reported both with lesions and inactivation w152,251x. Pursuit deficits typically occur for targets moving in a direction ipsilateral to the side of the lesion. Deficits in pursuit are still apparent beyond 10 weeks after an FEF ablation w120x. To obtain clear deficits in visually guided pursuit, the deeper portions of the arcuate, including the fundus and portions of the posterior bank, must be ablated or inactivated w154x. The deficits in pursuit so obtained are in consonance with two related findings w84,85,154x: Ž1. there are neurons in the depth of arcuate sulcus, including Fig. 9. Size and location of FEF lesions in monkeys. ŽA. The size and location of an FEF lesion were determined by noting the extent of a lesion with respect to the superior ŽAs. and inferior ŽAi. limbs of the arcuate and with respect to an axis originating at the posterior tip of the arcuate ŽPa. and extending anteriorly toward the principle sulcus ŽPs.. The anterior Žant. and posterior Žpost. portions of the arcuate sulcus are also shown so that the depth of the lesion into each bank can be assessed. The shaded area is a hypothetical FEF lesion. ŽB. The length of each axis Ž Y, X, and Z . was set to 1. For the hypothetical lesion shown in ‘‘A’’, the length of the Y vector is 0.7, the X vector is 0.6, and the Z vector is 0.4. ŽC. By joining the endpoints of each vector Ždotted line in B. in combination with the borders of the lesion into the anterior and posterior bank, the size and location of an FEF lesion are described. 425 Fig. 10. FEF lesions in monkeys. The size and location of FEF lesions are shown for various studies investigating oculomotor and skeletomotor behaviour. The shaded area represents the lidocaine injection sites of ŽA. Monkey L and ŽB. Monkey I in Sommer and Tehovnik w263,264x. The dotted area represents the maximal effective spread of lidocaine. Each injection was estimated to spread 1.5 mm from the cannula tip w287x. All injections were unilateral. ŽC. The shaded area represents the extent of the chronic lesion in the monkey used by Deng et al. w52x. The lesion was unilateral. ŽD. The shaded area represents the chronic lesion of a monkey used by Funahashi et al. w70x: monkey YM. The lesion was unilateral. ŽE. The shaded area represents a typical chronic lesion in the study of Schiller and Chou w234x: monkey 3. The lesions were usually unilateral. ŽF. The overlapped dotted regions show the location of chronic lesions of monkeys used in various studies: Keating w120x: monkeys Big1, Big2, SML; Keating w121x: monkey S1, S2, S3; Keating et al. w122x: monkey 3; Lynch w152x: monkey R5; MacAvoy et al. w154x: monkey 420. ŽG. The shaded area represents the chronic lesion of a monkey used by Schiller et al. w240x: monkey Bn. The lesion was bilateral. ŽH. The overlapped dotted regions show the location of chronic lesions of monkeys used by van der Steen et al. w291x: monkeys F1, F2, F3. Lesions were unilateral. ŽI. The overlapped dotted regions show the location of chronic lesions of monkeys used by Halsband and Passingham w95x: monkeys FEF33, FEF34. Lesions were bilateral. For other details, see Fig. 9. the fundus, that are modulated during smooth pursuit; and Ž2. electrical stimulation of this region evokes smooth pursuit eye movements. A number of studies have also investigated the effect of FEF ablations on visually guided forelimb movements w46,95,214,240,291x. Schiller et al. w240x trained monkeys to pick apple pieces from slots imbedded in a board. The slots in the board spanned 608 = 608 of visual space. Immediately after a bilateral FEF lesion ŽFig. 10G., monkeys were impaired at retrieving apple pieces from the board; complete recovery was evident by the fourth postoperative week. van der Steen et al. w291x trained monkeys to press, within 1 s following its illumination, a button which was presented at one of 19 positions spaced at 58 intervals over a horizontal 908 arc in front of an animal. 426 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Two days after a unilateral FEF lesion ŽFig. 10H., monkeys failed to push the illuminated target that appeared anywhere contralateral to the side of the lesion. Thirty-five days after the lesion, monkeys were still impaired at contralateral locations beyond 308. Monkeys had difficulties in making large gaze shifts which were compensated by the animals making head movements of increased amplitude. In another set of experiments, Halsband and Passingham w95x trained monkeys on a conditional motor task. The animals had to push a sphere when a green light came on and a cube when the light turned orange. The position of the sphere and the cube was randomized. Following bilateral lesions of the FEF ŽFig. 10I., monkeys did very poorly and failed to relearn the task; they were exposed to 3500 trials over a 3-month period. Unoperated controls mastered the task within 1 month and 1200 trials. 4.3. Direct comparison of DMFC and FEF lesions and inactiÕation on Õisually guided eye moÕements Only two sets of experiments have compared the effects on eye movements of lesions or reversible inactivation of the DMFC and FEF in monkeys. In the first set of experiments, the DMFC or FEF was reversibly inactivated with lidocaine or muscimol ŽFigs. 8F and 10A,B w263,264x. while monkeys performed various eye movements. Inactivation of the DMFC did not interfere with the ability to fixate visual targets in different parts of visual space or to execute saccadic eye movements to single visual targets or to remembered target locations w264x. However, inactivation of the FEF, as already noted, had dramatic effects on these tasks; the most pronounced deficits were obtained under conditions when animals had to make saccadic eye movements to remembered target locations w263x. When monkeys were required to generate a sequence of two saccades to two remembered targets, both DMFC and FEF inactivation produced deficits w263,264x. Monkeys were often unable to generate saccades to either the first or the second target. The magnitude of the impairment was much greater after FEF inactivation than it was after DMFC inactivation. The second set of experiments compared eye-movement performance after FEF and DMFC ablations w234x and reported similar deficits to those found during reversible inactivation. DMFC lesions produced a mild impairment that recovered within weeks on the execution of successive saccadic eye movements made to sequentially presented targets. FEF lesions, on the other hand, produced a much more dramatic deficit on this task that lasted even after 2 years of continued testing. The areas lesioned are depicted in Figs. 8G and 10E. In addition to these tasks, the study comparing FEF and DMFC lesions used a test in which two identical targets were presented, with one appearing in each hemifield w234x. Monkeys were rewarded for choosing either target. This task was similar to the one used in clinical studies that explore the extinction phenomenon w46,124,147,214, 291,304x. In addition to having the targets appear simultaneously, they were also presented with various temporal onset asynchronies. This enabled the investigators to determine how choice of the targets shifted as a function of the temporal offset between them. In normal animals, the equal probability point at which the frequency of left and right target choices was similar occurred close to the time when they appeared simultaneously. However, after a unilateral DMFC or FEF lesion, a major shift arose in choice preference. The temporal shift was much greater and longer-lasting after an FEF lesion than after a DMFC lesion. To get equal probability choices for the left and right targets, a temporal asynchrony of more than 110 ms had to be introduced 2 weeks after removal of the FEF, with the target in the hemifield contralateral to the lesion having to appear first. The temporal offset for equal probability choice 2 weeks after a DMFC lesion was only 31 ms. Recovery, after the DMFC lesion, was complete after 4 months; after FEF lesions, even a year later, more than a 60-ms offset was necessary for equal probability choice. Fig. 11 shows data on this task. Fig. 11. Target selection. Data obtained from two monkeys are shown as obtained with paired targets presented with varied temporal asynchronies. Plotted is the percent of saccades made to the left target as a function of temporal offset between the paired targets. Data are shown at various times after recovery from a left FEF ŽLFEF. or right DMFC ŽRDMFC. lesion as well as pre-operatively Žpre-op.. Squares and solid lines depict data from the animal with the FEF lesion; circles and dotted lines depict data from the animal with the DMFC lesion. Records of saccadic eye movements made to paired targets with various temporal offsets in the intact animal appear as an inset. For each of the post-operative weeks plotted Žwk2–wk16., data were collected for five or six successive days. Data from Schiller and Chou w234x. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 One interpretation of these effects after unilateral ablation is that the lesions affect temporal discrimination during target selection. To test this, monkeys were trained to discriminate temporal differences in target onset w234x; they had to select the target in an array that came on various times prior to the other similar stimuli. The animals were rewarded only for making a saccadic eye movement to the target that had appeared first. This task revealed major deficits in making temporal discriminations after FEF lesions and mild but significant deficits after DMFC lesions. Thus, one major effect of FEF lesions, and to a lesser extent of DMFC lesions, is to impair temporal discrimination. One animal in this study was also examined after both the DMFC and the FEF had been removed. The deficits after such paired lesions were of about the same magnitude as single lesions to the FEF w235,236x. These lesions did not produce any deficits in smooth pursuit eye movements, probably because the FEF lesion did not include damage to the fundus and posterior bank of the arcuate. 4.4. The effects of DMFC and FEF damage in humans 4.4.1. Deficits on oculomotor tasks Effects of frontal lobe damage on the oculomotor behaviour of humans have been studied widely Že.g., Refs. w24,73,74,93,100,170,190,205–207,213x.. Unfortunately, the brain damage in such patients is usually not restricted to the FEF or DMFC; it often includes damage to several other regions. In considering the effects of damage to the DMFC and FEF, we focused on results obtained from patients whose lesions did not extend well beyond either the FEF or DMFC ŽFig. 12.. In the human, the FEF is generally considered to be in or near the precentral sulcus Že.g., Ref. w189x. and the DMFC straddles the midline with significant overlap into the mesial bank. Most of our FEF cases included damage to regions rostral to, but also including, the precentral sulcus. The findings may be summarized as follows. Ž1. Patients with lesions of either the FEF or DMFC could generate saccades as well as normal subjects to visual targets. The lesions are depicted in Fig. 13A. Ž2. Patients with a unilateral FEF lesion ŽFig. 13B, left. trained to make saccades to remembered target positions in total darkness Žtypically a 2-s delay. generated hypometric saccades to targets appearing contralateral to the side of the lesion; saccade reaction times were much longer than those exhibited by normal subjects. In contrast, DMFC patients ŽFig. 13B, right. were not at all impaired on this task. Ž3. Patients with a unilateral FEF lesion ŽFig. 13C, left. trained to generate saccades to a remembered target position in total darkness after head and body rotation showed no impairment. Similar tests after DMFC lesions ŽFig. 13C, right. revealed that patients undershot the location of 427 Fig. 12. Size and location of DMFC or FEF lesions in humans. Shown is a diagrammatic horizontal slice through the top portion of one hemisphere of the human cerebral cortex. To determine the location and size of a lesion centered on the dorsomedial frontal cortex ŽDMFC. or frontal eye field ŽFEF., the length and width of the cerebral cortex were set to 1.0. According to this scheme, the DMFC is situated between 0.5 and 0.8 units along the length dimension and between 0.0 and 0.3 units along the width dimension; the FEF is situated between 0.5 and 0.75 units along the length dimension and between 0.5 and 0.9 units along the width dimension. In Figs. 13 and 14, this measuring scheme was used to determine the borders and location of lesions confined to the FEF and DMFC of various studies. The prefrontal cortex ŽPFC., superior frontal sulcus ŽSFs., precentral sulcus ŽPCs., central sulcus ŽCs., and intraparietal sulcus ŽIPs. are indicated. the remembered target position for targets appearing both in the hemifield ipsilateral and contralateral to the lesion. Ž4. One patient with a unilateral FEF lesion ŽFig. 13D, left. showed deficits in making successive saccades to two remembered target positions in total darkness; on 20% of trials, saccades were executed in the wrong order. Another patient with a DMFC lesion tested in a similar manner showed error rates of less than 10%; this performance was comparable to that of normal control subjects ŽRef. w73x, case 1.. Ž5. Three of five DMFC patients ŽFig. 13D, right. tested on a three-target task failed to successfully generate saccades to the remembered target positions in the same order the targets were presented. These three patients were tested between 26 and 54 days following the lesion. Two of the five patients, tested between 38 and 84 days, did not exhibit any deficits. Ž6. Patients with unilateral FEF ŽFig. 13E, left. or DMFC ŽFig. 13E, right. lesion trained to generate ‘‘antisaccades’’ that forced them to shift their gaze into the hemifield opposite to where the target had appeared, performed on the task as well as did normal subjects. However, in patients with infarcts that included the prefrontal cortex, a significant deficit was observed on this task w207x. 428 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Fig. 13. DMFC or FEF lesions in humans performing oculomotor tasks. Shown are horizontal slices through the cerebral cortex. Within each slice, the grey area represents the size and location of many lesions combined from different patients. ŽA. Left plate shows lesions confined to the FEF of 19 patients from Braun et al. w24x Žcases 1 to 6., Pierrot-Deseilligny et al. w207x Žcases 1 to 10., and Rivaud et al. w213x Žcases 1 to 3.. Right plate shows lesions confined to the DMFC of 13 patients from Braun et al. w24x Žcases 13 to 15., Gaymard et al. w73x Žcases 1., and Pierrot-Deseilligny et al. w207x Žcases 1 to 9.. ŽB. Left plate shows lesions confined to the FEF of eight patients from Pierrot-Deseilligny et al. w205x Žcases 1 to 5. and Rivaud et al. w213x Žcases 1 to 3.. Right plate shows lesions confined to the DMFC of seven patients from Gaymard et al. w74x Žcases 1, 3, 4, and 5. and Pierrot-Deseilligny et al. w206x Žcases 1 to 3, with right-sided lesion.. ŽC. Left plate shows lesions confined to the FEF of five patients from Pierrot-Deseilligny et al. w205x Žcases 1 to 5.. Right plate shows lesions confined to the DMFC of three patients of Pierrot-Deseilligny et al. w205x Žcases 1 to 3.. ŽD. Left plate shows lesions confined to the FEF of one patient from Rivaud et al. w213x Žcase not specified.. Right plate shows lesions confined to the DMFC of five patients from Gaymard et al. w73x Žcase 1. and Gaymard et al. w74x Žcases 1, 3, 4, and 5.. ŽE. Left plate shows lesions confined to the FEF of 13 patients from Pierrot-Deseilligny et al. w207x Žcases 1 to 10. and Rivaud et al. w213x Žcases 1 to 3.. Right plate shows lesions confined to the DMFC of 10 patients from Gaymard et al. w73x Žcase 1. and Pierrot-Deseilligny et al. w207x Žcases 1 to 9.. ŽF. The plate shows lesions confined to the FEF of six patients from Morrow et al. w170x Žcases 1 to 3. and Rivaud et al. w213x Žcases 1 to 3.. See Fig. 12 for other details. mally, under such conditions after the hand releases the load, the forelimb exhibits a slight flexion response. After a DMFC patient’s hand released the load, a larger flexion response than normal was observed, suggesting that the DMFC in humans is involved in bimanual postural adjustments. Halsband and Freund w94x trained patients with frontal lobe damage on a task wherein a particular visual stimulus had to be associated with a specific forelimb movement. At the onset of a stimulus, a patient was required to produce a correct movement Že.g., after stimulus onset, a patient had to put the right hand on the right shoulder unfolding it through about 908 downward.. Of the patients studied, two had lesions confined to the DMFC ŽFig. 14B. and one had a lesion confined to the FEF ŽFig. 14C.. All the patients had a deficit in learning the association task but had no difficulty imitating forelimb movements by visual example or from memory; nor did they have problems in evoking visually guided reaching movements toward different positions in visual space. 4.5. Summary The nature of deficits induced by ablation of the DMFC and FEF is quite different both in kind and in magnitude. After DMFC lesions, deficits are common both in limb and eye movement control with the latter quite mild, whereas after FEF lesions, there are no deficits in limb movements but impairment in some aspects of eye-movement control are sizeable and long-lasting. Such sizeable and long-lasting deficits following FEF ablation have been observed for the selection of two simultaneously presented targets, for the execution of sequences of eye movements to successive targets, and for eye movements made to briefly presented stimuli. DMFC lesions produced only mild deficits on these tasks that recovered within a few months. Lesions of the posterior bank and fundus of the arcuate, but not of the DMFC, produce deficits in pursuit eye movements. The impairment observed in limb movements after DMFC lesions is most pronounced when a bimanual task is employed. The deficits documented in the FEF and DMFC Ž7. Patients with unilateral FEF lesions ŽFig. 13F. tested on smooth pursuit were all impaired for ipsilaterally directed motion relative to the lesion but not for contralaterally directed motion. 4.4.2. Deficits on forelimb moÕement tasks Few studies have looked at the effect of DMFC or FEF damage on the execution of forelimb movements in patients Že.g., Refs. w94,293x.. Viallet et al. w293x trained patients with DMFC lesions ŽFig. 14A. on a bimanual forelimb movement task. At the onset of a tone burst, patients were required to remove a weight from the forelimb contralateral to the lesion with the other hand. Nor- Fig. 14. DMFC or FEF lesions in humans performing skeletomotor tasks. Shown are horizontal slices through the cerebral cortex. Within each slice, the grey area represents the size and location of many lesions combined from different patients. ŽA. Lesions confined to the DMFC of four patients from Viallet et al. w293x Žcases 1, 3, 4, and 5.. ŽB. Lesions confined to the DMFC of two patients from Halsband and Freund w94x Žcases 1 and 9.. ŽC. Lesion confined to the FEF of one patient from Halsband and Freund w94x Žcase 5.. See Fig. 12 for other details. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 429 upon reversible inactivation are similar to those found with lesions. Human studies for the most part are consistent with the findings reported in monkeys: lesions of the FEF or DMFC do not disrupt visually guided saccades; lesions of the FEF, but not the DMFC, impair saccades made to one remembered target position; lesions of the FEF or DMFC affect saccades generated to two or more remembered target positions; lesions of the FEF interfere with smooth pursuit eye movements; and finally, lesions of the DMFC disrupt bimanual movements. 5. Single-cell recordings in the DMFC and FEF 5.1. The characteristics of single neurons in the DMFC 5.1.1. Eye and forelimb moÕements Accumulating evidence suggests that neurons throughout the full extent of the DMFC are modulated by forelimb movements ŽFig. 15A.. Limb movements that are associated with neuronal activity include the displacement of a manipulandum, reaching toward and pressing buttons, and wrist extension or flexion. Unfortunately, many studies examining limb movements have not recorded and studied the eye movements that are an integral part of coordinated limb movements. Since Schlag and Schlag-Rey w242x have shown that the anterior DMFC Žanterior to the posterior tip of the arcuate. contains neurons that respond in association with eye movements, it is possible that the neuronal responses interpreted to occur with limb movements may be, at least in part, due to eye movements or the combined action involved in hand–eye coordination ŽFig. 15B.. Cells that respond in association with eye movements typically have been shown to be located in anterior DMFC, but a few studies have found such cells further back, in regions a few millimeters caudal to the posterior tip of the arcuate ŽFig. 15B.. Some studies have measured both eye and forelimb movements while recording from the DMFC w18–20,156,173,228x. Of these, only Mushiake et al. w173x have dissociated the contribution of eye movement from the contribution of forelimb movement. In this study, a monkey was trained to reach to and look at a visual target. Units were examined under three conditions: Ž1. execution of eye movements in the absence of limb movements; Ž2. execution of limb movements in the absence of eye movements; and Ž3. execution of both movements together. Of 12 cells studied in anterior DMFC, all exhibited their best response when the eyes and forelimb movements occurred together. Chou and Schiller w37,38x used a paradigm similar to that of Mushiake et al. w173x while recording from hundreds of units located within the full rostrocaudal extent of the DMFC. They found that the vast majority of cells responded under the above noted three conditions, albeit to different degrees. These findings suggest that most neurons in this area are not dedicated to the execu- Fig. 15. Location of unit recording sites in monkeys. ŽA. Top view of one side of the DMFC showing regions from which units were recorded by investigators who studied monkeys as they performed a forelimb movement task. The cerebral midline is represented by the horizontal line. Each tick mark on this line is spaced by 1 mm. The arcuate sulcus ŽAs. and central sulcus ŽCs. are indicated. The posterior tip of the arcuate sulcus ŽPa. is shown. Each oval represents the size and location of the region from which units modulated by a given forelimb movement task were found. The length and width of an oval and its center were determined using the method of Tehovnik w280x. Studies pertaining to a particular recording zone are listed. ŽAlso see Ref. w130x.. ŽB. Top view of one side of the DMFC showing regions from which units were recorded by investigators who studied monkeys as they performed an oculomotor task. tion of a single, specific motor act. These neurons may in fact be involved in hand–eye coordination. 5.1.2. Eye moÕements Neurons within the DMFC are modulated by saccadic and pursuit eye movements w19,101,105,139,156,223, 228,241–243x and discharge during the presentation of visual stimuli w223,228,243x. A large proportion of cells within the anterior DMFC also respond vigorously when the animal engages in active fixation w17,19,21,139,180, 244x. Neurons at rostral sites respond best when the center of gaze is directed toward contralateral craniotopic space, whereas neurons at caudal sites respond best when gaze is directed toward ipsilateral craniotopic space w139x. At lat- 430 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 eral sites, responses are best for upward gaze and at medial sites, for downward gaze. Recently, the rostrocaudal topographic order has been alluded to by the work of Bon and Lucchetti w21x. Here, a monkey was required to remain fixated and to displace a lever to detect the color change of a peripherally located visual target. Cells in rostral DMFC tended to respond to such target changes occurring in contralateral craniotopic space, whereas cells in caudal DMFC tended to respond to such changes occurring in ipsilateral craniotopic space. A population of neurons in rostral DMFC also seems to be modulated differentially according to what side of an object a monkey generates an eye movement w180,181x. Thus, DMFC neurons, especially those in anterior DMFC, discharge during saccadic eye movements, during smooth pursuit eye movements, during active fixation, and during the onset of visual stimuli. The region contains a topographic map for the representation of various gaze positions. 5.1.3. Anterior Õs. posterior DMFC Considerable evidence has been presented suggesting that the anterior and posterior regions of the DMFC are functionally distinct w69,161,162,173,254,274,277,279x. Matsuzaka et al. w161x found that neurons in the anterior DMFC are modulated differently from neurons in the posterior DMFC. In their experiment, monkeys faced a panel containing two switches. A light over either the left or right switch was illuminated briefly. A visual trigger then signalled the monkey to press the illuminated switch. Compared to cells in the anterior DMFC, cells located more caudally discharged best immediately before, during, and after the execution of the forelimb movement. In contrast, cells in anterior DMFC were more vigorously modulated when the light was on as well as during the period between the termination of the light and the onset of the target that triggered the forelimb movement. That cells in anterior DMFC are more readily modulated by visual stimuli and respond less at the time of the forelimb movement should not be surprising, since the region contains many cells that discharge to the presentation of visual stimuli w223,228,243x. When tactile rather than visual stimuli were used to initiate forelimb movement, no difference was observed in the modulation of cells in anterior and posterior DMFC w220x. Thus, to reveal a difference between the anterior and posterior regions of the DMFC, a visual task must be employed. Tanji and Shima w279x found that 54 out of 206 units located in posterior DMFC were modulated by a specific sequence of three movements Žpush–pull–turn. of a manipulandum guided by visual memory. Mushiake et al. w174x found that a small proportion of cells Ž21r328. located throughout the DMFC was modulated by a specific sequence of button presses. With the push–pull–turn paradigm of Tanji and Shima w279x, a larger proportion of cells in anterior DMFC Ž64r251., as compared to posterior DMFC Ž6r385., exhibited a diminution of responsivity during the continued execution of a movement sequence under visual guidance w254x. At this time, it is not known whether the sequencing-specific properties of cells in posterior DMFC would also diminish after prolonged task repetition. This question arises because it has been demonstrated that cells in posterior DMFC become quite unresponsive after overtraining on a simple key-press task w1x. One of the problems with the studies just described is that in none of them were eye movements under continuous measurement. This is of concern because the pattern of saccadic eye movements changes when animals learn a task that requires forelimb movements w167x. Another problem is that these studies have not mapped the receptive fields 5 or fixation fields of neurons before they were tested on any sequencing task. Issues of overtraining, eye movement control, and visual receptive field characteristics will need to be addressed before one can fully accept claims that go beyond anterior DMFC having a visual bias and posterior DMFC having a skeletomotor bias w161,162x. 5.2. The characteristics of single neurons in the FEF In 1968, Bizzi w14x found that neurons in the FEF respond during fixation and during and after the execution of saccadic eye movements. Subsequently, it was shown that cells in FEF also discharge in response to visual stimuli and before saccadic eye movements w15,30, 81,169,271x. Some cells in this region are also responsive during active fixation w270x. In addition, cells have been discovered in deeper portions of the bank of the arcuate that are modulated during ipsilaterally directed pursuit w154x. Finally, the FEF appears to be topographically organized such that neurons in dorsomedial sites discharge best for large-amplitude saccades, whereas neurons in ventrolateral sites discharge best for small-amplitude saccades w31x. Identifying neurons by antidromic activation, Segraves and Goldberg w248x described the basic properties of FEF cells that project to the superior colliculus. Using similar methods, Segraves w247x also characterized the FEF neurons that project to the pons. Both projections were found to relay primarily saccadic and fixation-related discharges, and they rarely had only visual responses. Recently, Sommer and Wurtz w265x presented the first description of FEF neurons that may receive input from the superior colliculus. Some FEF neurons could be activated via synapses by electrical stimulation in the superior colliculus, presumably driven via a tectothalamocortical route w153x. These FEF input neurons all had visual responses, and half had presaccadic bursts as well. Taken together with the results of 5 Tolias et al. w290x describes a method to map the entire receptive field of cortical cells within the context of the behavioural paradigm, a method that could be employed when studying sequencing behaviour in visual areas. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Segraves and Goldberg w248x, this provides the first evidence that FEF receives visual signals at its input and produces saccade commands at its output Žas predicted by Bruce and Goldberg w30x.. Segraves and Park w249x determined that movement-related presaccadic FEF neurons begin their discharge approximately 150 ms before saccadic onset and show peak activity approximately 13 ms before the onset of the saccade. These neurons can have multiple phases of activity: prelude activation and a burst that occurs later. Hanes et al. w96x performed an analysis of activity in the FEF and DMFC to determine the onset time of neural activity. For presaccadic neurons in both the FEF and DMFC, the onset time of the burst was more tightly coupled to the beginning of the saccade, as opposed to the time that the instructional trigger signal was presented. On average, FEF neurons began bursting 28 ms ŽS.D.s 3.3 ms. before saccades. Presaccadic neurons in the DMFC, on the other hand, started to burst earlier than FEF neurons, and they showed greater variance in burst time Žmean s 135, S.D.s 6.6 ms.. Also the prelude activity in the DMFC occurred earlier and with a greater variance in onset time than it did in the FEF. Lee w138x trained monkeys to generate saccades to visual targets presented within a region of 608 = 408 of visual space. The monkeys were required to maintain fixation for 1000 ms after which they generated a saccade to a randomly selected target position. It was found that a higher percentage of cells in the DMFC than FEF was modulated during active fixation but that a higher percentage of cells in the FEF than DMFC was modulated during saccade execution. Also unlike cells in the DMFC that are modulated during both eye and forelimb movements w18, 37,38,156,173x, cells in the FEF are modulated exclusively during eye movements w173x. 431 5.3. Summary Neuronal responses associated with eye and limb movements are common in the DMFC and many single cells respond to both, indicating that these cells do not mediate a specific motor act. Cells in anterior portions of the DMFC tend to be more responsive to visual stimuli and the execution of saccadic eye movements whereas in posterior regions, cells are more responsive to limb movements. Cells that discharge during fixation and smooth pursuit eye movements have also been reported. Fig. 16. Imaging studies and the DMFC and FEF. ŽA. Shown is a top view of the human cerebral cortex. The box depicts the region from which studies located maximal-activity points measured with PET or fMRI while subjects executed saccadic eye movements or smooth pursuit eye movements. The approximate location of the DMFC and FEF is represented by the ellipses. The central sulcus ŽCs., precentral sulcus ŽPCs., and superior frontal sulcus ŽSFs. are indicated. ŽB. The graph shows the location of maximal-activity points in Brodmann’s area 6 observed with PET or fMRI while human subjects performed tasks involving the generation of saccadic or smooth pursuit eye movements. Each dot within the figure represents a maximal-activity point plotted in Talairach and Tournoux w273x coordinates along the caudorostral and mediolateral axes. A dot is based on an average from 5 to 12 subjects. The subtraction method was used to determine a maximal-activity point. Typically, the baseline behaviour used for the subtraction method was fixation of a visual target, or resting with eyes open or closed. Data were obtained from the following studies: Anderson et al. w4x, Doricchi et al. w55x, Kawashima et al. w119x, Luna et al. w148x, O’Driscoll et al. w178x, Paus et al. w191,192x, Petit et al. w197,198x, and Sweeney et al. w272x. The ellipses represent the distribution of maximal-activity points along the caudorostral and mediolateral axes. The DMFC and FEF were divided according Paus w189x and Roland and Zilles w219x. ŽC. The graph shows the location of maximal-activity points in the DMFC of Brodmann’s area 6 while human subjects performed forelimb movement tasks that did not engage the eyes. Movements could involve displacement of the shoulder, elbow, hand, or fingers or some combination of each. A dot is based on an average of 6–16 subjects. Typically, the baseline behaviour used for the subtraction method was the total immobility of the body; often this immobility was conducted in total darkness with the eyes closed. Data were obtained from the following studies: Colebatch et al. w41x, Deiber et al. w51x, Dettmers et al. w53x, Jahanshahi et al. w111x, Jenkins et al. w112x, Jueptner et al. w115,116x, Paus et al. w192x, Playford et al. w209x, Schlaug et al. w245x, Stephan et al. w269x, and van Mier et al. w292x. The ellipse represents the distribution of maximal-activity points along the caudorostral and mediolateral axes. Activation in the FEF ellipse is presumably from the arm area of the motor and premotor cortical areas. See B for other details. ŽD. The graph shows the location of maximal-activity points in the DMFC of Brodmann’s area 6 while humans subjects performed forelimb movement tasks that engaged the eyes. The movements could involve a key press, pointing movements, or movements of a manipulandum whose displacement was represented visually on a monitor. A dot is based on an average of 6–24 subjects. Typically, the baseline behaviour used for the subtraction method was fixation of a visual target. Data were obtained from the following studies: Corbetta et al. w42x, Grafton et al. w87–90x, Kawashima et al. w117,118x, Paus et al. w192x, Sakai et al. w224x, and Sergent et al. w250x. The ellipse represents the distribution of maximal-activity points along the caudorostral and mediolateral axes. See B for other details. 432 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 In the FEF, neurons have not been found that respond specifically to skeletal movement. Several populations of cells have been discerned: some have visual receptive fields, some discharge before, during and after saccadic eye movements, and some respond in association with fixation as well as smooth pursuit eye movements. The saccade-associated responses of many cells are tightly time-locked to saccade execution, suggesting that these cells contribute centrally to the initiation of eye movements. For both the DMFC and FEF, recording studies support the topographic layout of saccadic eye movements as revealed by electrical stimulation. 6. Event-related potentials in humans When human subjects make self-initiated finger or hand movements, a DC shift in cortical potential can be recorded from scalp electrodes placed over the frontal lobes prior to these movements. This potential was first termed ‘‘bereitschaftpotential’’, or ‘‘readiness potential’’ by Kornhuber and Deecke w127x. It emanates primarily from rostromedial recording locations in the frontal lobe Želectroencephalogram ŽEEG. electrode locations Cz, C3, and C4. and is evident as a slow increase in surface negativity before self-initiated movements. The readiness potential has been divided into different phases w50,252x. The first component is associated with a slow bilateral buildup of negativity. This buildup generally occurs up to 1–2 s before the initiation of a voluntary movement as measured by the onset of EMG activity, and is most prominent over the midline. Approximately 400 ms prior to EMG onset, the negative potential increases in slope and becomes more pronounced in the hemisphere contralateral to the hand being used w49x. This marks the second component. Based on differences in the location of their maxima, the two components were posited to emanate from different cortical areas w49x. Since the first component was most prominent over the vertex, and was bilaterally distributed, it was proposed that its source was bilateral activation of the DMFC. The second component was centered more caudally and contralateral to the hand being used. This corresponded roughly with the hand region of motor cortex. Thus, it was proposed that the second component has its source primarily in contralateral motor cortex. This hypothesis of bilateral DMFC activation followed by contralateral primary motor cortex activity was consistent with single-unit recording evidence showing that DMFC, but not motor cortex, was organized bilaterally w278x, and that it was activated earlier than was motor cortex w2,276x. The DMFC motor cortex hypothesis was challenged in humans, however, by the failure to localize the early component to the DMFC. Modeling the overall potential by fitting different possible dipole sources, some studies did not find a stable solution with DMFC as the exclusive source of the early component w22x. An alternate hypothesis was formulated that attributed both components to the Fig. 17. Eye movements vs. forelimb movements and DMFC activity. The frequency of maximal-activity points for a given location in the DMFC along the caudorostral ŽA., mediolateral ŽB., and ventrodorsal ŽC. axes is shown for imaging experiments that studied eye movements ŽEye. and forelimb movements ŽLimb.. The data for the eye movement and forelimb movement studies are based on the data in Fig. 16B and C, respectively. Values along each ordinate are expressed in Talairach and Tournoux w273x coordinates. Statistical tests were conducted to determine whether the center of the eye field differed from the center of the forelimb field. Along the caudorostral axis, the center of the eye field was anterior to the center of the forelimb field Ž t Ž27. s 3.2, p - 0.01.; along the mediolateral axis, the center of the eye field was not different from the center of the forelimb field Ž t Ž27. s 0.006, p ) 0.05.; along the ventrodorsal axis, the center of the eye field was ventral to the center of the forelimb field Ž t Ž27. s 3.1, p - 0.01.. See Fig. 16 for other details. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 motor cortex, with early bilateral activation followed by exclusively contralateral activation. Other studies using different recording techniques have also failed to observe consistent early activation of the DMFC. For example, Neshige et al. w176x found a clear signal in motor cortex just prior to movement, but found only weak evidence for early DMFC activity using subdural electrodes in epileptic patients. Researchers have also failed to observe the readiness potential over the DMFC using magnetoencephalography ŽMEG.. Countering these claims is a report by Cheyne and Weinberg w36x, who suggested that these negative findings were a product of the recording technique of MEG; they proposed instead that symmetric bilateral activation of both DMFCs would lead to mutually canceling dipoles. Lang et al. w134x tested this hypothesis by recording from a patient with a unilateral DMFC lesion and found that a dipole was indeed consistently localized over the intact DMFC. Several studies have shown activation of DMFC as well as motor cortex during self-paced movements using PET w155,218x but the poor temporal resolution of this technique does not make it possible to distinguish between early and late components of activation. Thus, the evidence is mixed regarding the role of the DMFC in the generation of the readiness potential. If the DMFC is the source of the readiness potential, the potential should be modulated by tasks that involve the DMFC to various degrees. Functions that have been at- Fig. 18. Eye movements vs. eye and forelimb movements and DMFC activity. The frequency of maximal-activity points for a given location in the DMFC along the caudorostral axis is shown for the imaging experiments that studied limb movements ŽLimb. and limb and eye movements ŽLimb and Eye.. The data for Limb and Limb and Eye are based on the data in Fig. 16C and D, respectively. Values along each ordinate are expressed in Talairach and Tournoux w273x coordinates. A statistical test was conducted to determine whether the center of the eye and forelimb field differed from the center of the forelimb field. Along the caudorostral axis, the center of the eye and forelimb field was anterior to the center of the forelimb field Ž t Ž27. s 3.5, p- 0.01.. See Fig. 16 for other details. 433 Fig. 19. Simple vs. complex tasks and DMFC activity. The frequency of maximal-activity points for a given location in the DMFC along the rostrocaudal axis is shown for the imaging experiments that studied eye movements ŽEye. and forelimb movements ŽLimb., sorted according to whether the movements were simple Žsimple. or complex Žcomplex.. The data for Eye and Limb are based on the data in Fig. 16B and C, respectively. For the eye movement experiments, simple movements included self-paced saccades, visually guided saccades, or visually guided pursuit, and complex movements included memory-guided saccades, anti-saccades, or conditional saccades Že.g., visual objectrsaccade direction associations or gornogo saccade tasks.. For the forelimb movement experiments, simple movements included tactile- or auditory-triggered finger, hand, or shoulder movements, movements of a joy stick, or self-paced forelimb movements, and complex movements included antifinger flexion movements Že.g., to move the finger next to the one stimulated., sequential movements of the fingers or joy stick, or conditional forelimb movements Že.g., to move a particular finger according to the auditory tone delivered.. Only data from experiments in which subject were over trained are included. Values along each ordinate are expressed in Talairach and Tournoux w273x coordinates. Statistical tests were conducted to determine whether the center of the eye and forelimb fields differed depending on whether the task performed was simple or complex. The center of the eye field for simple and complex tasks did not differ Ž t Ž9. s1.3, p) 0.05.. Also, the center of the forelimb field for simple and complex tasks did not differ Ž t Ž15. s1.64, p) 0.05.. See Fig. 16 for other details. tributed to the DMFC are sequencing of multiple movements and also the learning of motor tasks. The magnitude of the readiness potential does seem to reflect the complexity of the task being performed and is greater prior to sequences of movements as opposed to single movements w48x. As previously discussed, one class of movements that the DMFC is thought to be specialized for is bimanual coordination. Lang et al. w136x showed that the magnitude of the readiness potential was not only greater when subjects performed a bimanual coordination task as compared to a unimanual one but it was also modulated by the difficulty of the task. They recorded from trained musicians performing both an easy and a difficult bimanual rhythm and found that the readiness potential had a greater magnitude when subjects performed the difficult task. 434 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Fig. 20. Eye movements vs. forelimb movements and FEF activity. The frequency of maximal-activity points for a given location in the FEF along the rostrocaudal ŽA., mediolateral ŽB., and ventrodorsal ŽC. axes is shown for imaging experiments that studied eye movements ŽEye. and forelimb movements ŽLimb.. The data for the eye movement and forelimb movement studies are based on the data in Fig. 16B and C, respectively. Values along each ordinate are expressed in Talairach and Tournoux w273x coordinates. Statistical tests were conducted to determine whether the center of the eye field differed from the center of the forelimb field. Along the caudorostral axis, the center of the eye field was anterior to the center of the forelimb field Ž t Ž30. s 2.6, p - 0.01.; along the mediolateral axis, the center of the eye field was lateral to the center of the forelimb field Ž t Ž30. s 4.3, p - 0.01.; along the ventrodorsal axis, the center of the eye field was ventral to the center of the forelimb field Ž t Ž30. s 4.9, p - 0.01.. See Fig. 16 for other details. The readiness potential is also modulated during motor learning. Several studies have observed a change in the magnitude of the readiness potential over the course of learning of a difficult motor task, such as mirror tracing w48,135,177x. Although the majority of studies examining the readiness potential have used hand or finger movements, a readiness potential can also be seen prior to saccadic eye movements w9,57,126x and speech w91x. These findings suggest that the readiness potential is a generalized premovement signal. Because of the low spatial resolution of EEG recording, it is not known whether the pre-movement signals all emanate from one source, or from several sources close to each other. The readiness potential observed before arm, eye, foot or mouth movements could actually be emanating from each of the respective representations in the topography known to exist in the DMFC. Experiments using finer resolution techniques, such as MEG or subdural recording, will be required to resolve this question. In monkeys, correlates of the readiness potential have been observed using a variety of techniques. Field potential recordings revealed a slow potential prior to movements w97x, particularly during learning of new movements w75,225x. At the single-cell level, many studies have observed a preponderance of neurons in DMFC that discharge in advance of movement. Such discharge before arm movements has been termed ‘‘preparatory set’’ w2,179x. Early pre-movement activity has also been observed prior to saccadic and smooth pursuit eye movements w105, Fig. 21. Effects of learning on DMFC and FEF activity. ŽA. Shown is a top view of the human cerebral cortex. The approximate location of the DMFC and FEF is represented by the ellipses. The central sulcus ŽCs., precentral sulcus ŽPCs., and superior frontal sulcus ŽSFs. are indicated. ŽB. The graph shows the location of maximal-activity points in Brodmann’s area 6 observed with PET or fMRI while human subjects learned eye movement Že., limb movement Žl., or eye and limb movement Že & l. tasks. Each symbol within the figure represents the location of a maximal-activity point plotted in Talairach and Tournoux w273x coordinates. A maximal activity point is based on the average of 6–12 subjects. The subtraction method was used to determine a maximal-activity point. Typically, the baseline behaviour used for the subtraction method was the performance of a task that resembled the learned task in terms of the types of movements executed except that there was no change in performance over trials. Data were obtained from the following studies: for eye movements experiments Že.: Kawashima et al. w119x; for limb movement experiments Žl.: Jenkins et al. w112x; Jueptner et al. w115,116x; for eye and limb movement experiments Že and l.: Grafton et al. w88,89x; Sakai et al. w224x. The vertically oriented ellipse represents the approximate location of the DMFC and the horizontally oriented ellipse represents the approximate location of the FEF. See Fig. 16 for other details. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 228,243x. It remains to be seen whether such activity is truly analogous with human readiness potentials, and if it is modulated by the same factors. In conclusion, the readiness potential has been observed over the DMFC of both humans and monkeys. There is some question as to whether the DMFC is the true source of this potential. The magnitude of this potential is affected by motor learning, task difficulty, and whether the task is bimanual or unimanual. The readiness potential has been studied mainly using forelimb-movement tasks, and to a lesser degree, oculomotor tasks. The somatotopic map 435 described for the DMFC has yet to be verified using EEG recordings. 7. Metabolic imaging of the DMFC and FEF in humans PET and fMRI have been used on humans to measure the metabolic activity of the cortex as subjects performed eye and forelimb movement tasks. The metabolic activity is assumed to reflect neuronal activity. A large region of Fig. 22. Monkey and ape frontal lobe functional maps. All panels show lateral views, with anterior to the left and dorsal at the top. ŽA. Schematic of monkey frontal lobe and the movements elicited by stimulation from each region w11x. Note that eye movements are represented in the curve of the arcuate sulcus, but eyelid blinking is represented caudally, in the motor strip. ŽB. Photograph of the orangutan brain studied by Beevor and Horsley w11x; the stippling they used to show functional areas has been modified to hatched areas for clarity. FEF, frontal eye field; EBF, eyelid blinking field; MSEF, motor strip eye field. ŽC. A representative chimpanzee brain, modified from a figure by Leyton and Sherrington w144x. In this particular animal, the MSEF was not found, but the general location of this region in other chimpanzees is indicated. ŽD. A representative gorilla brain w144x. Sites from both hemispheres pooled together. In panels C and D, eye rotation was the primary movement evoked at sites 373 and 376; eyelid closing was the primary movement evoked from sites c, p, m, and n; and eye rotation occasionally accompanied the movements Žhead turnings. that were primarily evoked from sites N and III. 436 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Brodmann’s area 6 becomes active when subjects generate saccadic or smooth pursuit eye movements ŽFig. 16A.. The medial portion of this region is presumed to be the DMFC, and the lateral portion is presumed to be the FEF ŽFig. 16B w189,219x.. Here, we review imaging studies and compare the locations of the center of activation for eye movement tasks and forelimb movement tasks. In the DMFC of monkeys, the eye field overlaps with the forelimb field Žsee Fig. 15.. Fig. 16B, C, and D show the distributions of maximal activity points obtained from imaging studies conducted on the DMFC of humans as they performed eye movements alone, or forelimb movements alone, or eye and forelimb movement together. The distributions ŽFig. 16B and C. suggest that the eye and forelimb fields of the human DMFC are overlapped. Despite this overlap, the center of the eye field in the DMFC was situated anterior to the center of the forelimb field ŽFig. 17A.. This result is consistent with the somatotopy of the DMFC of monkeys, and is also consistent with earlier observations made in humans w64x. Furthermore, the center of the DMFC’s eye field was ventral to the center of forelimb field ŽFig. 17C.. This is due to the fact that the surface of the frontal lobes in humans curves downward as one advances rostrally. Finally, there was no difference in the location of the center of the eye field in the DMFC as compared with the center of the forelimb field along the mediolateral dimension ŽFig. 17B.. In most studies so far, comparisons have been based on experiments that studied either eye movements with no forelimb involvement or forelimb movements with no eye involvement. When comparisons were made between the center of a field based on forelimb movements that engaged the eyes and the center of a field based on forelimb movements that did not engage the eyes, it was found that the center of the former, the limb and eye case, was anterior to the center of the latter, the limb-only case ŽFig. 18.. This indicates that by engaging the eyes during a forelimb movement task, the center of the activity field within the DMFC shifts rostrally. A recent review by Picard and Strick w204x have suggested that the performance of complex tasks activates anterior regions of the DMFC and that the performance of simple tasks activates more posterior regions of the DMFC w204x. We compared the centers of activity within the DMFC for simple and complex tasks that involved the eyes only and forelimbs only Žsee caption of Fig. 19 for details.. For both behaviours, there was no tendency for the performance of complex tasks to activate regions anterior to those activated for the performance of simple tasks; if anything, the center of activity was situated somewhat more caudally during the performance of complex tasks ŽFig. 19.. Only experiments in which subjects were overtrained on the task were considered here, since studies have shown that learning a new task greatly augments the activity of the DMFC when compared to the overtrained condition w224x. Maximal activity points for the eye and forelimb fields of the human FEF region were compared ŽFig. 16B and C, FEF.. 6 It was found that forelimb tasks activated more caudal and medial locations of the FEF region, whereas eye movement tasks activated more rostral and lateral regions ŽFig. 20A and B.. Consistent with this was the observation that the forelimb tasks activated more dorsal sites in the cortex and the eye movement tasks activated more ventral sites ŽFig. 20C.. This can be explained by the fact that the surface of the frontal lobes in humans curves downward moving rostrally and laterally. To summarize, the eye and forelimb fields of the human DMFC are highly overlapped, with the center of the eye field being somewhat more anterior to the center of the forelimb field. For comparison, the eye field within the FEF region is also situated more anterior to the forelimb field of this region, although the forelimb field here represents the forelimb area of the premotor and motor cortices. Contrary to the findings of Picard and Strick, we do not find evidence for the idea that the anterior DMFC mediates ‘‘complex’’ tasks and that the posterior DMFC mediates ‘‘simple’’ tasks. When forelimb movements are accompanied by eye movements, the center of the activity field within the DMFC shifts rostrally. This concurs with the general topographic order of human as well as monkey DMFC. 8. Motor learning and the DMFC and FEF Imaging experiments performed on human subjects have disclosed that the DMFC becomes very active during the learning of new tasks ŽFig. 21 w88,89,112,115,116,119, 224x.. Learning tasks that engage both the eyes and limbs tend to similarly activate anterior and posterior sites of the DMFC w88,89,224x. One study has been done on the learning of an eye movement task, and in this case, the anterior DMFC was activated w119x. As for the FEF, one site only has been identified that is maximally active during the learning of an eye or forelimb movement task. This difference between the DMFC and FEF is quite revealing given that in all these studies, neuronal activity was assessed over both the DMFC and FEF. Sakai et al. w224x found that once human subjects master the performance of a task, the activity of the DMFC drops. They trained subjects to depress a set of buttons in a particular order using visual guidance. The DMFC was more active during the performance of an underlearned task as compared to the performance of an overlearned task. Also, the readiness potential that has been attributed to the DMFC was found to exhibit a decrease in amplitude once human subjects were overtrained w133,177x. 6 Limb-related activation in the FEF region presumably is due to neurons of the primary motor cortex and premotor cortex, which abuts human FEF. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 Chen and Wise w33,34x found that cells in the DMFC of monkeys were more readily modulated during the learning of an object–saccade association task than were cells in the FEF. For this task, a monkey was required to fixate a spot where an object stimulus was subsequently presented. Following the dousing of the object stimulus and fixation spot, the monkey was required to generate a saccade to one of four identical targets. Making a saccade to one of the four targets produced a reward. Through trial and error, the animal had to determine which of four saccade directions Ži.e., up, down, left, or right. yielded a reward for a given object stimulus. Neurons in the DMFC, as compared with neurons in the FEF, more often changed their firing rate with changes in the performance of the object–saccade association task. Aizawa et al. w1x found that training a monkey for 1 year to press two keys in response to a visual target resulted in fewer DMFC units than normal being modulated by the task. Only two units of 1120 tested were modulated after 1 year of training, compared with the normal rate of 40% observed after 1–4 months of training w278x. Also more cells in the DMFC of monkeys are modulated during the performance of a new task than during the performance of an overlearned task w175x. As mentioned previously, once monkeys are overtrained on a task, inactivation of the DMFC is no longer disruptive w168,227x. Any area of the brain that participates in the learning of motor tasks, as the DMFC seems to, should be influenced by reward delivery given that it is usually reward that motivates subjects to learn. Cells in the DMFC are modulated while monkeys fixate different positions in craniotopic space for a juice reward, but not when monkeys look about freely and obtain no reward w19,139x. Mann et al. w156x observed that cells in the DMFC respond to the delivery of juice only when the juice is delivered as a reward for task execution but not when delivered outside of task execution Že.g., as during the intertrial interval.. Also while monkeys learned an object–saccade association task for juice reward, many cells within the DMFC were modulated immediately before and after the reward period. Of the seven cells illustrated by Chen and Wise w33x showing changes in responsivity with changes in task performance, five cells showed clear changes in modulation during the reward period. That the DMFC has access to a reward signal should not be surprising given that the substantia nigra of monkeys sends a rich dopaminergic projection to this cortical area w12,13,63,72,143,210x, an input that is more robust than that to the FEFrprefrontal region and other cortical areas w13,71,143x. Like the DMFC w1,33,34,133,175,177, 224x, the substantia nigra is maximally active during the early stages of learning a motor task w145,246x. In short, the DMFC seems more involved in motor learning than the FEF. This conclusion is based on imaging data from humans and on single-unit recording data 437 from monkeys. It is apparent that the activity of the DMFC drops with protracted training on a variety of motor tasks. After such training, lesions of the DMFC are no longer disruptive. The DMFC has a substantial dopaminergic input that might mediate motor learning. 9. Misplaced human FEF? PET and fMRI studies have found that the human FEF is in the precentral sulcus, abutting or lying within the primary motor strip. These results differ from the location of the FEF in monkeys. The key to reconciling this difference may lie in a consideration of behavioural methods used by imaging investigators, who assume that their images reflect neuronal activity related to moving the eyes. To obtain these images, data derived during control scans Ži.e., while subjects fixate or are at rest. are subtracted from data derived during saccadic test scans. 7 This is intended to localize brain areas related to moving the eyes, because this is the only movement that supposedly differentiates the test scans from the control scans. However, another action has been well-documented to occur with saccades: eyelid blinking w58,62,298,303x. Unless subjects are specifically instructed to inhibit blinking, it is common for blinking to occur when saccades are made. In contrast, subjects blink less frequently during steady fixation or during rest. Subjects rarely blink during task-related saccades Žthose made toward a visual target, for instance. but they compensate by blinking with high likelihood during ‘‘return’’ saccades in the intertrial interval w58,62,303x. Blinking is particularly common for saccades greater than 108 in amplitude w303x, which means that blinking may have frequently accompanied saccades during test scans in six of the eight PET studies reviewed recently ŽPaus w189x: Refs. w4,64,137,178,191,199x., in four out of the five published fMRI studies ŽRefs. w43,47,171,197x; the exception is Luna et al. w148x., and even in the original 133 Xe regional cerebral blood flow study that started the era of FEF imaging w164x. No oculomotor imaging study, to our knowledge, has mentioned instructing the subjects not to blink, nor has any used control scans in which subjects blinked at similar rates as detected during test scans. Accordingly, most imaging studies have probably mislocated the FEF caudally, toward the motor strip which contains a region that mediates blinking responses Žan eyelid blinking field. as well as another region that medi- 7 Recently w43x, the method of fMRI has made it possible to compare data from different epochs of a task with each other Ž‘‘epoch 1’’ image–‘‘epoch 2’’ image.. The problems discussed in this section are still relevant. 438 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 ates eye movements in humans Ža motor strip eye field. ŽFig. 22 and Fig. 23A–C.. 8 Only a single imaging study that examined saccade generation failed to find the FEF near the expected precentral sulcus location w119x. In this study by Kawashima et al., subjects made a series of seven successive saccades to visual targets, each saccadic period lasting a second, and then subjects rested for a second, on average. The saccades were of relatively short amplitude 9 and nearly all saccades were task-related, such that blinking was probably inhibited until the intertrial period, when ‘‘return’’ saccades were allowed w58,62,303x. This intertrial period only occurred for about 1 s out of approximately every 9 s. It would seem that blinking once every 9 s or so is about the rate expected to occur during fixation, as well. Therefore, 8 Electrical stimulation of the FEF region in monkeys causes conjugate eye movements Že.g., Refs. w10,59,294x. ŽFig. 22A.. Eyelid blinking is evoked from the face area of the precentral motor strip of monkey ŽFig. 22A w262x.. Beevor and Horsely w11x stimulated the brain of an orangutan and found that conjugate eye movements Žwith no eyelid movements. were evoked from a region anterior to, and well-separated from, the motor strip Ž‘‘FEF’’ in Fig. 22B.. Eyelid blinking was evoked from the face area in the motor strip Ž‘‘EBF’’, for eyelid blinking field, in Fig. 22B.. A complex combination of head rotation, eye movement, and eyelid opening was also evoked within the motor strip itself, just medial to the blinking area Ž‘‘MSEF’’, for motor strip eye field, in Fig. 22B.. Leyton and Sherrington w144x confirmed all these findings in their subsequent study of orangutans, chimpanzees ŽFig. 22C., and gorillas ŽFig. 22D.. When neurosurgeons began stimulating the human brain, they found that the oculomotor layout of humans was similar to that of anthropoid apes. Rasmussen and Penfield w212x evoked eye movements from 48 stimulation sites in 31 patients and separated their eye movement sites into two groups. A posterior group consisted of 18 sites Ž37.5% of the total. on the lip of the central sulcus ŽFig. 23A.. Here, contraversive and as well as ipsiversive movements were often evoked. An anterior group consisted of 30 sites Ž62.5% of the total. that were mostly rostral to the precentral sulcus ŽFig. 23A.. At these anterior sites, nearly all the evoked movements were contraversive. Comparing the two groups of eye movement sites with data from monkey and ape Žcf. Fig. 22., it seems that the anterior group represents the FEF and that the posterior group represents the motor strip eye field. Rasmussen and Penfield also evoked eyelid movements from 27 sites in 18 patients. In contrast to the distribution of eye movement sites, the majority of eyelid sites Ž63% of the total. were on the lip of the central sulcus ŽFig. 23B.. In order to summarize these results, we calculated the average locations of Rasmussen and Penfield’s anterior eye field, posterior eye field, and eyelid field, depicting each field with an ellipse ŽFig. 23C.. The ellipses were constructed by scanning the original figures w212x into a computer graphics program, finding the x – y location of each point, and drawing an ellipse to represent each field where the ellipse’s center was at the mean x – y location and the radii of the ellipses in the x and y direction equaled the standard deviations of the points in each direction. Rasmussen and Penfield’s anterior eye field ŽFEF. is almost completely rostral to the precentral sulcus Žand mostly within the middle frontal gyrus w193x.. Note that this location is in excellent agreement with the location of Foerster’s FEF ŽFig. 1C.. In contrast to the FEF, the eyelid field and posterior eye field are mostly within the motor strip. 9 Mean 15"S.D. 58, range 78–258; calculated from their Fig. 1. we suspect that blinking activations may have been canceled out in the study of Kawashima et al. w119x when control scans Žfixation. were subtracted from the test scans. Kawashima et al. w119x found that, although precentral sulcus activation was not significant, activation in the middle frontal gyrus was strong and bilateral Žsee their Tables 1 and 3.. The location of this activation zone in Talairach space was x s 36.5 " 5.3, y s 25.8 " 12.3, and z s 29.3 " 7.7. 10 Could this be within the true human FEF? Its location in the middle frontal gyrus w119x puts it within granular cortex ŽFig. 23D. and within the mediodorsal thalamus-recipient zone Žregio frontalis, Fig. 23E., making it anatomically homologous to the monkey FEF Žsee Section 2.2.1.. The location of this putative FEF in Talairach space is about 3.5 cm anterior to the precentral sulcus and thus is well-removed from the primary motor strip. Furthermore, it is not within the dorsolateral prefrontal cortex as mapped by imaging investigators; rather, it lies about 1.1 cm posterior to it. 11 This makes the putative human FEF location, as found by Kawashima et al. w119x, homologous with monkey FEF in terms of gross location w70x. The idea, that human FEF is in the middle frontal gyrus, has very recently been supported using a method independent of functional imaging: transcranial magnetic stimulation combined with structural MRI ŽFig. 23D,E; Ro et al. w215x.. Thus, human imaging studies are needed that explicitly control eyelid blinking so as to determine whether the human FEF is indeed located within the middle frontal gyrus. The oculomotor deficits that occur after damage to the precentral region in humans might in fact be due to damage of axons originating from the middle frontal gyrus. Finally, based on our analysis Žsee 8 ., a third eye field might exist in the frontal lobe of monkeys, a motor-strip eye field, immediately adjacent to the face area within the precentral sulcus. In human imaging studies, this eye field may also contribute to biasing the FEF activity field caudally toward the motor cortex as subjects perform oculomotor tasks. 10 Mean and standard deviations of their middle frontal gyrus activations, ns 4. Note, left and right hemispheres combined, so absolute value of x was used. 11 Significantly different from the activity focus of prefrontal cortex imaging, ps 0.038, Mann–Whitney rank sum test, from the dorsolateral prefrontal cortex mean location of x s 35.5"5.3, y s 37.1"5.7, z s 23.7"7.4 in Talairach coordinates; found by pooling 39 estimates of dorsolateral prefrontal cortex that were imaged during tasks that primarily involved working memory; right and left hemispheres combined Žabsolute value of x used.; values used only if authors indicate that the activation included at least part of Brodmann’s area 46. Studies used: Cohen et al. w39,40x; Courtney et al. w44,45x; Goldberg et al. w83x; Haxby et al. w98x; McCarthy et al. w163x; Owen et al. w182–185x; Petrides et al. w200,201x; Smith et al. w260,261x. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 10. Discussion This review makes it evident that the organization and the functions of the DMFC and FEF for the most part are quite different. In what follows, we shall discuss the 439 similarities and the differences that have been established between these two structures. Ž1. The first difference between these structures is in the kinds of coding operations they perform. There is now compelling evidence to the effect that the FEF carries a 440 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 retinocentric code whereas the DMFC carries a craniocentric one. Two major lines of evidence support the existence of a retinocentric code in the FEF. Ža. Electrical stimulation of the area evokes predominantly contraversive saccades of a specific size and direction irrespective of starting eye position w216x; there is topographic order in the layout within the FEF for different direction and amplitude saccadic eye movements. Žb. Neurons in the FEF are tuned for specific directions and sizes of saccades made into contralateral space w31x; this arrangement corresponds with what has been reported with electrical stimulation. Supportive, if less compelling, is the evidence from lesion studies that show deficits in saccade execution associated with the execution of vectors w54,213,263x. Two major lines of evidence support the claim that the DMFC carries a craniocentric code. Ža. Electrical stimulation of the DMFC at most sites drives the eyes to a particular orbital position and these orbital positions are topographically ordered in the DMFC w282x. Žb. The response characteristics of single cells confirm this observation w139x. Lesion studies, again less compelling, are also in support of a craniocentric code in the DMFC; deficits have been reported in both monkeys and humans for the execution of saccades to two or more remembered target positions w73,74,264x; the deficit has been attributed to a failure to store the second or subsequent target positions in craniotopic coordinates w208x. Ž2. The second distinction between the two eye-movement areas in the frontal lobe is that the DMFC is less dedicated to the execution of saccadic eye movements than is the FEF. The DMFC contains a representation of the entire body; stimulation evokes oculomotor as well as skeletomotor responses Že.g., Mitz and Wise w166x.; and many individual neurons in the DMFC respond both to eye and limb movements, which is not the case for FEF neurons w37,38,173x. Ž3. The third difference between these two structures is revealed by the deficits that arise after ablation or reversible inactivation. Inactivation or lesions of the FEF produce deficits in saccadic latencies and velocities, in generating saccades to extinguished targets, in the selection of simultaneously presented targets, in the execution of sequences of saccadic eye movements to successive targets, and in pursuit eye movements; deficits in making saccades to extinguished targets in the selection of simultaneously presented targets, and in the execution of sequences of saccades are long-lasting. In contrast, DMFC inactivation and lesions produce virtually no deficits in saccades made to a single visual target or in pursuit eye movements; the deficits seen in initiating saccades to extinguished targets and in the execution of sequences of saccades are of small magnitude and recover rapidly. In support of the FEF lesion work, physiological studies have reported neurons involved in pursuit eye movement in the FEF w154x. In conflict with the DMFC lesion work, however, is the finding that DMFC neurons are modulated during pursuit eye movements w101,105x and that stimulation of the DMFC interferes with pursuit w102–104x. It is unclear whether this latter effect is due to direct interference with neural structures involved in the execution of pursuit eye movements. The effects reported could be due to interference produced by the stimulation that normally drives the eye to a termination zone. The DMFC and the FEF are similar in having neurons that alter their responses as animals and humans learn new visuomotor tasks although such neurons are more preva- Fig. 23. Results from intraoperative stimulation in humans, and anatomical implications of our human FEF hypothesis. Anterior is to the left in all the panels, and dorsal is to the top. Pictures in panels A and B were flipped in mirror image from the originals w212x so as to put anterior to the left. ŽA. Human Frontal lobe eye movement sites found by Rasmussen and Penfield w212x. They distinguished two groups separated by function and location, shown here as ‘‘Anterior Sites’’ Žwhich includes the sites described by arrow diagrams at top and bottom. and ‘‘Posterior Sites’’. Arrows describe the directions of eye movements evoked at each site; leftward arrow s purely contraversive movement and rightward arrow s purely ipsiversive movement. Some movements have a vertical component as well, and some are purely vertical. Also, converging arrowss convergence and ? s movement not described. ŽB. Frontal lobe eyelid movement sites found by Rasmussen and Penfield w212x. ŽC. Summary of the general locations of the FEF, the eyelid blinking field ŽEBF., and the motor strip eye field ŽMSEF., according to the data of Rasmussen and Penfield w212x Žsee 8 for details of constructing the ellipses.. ŽD. Basic cytoarchitecture of human frontal lobe, above, compared with monkey, below. Region 2 Žhorizontal lines. is the extent of granular frontal cortex, and caudal to that Žbut anterior to the central sulcus ŽCS.. is agranular frontal cortex. The region within the bold-outlined polygon is the general predicted location of the human FEF, lying in the middle frontal gyrus where neocortex changes from granular layer IV Žrostrally. to agranular Žcaudally.. A similar transition is seen in monkey FEF, lying in the bank of the arcuate sulcus ŽAS. where granular cortex changes to agranular cortex. Figures derived from Walker w301x. ŽE. Known connections of the human brain with respect to thalamus, above; compared with monkey, below. In human brain, the ‘‘regio frontalis’’ is connected primarily with the mediodorsal ŽMD. nucleus of the thalamus; the region caudal to that is connected primarily with the ventrolateral ŽVL. thalamic nucleus. Our predicted FEF region in the middle frontal gyrus is probably at a transition point, being connected with both thalamic zones. Similarly, FEF in monkey is also at a transition point where cortex changes from being more connected with MD thalamus Žstippled. to being more connected with VL thalamus Žcross-hatched.. Human brain map is modified from Bailey and von Bonin w7x; monkey brain map is modified from Walker w300x, chosen because he used similar methods Žretrograde degeneration. as was used to derive the human map w65,165x. PCS, precentral sulcus. In both D and E, the triangle shows the approximate mean location of middle frontal gyrus activation found by Kawashima et al. w119x, and the ŽX. shows the approximate regions in two human subjects where transcranial magnetic stimulation delayed saccades ŽRo et al. w215x; plotted with reference to anatomy of Yousry et al. w312x.. For explanation of other notations and area fills in panels D and E, see the original papers. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 lent in the DMFC ŽFig. 21; Chen and Wise w33,34x.. In the DMFC, a frequently made observation is a decrease in the responses of neurons as proficiency is reached on a task. No such decrease has been reported so far for the FEF. Dopaminergic inputs to the DMFC are greater than those to the FEF. Whether this difference is functionally significant as it relates to learning is unclear. Many outstanding issues pertaining to the functions of the DMFC and FEF remain to be addressed. Here we note three. First, it is not known what the magnitude is of the modifiability of DMFC and FEF neurons. At the present time, most unit recordings are limited to a time span of a few hours; changes that arise in the course of long-term learning have not been amenable to study. Second, it remains unclear as to what extent neurons in these areas play a role in memory functions. Does the execution of motor acts in the absence of direct sensory input rely on neural circuits within these structures or are they relayed to these areas from other regions? How are these structures engaged during task performance once overtraining has occurred? Third, the exact nature of the neural code in these areas needs to be clarified. While there is general agreement to the effect that the FEF carries a retinocentric code, the nature of the code in the DMFC is still under debate, as both retinocentric and craniocentric coding operations have been noted. Whether, in addition, other egocentric codes w205x or even allocentric codes w180x are operative in these or related areas needs to be assessed. Acknowledgements We would like to thank Andreas Tolias and David Leopold for comments on this review. This work was supported by NEI grant EY-08502 to P.H. Schiller. References w1x H. Aizawa, M. Inase, H. Mushiake, K. Shima, J. Tanji, Reorganization of activity in the supplementary motor area associated with motor learning and functional recovery, Exp. Brain Res. 84 Ž1991. 668–671. w2x G.E. Alexander, M.D. Crutcher, Preparation for movement: neural representation of intended direction in three motor areas of the monkey, J. Neurophysiol. 64 Ž1990. 133–150. w3x G.E. Alexander, M.R. DeLong, P.L. Strick, Parallel organization of functionally segregated circuits linking basal ganglia and cortex, Annu. Rev. Neurosci. 9 Ž1986. 357–381. w4x T.J. Anderson, I.H. Jenkins, D.J. Brooks, M.B. Hawken, R.S.J. Frackowiak, C. Kenard, Cortical control of saccades and fixations in man. A PET study, Brain 117 Ž1994. 1073–1084. w5x T. Arikuni, K. Watanabe, K. Kubota, Connections of area 8 with area 6 in the brain of macaque monkey, J. Comp. Neurol. 277 Ž1988. 21–40. w6x M. Azuma, H. Nakayama, Y. Sasaki, H. Suzuki, Relation between visual input and motor output for eye movements in monkey frontal eye field, Behav. Brain Res. 27 Ž1988. 93–98. w7x P. Bailey, G. von Bonin, The Isocortex of Man, University of Illinois Press, Urbana, IL, USA, 1951. 441 w8x H. Barbas, D.N. Pandya, Architecture and frontal cortical connections of the premotor cortex Žarea 6. in the rhesus monkey, J. Comp. Neurol. 286 Ž1987. 211–228. w9x W. Becker, O. Hoehne, K. Iwase, Bereitschaftpotential, pramotorische positivierung und andere hirnpotentiale bei sakkadis¨ chen augenbewegungen, Vision Res. 12 Ž1972. 421–436. w10x C.E. Beevor, V. Horsley, A further minute analysis by electric stimulation of the so-called motor region of the cortex cerebri in the monkey Ž Macacus sinicus ., Philos. Trans. R. Soc. London, Ser. B 179 Ž1888. 205–256. w11x C.E. Beevor, V. Horsley, A record of the results obtained by electrical excitation of the so-called motor cortex and internal capsule in an orangoutang Ž Simia satyrus ., Philos. Trans. R. Soc. London, Ser. B 181 Ž1890. 129–158. w12x B. Berger, S. Trottier, P. Gaspar, C. Verney, C. Alvarez, Major dopamine innervation of the cortical motor areas in the cynomologus monkey. A radioautographic study with comparative assessment of serotonergic afferents, Neurosci. Lett. 72 Ž1986. 121–127. w13x B. Berger, S. Trottier, C. Verney, P. Gaspar, C. Alvarez, Regional and laminar distribution of the dopamine and serotonin innervation in the macaque cerebral cortex: a radiographic study, J. Comp. Neurol. 273 Ž1988. 99–119. w14x E. Bizzi, Discharge of frontal eye field neurons during saccadic and following eye movements in unanesthetized monkeys, Exp. Brain Res. 6 Ž1968. 69–80. w15x E. Bizzi, P.H. Schiller, Single unit activity in the frontal eye field of unanesthetized monkeys during eye and head movement, Exp. Brain Res. 10 Ž1970. 151–158. w16x S.T. Bok, Histonomy of the Cerebral Cortex, Elsevier, Amsterdam, 1959. w17x L. Bon, C. Lucchetti, Neurons signalling the maintenance of attentive fixation in frontal area 6ab of macaque monkey, Exp. Brain Res. 82 Ž1990. 231–233. w18x L. Bon, C. Lucchetti, Behavioural and motor mechanisms of dorsomedial frontal cortex of Macaca monkey, Int. J. Neurosci. 60 Ž1991. 187–193. w19x L. Bon, C. Lucchetti, The dorsomedial frontal cortex of the Macaca monkey: fixation and saccade-related activity, Exp. Brain Res. 89 Ž1992. 571–580. w20x L. Bon, C. Lucchetti, Ear and eye representation in the frontal cortex, area 8b, of macaque monkey: an electrophysiological study, Exp. Brain Res. 102 Ž1994. 259–271. w21x L. Bon, C. Lucchetti, Attentional-related neurons in the supplementary eye field of the macaque monkey, Exp. Brain Res. 113 Ž1997. 180–185. w22x K. Botzel, H. Plendl, W. Paulus, M. Scherg, Bereitschaftpotential: ¨ is there a contribution of the supplementary motor area?, Electroencephalographr. Clin. Neurophysiol. 89 Ž1993. 187–196. w23x V. Braitenberg, A. Schuz, Anatomy of the Cortex: Statistic and Geometry, Springer-Verlag, London, 1991. w24x D. Braun, H. Weber, T.H. Mergner, J. Schulte-Monting, Saccadic reaction times in patients with frontal and parietal lesions, Brain 115 Ž1992. 1359–1386. w25x C. Brinkman, Lesions in supplementary motor area interfere with a monkey’s performance of a bimanual coordination task, Neurosci. Lett. 27 Ž1981. 267–270. w26x C. Brinkman, Supplementary motor area of the monkey’s cerebral cortex: short- and long-term deficits after unilateral ablation and the effects of subsequent callosal section, J. Neurosci. 4 Ž1984. 918–929. w27x C. Brinkman, R. Porter, Supplementary motor area in the monkey: activity of neurons during performance of a learned motor task, J. Neurophyiol. 42 Ž1979. 681–709. w28x K. Brodmann, Beitrage zur histologischen lokalisation der grosshirnrinde. Erste mitteilung: dei regio rolandica, J. Psychol. Neurol. 2 Ž1903. 79–107. 442 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w29x K. Brodmann, Beitrage zur histologischen lokalisation der grosshirnrinde: III. Mitteilung: die rhindenfelder der niederen afferen, J. Psychol. Neurol. Leipzig 4 Ž1905. 177–226. w30x C.J. Bruce, M.E. Goldberg, Primate frontal eye fields: I. Single neurons discharging before saccades, J. Neurophysiol. 53 Ž1985. 603–635. w31x C.J. Bruce, M.E. Goldberg, M.C. Bushnell, G.B. Stanton, Primate frontal eye fields: II. Physiological and anatomic correlates of electrically evoked eye movements, J. Neurophysiol. 54 Ž1985. 714–734. w32x A.W. Campbell, Histological Studies on the Localization of Cerebral Function, Cambridge Univ. Press, 1905. w33x L.L. Chen, S.P. Wise, Neural activity in the supplementary eye field during acquisition of conditional oculomotor associations, J. Neurophysiol. 73 Ž1995. 1101–1121. w34x L.L. Chen, S.P. Wise, Supplementary eye field contrasted with the frontal eye field during acquisition of conditional oculomotor associations, J. Neurophysiol. 73 Ž1995. 1122–1134. w35x Y.C. Chen, D. Thaler, P.D. Nixon, C.E. Stern, R.E. Passingham, The functions of the medial premotor cortex: II. The timing and selection of learned movements, Exp. Brain Res. 102 Ž1995. 461–473. w36x D. Cheyne, H. Weinberg, Neuromagnetic fields accompanying unilateral finger movements: premovement and movement-evoked fields, Exp. Brain Res. 78 Ž1989. 604–612. w37x I.-H. Chou, P.H. Schiller, Encoding of arm and eye movements in the dorsomedial frontal cortex ŽDMFC., Neural Control of Movement Abstract 2 Ž1997. 16. w38x I.-H. Chou, P.H. Schiller, Single cell responses in dorsomedial frontal cortex ŽDMFC. to eye and arm movements, Soc. Neurosci. Abstr. 23 Ž1997. 1552. w39x J.D. Cohen, S.D. Forman, T.S. Braver, B.J. Casey, D. ServanSchreiber, D.C. Noll, Activation of the prefrontal cortex in a non-spatial working memory task with functional MRI, Hum. Brain Mapp. 1 Ž1994. 293–304. w40x J.D. Cohen, W.M. Perlstein, T.S. Braver, L.E. Nystrom, D.C. Noll, J. Jonides, E.E. Smith, Temporal dynamics of brain activation during a working memory task, Nature 386 Ž1997. 604–608. w41x J.G. Colebatch, M.-P. Deiber, R.E. Passingham, K.J. Friston, R.S. Frackowiak, Regional cerebral blood flow during voluntary arm and hand movements in human subjects, J. Neurophysiol. 65 Ž1991. 1392–1401. w42x M. Corbetta, F.M. Miezin, G.L. Shulman, S.E. Petersen, A PET study of visuospatial attention, J. Neurosci. 13 Ž1993. 1202–1226. w43x S.M. Courtney, L. Petit, J.M. Maisog, L.G. Ungerleider, J.V. Haxby, An area specialized for spatial working memory in human frontal cortex, Science 279 Ž1998. 1347–1351. w44x S.M. Courtney, L.G. Ungerleider, K. Keil, J.V. Haxby, Object and spatial visual working memory activates separate neural systems in human cortex, Cereb. Cortex 6 Ž1996. 39–49. w45x S.M. Courtney, L.G. Ungerleider, K. Keil, J.V. Haxby, Transient and sustained activity in a distributed neural system for human working memory, Nature 386 Ž1997. 608–611. w46x D.P. Crowne, C.H. Yeo, I.S. Russell, The effects of unilateral frontal eye field lesions in the monkey: visual–motor guidance and avoidance behaviour, Behav. Brain Res. 2 Ž1981. 165–187. w47x D.G. Darby, A.C. Nobre, V. Thangaraj, R. Edelman, M.-M. Mesulam, S. Warach, Cortical activation in the human brain during lateral saccades using EPISTAR functional magnetic resonance imaging, Neuroimage 3 Ž1996. 53–62. w48x L. Deecke, H.H. Kornhuber, W. Lang, M. Lang, H. Schreiber, Timing function of the frontal cortex in sequential motor and learning tasks, Hum. Neurobiol. 4 Ž1985. 143–154. w49x L. Deecke, W. Lang, Generation of movement-related potentials and fields in the supplementary sensorimotor area and the primary motor area, in: H.O. Luders ŽEd.., Advances in Neurology, Supple- w50x w51x w52x w53x w54x w55x w56x w57x w58x w59x w60x w61x w62x w63x w64x w65x w66x w67x w68x w69x w70x mentary Sensorimotor Area, Vol. 70, Lippincott-Raven, Amsterdam, 1996. L. Deecke, P. Scheid, H.H. Kornhuber, Distribution of readiness potential, pre-motion positivity and motor potential of the human cerebral cortex preceding voluntary finger movements, Exp. Brain Res. 7 Ž1969. 158–168. M.-P. Deiber, R.E. Passingham, J.G. Colebatch, K.J. Friston, P.D. Nixon, R.S.J. Frackowiak, Cortical areas and the selection of movement: a study with positron emission tomography, Exp. Brain Res. 84 Ž1991. 393–402. S.Y. Deng, M.E. Goldberg, M.A. Segraves, L.G. Ungerleider, M. Mishkin, The effect of unilateral ablation of the frontal eye fields on saccadic performance in the monkey, in: E. Keller, D.S. Zee ŽEds.., Adaptive Processes in the Visual and Oculomotor Systems, Pergamon, Oxford, 1986, pp. 201–208. C. Dettmers, G.R. Fink, R.N. Lemon, K.M. Stephan, R.E. Passingham, D. Sibersweig, A. Holms, M.C. Ridding, D.J. Brooks, R.S.J. Frackowiak, Relation between cerebral activity and force in the motor area of the human brain, J. Neurophysiol. 74 Ž1995. 802–815. E.C. Dias, M.A. Segraves, Muscimol-induced inactivation of the monkey frontal eye field: effects on visually and memory-guided saccades, J. Neurophysiol. 81 Ž1999. 2191–2214. F. Doricchi, D. Perani, C. Inococcia, F. Grassi, S.F. Cappa, V. Bettinardi, G. Galati, L. Pizzamiglio, F. Fazio, Neural control of fast-regular saccades and antisaccades: an investigation using positron emission tomography, Exp. Brain Res. 116 Ž1997. 50–62. R.P. Dum, P.L. Strick, The origin of corticospinal projections from the premotor areas in the frontal lobe, J. Neurosci. 11 Ž1991. 667–689. I. Evdokimidis, T. Mergner, C.H. Lucking, Dependence of presaccadic cortical potentials on the type of saccadic eye movement, Electroencephalogr. Clin. Neuropsychol. 83 Ž1992. 179–191. C. Evinger, K.A. Manning, J.J. Pellegrini, M.A. Basso, A.S. Powers, P.A. Sibony, Not looking while leaping: the linkage of blinking and saccadic gaze shifts, Exp. Brain Res. 100 Ž1994. 337–344. D. Ferrier, Experiments on the brains of monkeys — No. I, Proc. R. Soc. London 23 Ž1875. 409–432. O. Foerster, The cerebral cortex in man, Lancet 2 Ž1931. 309–312. O. Foerster, The motor cortex in man in the light of Hughlings Jackson’s doctrines, Brain 59 Ž1936. 135–159. C. Fogarty, J.A. Stern, Eye movements and blinks: their relationship to higher cognitive processes, Int. J. Psychophysiol. 8 Ž1989. 35–42. S.L. Foote, J.H. Morrision, Development of the noradrenergic, serotonergic, and dopaminergic innervation of neocortex, Curr. Topics Dev. Biol. 21 Ž1987. 391–423. P.T. Fox, J.M. Fox, M.E. Raichle, R.M. Burde, The role of cerebral cortex in the generation of voluntary saccades: a positron emission tomographic study, J. Neurophysiol. 54 Ž1985. 348–369. W. Freeman, J.W. Watts, Retrograde degeneration of the thalamus following prefrontal lobotomy, J. Comp. Neurol. 86 Ž1947. 65–93. I. Fried, A. Katz, G. McCarthy, K.J. Sass, P. Williamson, S.S. Spencer, D. Spencer, Functional organization of human supplementary motor cortex studied by electrical stimulation, J. Neurosci. 11 Ž1991. 3656–3666. W. Fries, Cortical projections to the superior colliculus in the macaque monkey: a retrograde study using horseradish peroxidase, J. Comp. Neurol. 230 Ž1984. 55–76. G. Fritsch, E. Hitzig, Ueber die elektrische erregbarkeit des grosshirns, Archiv fur Anatomie, Physiologie, und Wissenschaftliche Medizin 37 Ž1870. 300–322. N. Fujii, H. Mushiake, M. Tamai, J. Tanji, Microstimulation of the supplementary eye field during saccade preparation, NeuroReport 6 Ž1995. 2565–2568. S. Funahashi, C.J. Bruce, P.S. Goldman-Rakic, Dorsolateral prefrontal lesions and oculomotor delayed-response performance: evi- E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w71x w72x w73x w74x w75x w76x w77x w78x w79x w80x w81x w82x w83x w84x w85x w86x w87x w88x w89x w90x dence for mnemonic ‘‘scotomas’’, J. Neurosci. 13 Ž1993. 1479– 1497. P. Gaspar, B. Berger, A. Febvret, A. Vigny, J.P. Henry, Catecholamine innervation of the human cerebral cortex as revealed by comparative immunohistochemistry of tyrosine hydroxylase and dopamine-b-hydroxylase, J. Comp. Neurol. 279 Ž1989. 249–271. P. Gaspar, I. Stephniewska, J.H. Kaas, Topography and collateralization of the dopaminergic projections to motor and lateral prefrontal cortex in owl monkeys, J. Comp. Neurol. 325 Ž1992. 1–21. B. Gaymard, C. Pierrot-Deseilligny, S. Rivaud, Impairment of sequences of memory-guided saccades after supplementary motor area lesions, Ann. Neurol. 28 Ž1990. 622–626. B. Gaymard, S. Rivaud, C. Pierrot-Deseilligny, Role of the left and right supplementary motor areas in memory-guided saccade sequences, Ann. Neurol. 34 Ž1993. 404–406. H. Gemba, K. Sasaki, Studies on cortical field potentials recorded during learning processes of visually initiated hand movements in monkeys, Exp. Brain Res. 55 Ž1984. 26–32. M. Giguere, P.S. Goldman-Rakic, Mediodorsal nucleus: areal, laminar, and tangential distributions of afferents and efferents in the frontal lobe of rhesus monkeys, J. Comp. Neurol. 277 Ž1988. 195–213. M. Glickstein, Ferrier’s mistake, Trends Neurosci. 8 Ž1985. 341– 344. J. Godoy, H. Luders, D.S. Dinner, H.H. Morris, E. Wyllie, Versive ¨ eye movements elicited by cortical stimulation of the human brain, Neurology 40 Ž1990. 296–299. M. Godschalk, R.N. Lemon, H.G.J.M. Kuypers, H.K. Ronday, Cortical afferents and efferents of monkey postarcuate area: an anatomical and electrophysiological study, Exp. Brain Res. 56 Ž1984. 410–424. M.E. Goldberg, C.J. Bruce, Primate frontal eye fields: III. Maintenance of spatially accurate saccade signal, J. Neurophysiol. 64 Ž1990. 489–508. M.E. Goldberg, M.C. Bushnell, Behavioral enhancement of visual responses in monkey cerebral cortex: II. Modulation in frontal eye fields specifically related to saccades, J. Neurophysiol. 46 Ž1981. 773–787. M.E. Golberg, M.C. Bushnell, C.J. Bruce, The effect of attentive fixation on eye movements evoked by electrical stimulation of the frontal eye fields, Exp. Brain Res. 61 Ž1986. 579–584. T.E. Goldberg, K.F. Berman, C. Randolph, J.M. Gold, D.R. Weinberger, Isolating the mnemonic component in spatial delayed response: a controlled PET 15 O-labelled water regional cerebral blood flow study in normal humans, Neuroimage 3 Ž1996. 69–78. J.P. Gottlieb, C.J. Bruce, M.G. MacAvoy, Smooth eye movements elicited by microstimulation in the primate frontal eye field, J. Neurophysiol. 69 Ž1993. 786–799. J.P. Gottlieb, M.G. MacAvoy, C.J. Bruce, Neural responses related to smooth pursuit eye movements and their correspondence with electrically elicited smooth eye movements in the primate frontal eye field, J. Neurophysiol. 72 Ž1994. 1634–1653. H.J. Gould, C.G. Cusick, T.P. Pons, J.H. Kaas, The relationship of corpus callosum connections to electrical stimulation maps of motor, supplementary motor and frontal eye fields in owl monkeys, J. Comp. Neurol. 247 Ž1986. 297–325. S.T. Grafton, A.H. Fagg, R.P. Woods, M.A. Arbib, Functional anatomy of pointing and grasping in humans, Cereb. Cortex 6 Ž1996. 226–237. S.T. Grafton, E. Hazeltine, R. Ivry, Functional mapping of sequence learning in normal humans, J. Cognit. Neurosci. 7 Ž1995. 497–510. S.T. Grafton, J.C. Mazziotta, S. Presty, K.J. Friston, R.S.J. Frackowiak, M.E. Phelps, Functional anatomy of human procedural learning determined with regional cerebral blood flow and PET, J. Neurosci. 12 Ž1992. 2542–2548. S.T. Grafton, R.P. Woods, J.C. Mazziotta, Within-arm somatotopy w91x w92x w93x w94x w95x w96x w97x w98x w99x w100x w101x w102x w103x w104x w105x w106x w107x w108x w109x w110x w111x 443 in human motor areas determined by positron emission tomography imaging of cerebral blood flow, Exp. Brain Res. 95 Ž1993. 172–176. B. Grozinger, H.H. Kornhuber, J. Kriebel, Participation of mesial ¨ cortex in speech: evidence from cerebral potentials preceding speech production in man, in: O. Creutzfeldt, H. Scheich, C. Schreiner ŽEds.., Hearing Mechanisms and Speech. Exp. Brain Res., Suppl. II, 1979, pp. 189–192. A.S.F. Grunbaum, C.S. Sherrington, Observations on the physiol¨ ogy of the cerebral cortex of anthropoid apes, Proc. R. Soc. London 72 Ž1903. 152–155. D. Guitton, H.A. Buchtel, R.M. Douglas, Frontal lobe lesions in man cause difficulties in suppressing reflexive glances and in generating goal-directed saccades, Exp. Brain Res. 58 Ž1985. 455– 472. U. Halsband, H.J. Freund, Premotor cortex and conditional motor learning in man, Brain 113 Ž1990. 207–222. U. Halsband, R.E. Passingham, Premotor cortex and the conditions for movement in monkeys Ž Macaca fascicularis., Behav. Brain Res. 18 Ž1985. 269–277. D.P. Hanes, K.G. Thompson, J.D. Schall, Relationship of presaccadic activity in frontal eye field and supplementary eye field to saccade initiation in macaque: Poisson spike train analysis, Exp. Brain Res. 103 Ž1995. 85–96. S. Hashimoto, H. Gemba, K. Sasaki, Analysis of slow cortical potentials preceding self-paced hand movements in the monkey, Exp. Neurol. 65 Ž1979. 218–229. J.V. Haxby, L.G. Ungerleider, B. Horwitz, S.I. Rapoport, C.L. Grady, Hemispheric differences in neural systems for face working memory, Hum. Brain Mapp. 3 Ž1995. 68–82. S.-Q. He, P.D. Richard, P.L. Strick, Topographic organization of corticospinal projections from the frontal lobe: motor areas on the lateral surface of the hemisphere, J. Neurosci. 13 Ž1993. 952–980. W. Heide, M. Blankenburg, E. Zimmermann, D. Kompf, Cortical control of double-step saccades: implications for spatial orientation, Ann. Neurol. 38 Ž1995. 739–748. S.J. Heinen, Single neuron activity in the dorsomedial frontal cortex during smooth pursuit eye movements, Exp. Brain Res. 104 Ž1995. 357–361. S.J. Heinen, Microstimulation in dorsomedial frontal cortex affects smooth pursuit eye movements, Soc. Neurosci. Abstr. 22 Ž1996. 968. S.J. Heinen, Miscrostimulation in dorsomedial frontal cortex inhibits smooth pursuit, Soc. Neurosci. Abstr. 23 Ž1997. 8. S.J. Heinen, A.N. Anbar, Stimulation of the supplementary eye fields affects saccade timing, Soc. Neurosci. Abstr. 24 Ž1998. 1147. S.J. Heinen, M. Liu, Single-neuron activity in the dorsomedial frontal cortex during smooth-pursuit movements to predictable target motion, Visual Neurosci. 14 Ž1997. 853–865. E. Hitzig, Ueber die galvanischen schwindelempfindungen und eine neue methode galvanischer reizung der augenmuskeln, Berliner Klinische Wochenschrift 11 Ž1870. 137–138. V. Horsley, E.A. Schafer, A record of experiments upon the ¨ functions of the cerebral cortex, Philos. Trans. R. Soc. London, Ser. B 179 Ž1888. 1–45. M.F. Huerta, J.H. Kaas, Supplementary eye field as defined by intracortical microstimulation: connections in macaques, J. Comp. Neurol. 293 Ž1990. 299–330. M.F. Huerta, L.A. Krubitzer, J.H. Kaas, Frontal eye field as defined by intracortical microstimulation in squirrel monkeys, owl monkeys, and macaque monkeys: II. Cortical connections, J. Comp. Neurol. 265 Ž1987. 332–361. K.D. Hutchins, A.M. Martino, P.L. Strick, Corticospinal projections from the medial wall of the hemisphere, Exp. Brain Res. 71 Ž1988. 667–672. M. Jahanshahi, I.H. Jenkins, R.G. Brown, C.D. Marsden, R.E. Passingham, D.J. Brooks, Self-initiated versus externally triggered movements: I. An investigation using measurement of regional 444 w112x w113x w114x w115x w116x w117x w118x w119x w120x w121x w122x w123x w124x w125x w126x w127x w128x w129x w130x E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 cerebral blood flow with PET and movement-related potentials in normal and Parkinson’s disease subjects, Brain 118 Ž1995. 913– 933. I.H. Jenkins, D.J. Brooks, P.D. Nixon, R.S.J. Frackowiak, R.E. Passingham, Motor sequence learning: a study with positron emission tomography, J. Neurosci. 14 Ž1994. 3775–3790. E.G. Jones, T.P.S. Powell, An anatomical study of converging sensory pathways within the cerebral cortex of the monkey, Brain 93 Ž1970. 793–820. E.G. Jones, S.P. Wise, Size, laminar and columnar distribution of efferent cells in the sensory–motor cortex of monkeys, J. Comp. Neurol. 175 Ž1978. 391–438. M. Jueptner, C.D. Frith, D.J. Brooks, R.S.J. Frackowiak, R.E. Passingham, Anatomy of motor learning: II. Subcortical structures and learning by trail and error, J. Neurophysiol. 77 Ž1997. 1327– 1337. M. Jueptner, K.M. Stephan, C.D. Frith, D.J. Brooks, R.S.J. Frackowiak, R.E. Passingham, Anatomy of motor learning: I. Frontal cortex and attention to action, J. Neurophysiol. 77 Ž1997. 1313– 1324. R. Kawashima, P.E. Roland, B.T. O’Sullivan, Fields in human motor areas involved in preparation for reaching, actual reaching, and visuomotor learning: a positron emission tomography study, J. Neurosci. 14 Ž1994. 3462–3474. R. Kawashima, K. Satoh, H. Itoh, S. Ono, S. Furumoto, R. Gotoch, M. Koyama, S. Yoshioka, K. Takahashi, K. Takahiashi, T. Yanagisawa, H. Fukada, Functional anatomy of GOrNO-GO discrimination and response selection — a PET study in man, Brain Res. 728 Ž1996. 79–89. R. Kawashima, J. Tanji, K. Okada, M. Sugiura, K. Sato, S. Kinomura, K. Inoue, A. Ogawa, H. Fukuda, Oculomotor sequence learning: a positron emission tomography study, Exp. Brain Res. 122 Ž1998. 1–8. E.G. Keating, Frontal eye field lesions impair predictive and visually guided pursuit eye movements, Exp. Brain Res. 86 Ž1991. 311–323. E.G. Keating, Lesions of the frontal eye field impair pursuit eye movements, but preserve the predictions driving them, Behav. Brain Res. 53 Ž1993. 91–104. E.G. Keating, A. Peirre, S. Chopra, Ablation of the pursuit area in the frontal cortex of the primate degrades foveal but not optokinetic smooth eye movements, J. Neurophysiol. 76 Ž1996. 637–641. K. Keizer, H.G.J.M. Kuypers, Distribution of corticospinal neurons with collaterals to the lower brain stem reticular formation in monkey Ž Macaca fascicularis., Exp. Brain Res. 74 Ž1989. 311– 318. M.A. Kennard, L. Ectors, Forced circling in monkeys following lesions of the frontal lobes, J. Neurophysiol. 65 Ž1938. 1243–1253. J. Kievit, H.G.J.M. Kuypers, Organization of the thalamocortical connections to the frontal lobe in the rhesus monkey, Exp. Brain Res. 29 Ž1977. 299–322. W. Klostermann, D. Klompf, W. Heide, R. Verleger, B. Wauschkuhn, T. Seyfert, The presaccadic cortical negativity prior to self-paced saccades with and without visual guidance, Electroencephalogr. Clin. Neuropsychol. 91 Ž1994. 219–228. H.H. Kornhuber, L. Deecke, Hirnpotentialanderungen bei ¨ willkurbewegungen und passiven bewegungen des menschen: bere¨ itschaftpotential und reafferente potentiale, Pflugers Archiv fur ¨ ¨ die Gesamte Physiologie 284 Ž1965. 1–17. H. Kunzle, An autoradiographic analysis of the efferent connections from premotor and adjacent prefrontal regions Žarea 6 and 9. in Macaca fascicularis, Brain Behav. Evol. 15 Ž1978. 185–234. H. Kunzle, Cortico-cortical efferents of primary motor and somatosensory regions of the cerebral cortex in Macaca fascicularis, Neuroscience 3 Ž1978. 25–39. K. Kurata, S.P. Wise, Premotor and supplementary motor cortex in w131x w132x w133x w134x w135x w136x w137x w138x w139x w140x w141x w142x w143x w144x w145x w146x w147x w148x rhesus monkeys: neuronal activity during externally and internally instructed motor tasks, Exp. Brain Res. 72 Ž1988. 237–248. D.D. Kurylo, A.A. Skavenski, Eye movements elicited by electrical stimulation of area PG in the monkey, J. Neurophysiol. 65 Ž1991. 1243–1253. H.G.J.M. Kuypers, C.E. Catsman-Berrevoets, Frontal corticosubcortical projections and their cells of origin, in: F. Reinoso-Suarez, C. Ajmone-Marsan ŽEds.., International Brain Research Organization Monograph Series, Cortical Integration: Basic, Archicortical, and Cortical Association Levels of Neural Integration, Vol. 11, Raven Press, New York, 1984, pp. 171–193. M. Lang, R. Beisteiner, G. Lindinger, L. Deecke, Changes of cortical activity when executing learned motor sequences, Exp. Brain Res. 89 Ž1992. 435–440. W. Lang, D. Cheyne, R. Kristeva, R. Beisteiner, G. Lindinger, L. Deecke, Three-dimensional location of SMA activity preceding voluntary movement: a study of electric and magnetic fields in a patient with infarction of the right supplementary motor area, Exp. Brain Res. 87 Ž1991. 688–695. M. Lang, A. Kornhuber, L. Deecke, H.H. Kornhuber, Human cerebral potentials and visuomotor learning, Eur. J. Physiol. 399 Ž1983. 342–344. W. Lang, H. Obrig, G. Lindinger, D. Cheyne, L. Deecke, Supplementary motor area activation while tapping bimanually different rhythms in musicians, Exp. Brain Res. 79 Ž1990. 506–514. W. Lang, L. Petit, P. Hollinger, U. Pietrzyk, N. Tzourio, B. ¨ Mazoyer, A. Berthoz, A positron emission tomography study of oculomotor imagery, NeuroReport 5 Ž1994. 921–924. K.-M. Lee, Electrophysiological characterization of the dorsomedial frontal cortex of the rhesus monkey, PhD thesis, Massachusetts Institute of Technology, Cambridge, 1992. K.-M. Lee, E.J. Tehovnik, Topographic distribution of fixation-related units in the dorsomedial frontal cortex of the rhesus monkey, Eur. J. Neurosci. 7 Ž1995. 1005–1011. G.R. Leichnetz, Afferent and efferent connections of the dorsolateral precentral gyrus Žarea 4, handrarm region. in the macaque monkey, with comparisons to area 8, J. Comp. Neurol. 254 Ž1986. 460–492. G.R. Leichnetz, M.E. Goldberg, Higher centers concerned with eye movements and visual attention: cerebral cortex and thalamus, in: ŽEd.., Reviews of Oculomotor Research, J.A. Buttner-Ennever ¨ Neuroanatomy of the Oculomotor System, Vol. 2, Elsevier, Amsterdam, 1988, pp. 365–429. G.R. Leichnetz, D.L. Smith, R.F. Spencer, Cortical projections to the paramedian tegmental and basilar pons in the monkey, J. Comp. Neurol. 228 Ž1984. 388–408. D.A. Lewis, M.J. Campbell, S.L. Foote, M. Goldstein, J.H. Morrision, The distribution of tyrosine hydroxylase-immunoreactive fibres in primate neocortex is widespread but regionally specific, J. Neurosci. 7 Ž1987. 279–290. A.S.F. Leyton, C.S. Sherrington, Observations on the excitable cortex of the chimpanzee, orangutan and gorilla, Q. J. Exp. Psychol. 11 Ž1917. 135–222. T. Ljungberg, P. Apicella, W. Schultz, Responses of monkey dopamine neurons during learning of behavioural reactions, J. Neurophysiol. 67 Ž1992. 145–163. M.-T. Lu, J.B. Preston, P.L. Strick, Interconnections between the prefrontal cortex and the premotor areas in the frontal lobe, J. Comp. Neurol. 341 Ž1994. 375–392. C. Lucchetti, F. Lui, L. Bon, Neglect syndrome for aversive stimuli in a macaque monkey with dorsomedial frontal cortex lesion, Neuropsychologia 36 Ž1998. 251–257. B. Luna, K.R. Thulorn, M.H. Strojwas, B.J. McCurtain, R.A. Berman, C.R. Genovese, J.A. Sweeney, Dorsal cortical regions subserving visually guided saccades in humans: an fMRI study, Cereb. Cortex 8 Ž1998. 40–47. E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w149x G. Luppino, M. Matelli, R. Camarda, G. Rizzolatti, Multiple representations of body movements in mesial area 6 and the adjacent cingulate cortex: an intracortical microstimulation study in the macaque monkey, J. Comp. Neurol. 311 Ž1991. 463–482. w150x G. Luppino, M. Matelli, R. Camarda, G. Rizzolatti, Corticocortical connections of area F3 ŽSMA-Proper. and area F6 ŽPre-SMA. in the macaque monkey, J. Comp. Neurol. 338 Ž1993. 114–140. w151x G. Luppino, M. Matelli, R. Camarda, G. Rizzolatti, Corticospinal projections from mesial frontal and cingulate areas in the monkey, NeuroReport 5 Ž1994. 2545–2548. w152x J.C. Lynch, Frontal eye field lesions in monkeys disrupt visual pursuit, Exp. Brain Res. 68 Ž1987. 437–441. w153x J.C. Lynch, J.E. Hoover, P.L. Strick, Input to the primate frontal eye field from the substantia nigra, superior colliculus, and dentate nucleus demonstrated by transneuronal transport, Exp. Brain Res. 100 Ž1994. 181–186. w154x M.G. MacAvoy, J.P. Gottlieb, C.J. Bruce, Smooth-pursuit eye movement representation in the primate frontal eye field, Cereb. Cortex 1 Ž1991. 95–102. w155x C.D. MacKinnon, S. Kapur, D. Hussey, M.C. Verrier, S. Houle, W.G. Tatton, Contributions of the mesial frontal cortex to the premovement potentials associated with intermittent hand movements in humans, Hum. Brain Mapp. 4 Ž1996. 1–22. w156x S.E. Mann, R. Thau, P.H. Schiller, Conditional task-related responses in monkey dorsomedial frontal cortex, Exp. Brain Res. 69 Ž1988. 460–468. w157x R.T. Marrocco, Saccades induced by stimulation of the frontal eye fields: interaction with voluntary and reflexive eye movements, Brain Res. 146 Ž1978. 23–34. w158x M. Matelli, G. Luppino, Thalamic input to mesial and superior area 6 in the macaque monkey, J. Comp. Neurol. 372 Ž1996. 59–87. w159x M. Matelli, G. Luppino, G. Rizzolatti, Architecture of superior and mesial area 6 and the adjacent cingulate cortex in the macaque monkey, J. Comp. Neurol. 311 Ž1991. 445–462. w160x M. Matsumura, K. Kubota, Cortical projection to hand–arm area from post-arcuate area in macaque monkeys: a histological study of retrograde transport of horseradish peroxidase, Neurosci. Lett. 11 Ž1979. 241–246. w161x Y. Matsuzaka, H. Aizawa, J. Tanji, A motor area rostral to the supplementary motor area Žpresupplementary motor area. in the monkey: neural activity during a learned motor task, J. Neurophysiol. 68 Ž1992. 653–662. w162x Y. Matsuzaka, J. Tanji, Changing directions of forthcoming arm movements: neural activity in the presupplementary and supplementary motor area of monkey cortex, J. Neurophysiol. 76 Ž1996. 2327–2342. w163x G. McCarthy, A. Pruce, R.T. Constable, J.H. Krystal, J.C. Gore, P. Goldman-Rakic, Activation of human prefrontal cortex during spatial and non-spatial working memory tasks measured by functional MRI, Cereb. Cortex 6 Ž1996. 600–611. w164x E. Melamed, B. Larsen, Cortical activation pattern during saccadic eye movements in humans: localization by focal cerebral blood flow increases, Ann. Neurol. 5 Ž1978. 79–88. w165x A. Meyer, E. Beck, T. McLardy, Prefrontal leucotomy: a neuroanatomical report, Brain 70 Ž1947. 18–49. w166x A.R. Mitz, S.P. Wise, The somatotopic organization of the supplementary motor area: intracortical microstimulation mapping, J. Neurosci. 7 Ž1987. 1010–1021. w167x K. Miyashita, K. Sakai, O. Hikosaka, Anticipatory saccades in sequential procedural learning in monkeys, J. Neurosci. 76 Ž1996. 1361–1366. w168x K. Miyashita, K. Sakai, O. Hikosaka, Effects of SMA and Pre-SMA inactivation on learning of sequential movements in monkey, Soc. Neurosci. Abstr. 22 Ž1996. 1862. w169x C.W. Mohler, M.E. Goldberg, R.H. Wurtz, Visual receptive fields of frontal eye field neurons, Brain Res. 61 Ž1973. 385–389. w170x M.J. Morrow, J.A. Sharpe, Deficits of smooth pursuit eye move- w171x w172x w173x w174x w175x w176x w177x w178x w179x w180x w181x w182x w183x w184x w185x w186x w187x w188x w189x 445 ment after unilateral frontal lobe lesions, Ann. Neurol. 37 Ž1995. 443–451. R.M. Muri, ¨ O. Heid, A.C. Nirkko, C. Ozdoba, J. Felblinger, G. Schroth, C.W. Hess, Functional organization of saccades and antisaccades in the frontal lobe in humans: a study with echo planar functional magnetic resonance imaging, J. Neurol. Neurosurg. Psychiatry 65 Ž1998. 374–377. E.A. Murray, J.D. Coulter, Organization of corticospinal neurons in the monkey, J. Comp. Neurol. 195 Ž1981. 339–365. H. Mushiake, N. Fujii, J. Tanji, Visually guided saccade versus eye–hand reach: contrasting neural activity in the cortical supplementary and frontal eye fields, J. Neurophysiol. 75 Ž1996. 2187– 2191. H. Mushiake, M. Inase, J. Tanji, Neural activity in the primate premotor, supplementary, and precentral motor cortex during visually guided and internally determined sequential movements, J. Neurophysiol. 66 Ž1991. 705–718. K. Nakamura, K. Sakai, O. Hikosaka, Neuronal activity in the medial frontal cortex during learning of sequential procedures, J. Neurophysiol. 80 Ž1998. 2671–2687. R. Neshige, H. Luders, H. Shibasaki, Recording of movement-re¨ lated potentials scalp and cortex in man, Brain 111 Ž1988. 719–736. J. Niemann, T. Winker, J. Gerling, B. Landwehrmeyer, R. Jung, Changes of slow cortical negative DC potentials during the acquisition of a complex finger motor task, Exp. Brain Res. 85 Ž1991. 417–422. G.A. O’Driscoll, N.M. Alpert, S.W. Matthysse, D.L. Levy, S.L. Rauch, P.S. Holzman, Functional anatomy of antisaccade eye movements investigated with positron emission tomography, Proc. Natl. Acad. Sci. U.S.A. 92 Ž1995. 925–929. K. Okano, J. Tanji, Neural activity in the primate motor fields of the agranular frontal cortex preceding visually triggered and selfpaced movement, Exp. Brain Res. 66 Ž1987. 155–166. C.R. Olson, S.N. Gettner, Object-centered direction selectivity in the macaque supplementary eye field, Science 269 Ž1995. 985–988. C.R. Olson, S.N. Gettner, Macaque SEF neurons encode objectcentered directions of eye movements regardless of the visual attributes of instruction cues, J. Neurophysiol. 81 Ž1999. 2340– 2346. A.M. Owen, J. Doyon, M. Petrides, A.C. Evans, Planning and spatial working memory: a positron emission tomography study in humans, Eur. J. Neurosci. 8 Ž1996. 353–364. A.M. Owen, A.C. Evans, M. Petrides, Evidence for a two-stage model of spatial working memory processing within the lateral frontal cortex: a positron emission tomography study, Cereb. Cortex 6 Ž1996. 31–38. A.M. Owen, B. Milner, M. Petrides, A.C. Evans, Memory for object features versus memory for object location: a positron emission tomography study of encoding and retrieval processes, Proc. Natl. Acad. Sci. U.S.A. 93 Ž1996. 9212–9217. A.M. Owen, C.E. Stern, R.B. Look, I. Tracey, B.R. Rosen, M. Petrides, Functional organization of spatial and non-spatial working memory processing within the human lateral frontal cortex, Proc. Natl. Acad. Sci. U.S.A. 95 Ž1998. 7721–7726. M. Pare, M. Crommelinck, D. Guitton, Gaze shifts evoked by stimulation of the superior colliculus in the head-free cat conform to the motor map but also depend on stimulus strength and fixation activity, Exp. Brain Res. 101 Ž1994. 123–139. H.B. Parthasarathy, J.D. Schall, A.M. Graybiel, Distributed but convergent ordering of corticostriatal projections: analysis of the frontal eye field and the supplementary eye field in the macaque monkeys, J. Neurosci. 12 Ž1992. 4466–4488. R.E. Passingham, Two cortical systems for directing movement. Motor Areas of the Cerebral Cortex, Ciba Foundation Symposium 132, Wiley, New York, 1987, pp. 151–164. T. Paus, Location and function of the human frontal eye field: a selective review, Neuropsychologia 34 Ž1996. 475–483. 446 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w190x T. Paus, M. Kalina, L. Patockova, Y. Anterova, R. Cerny, P. Mecir, J. Bauer, P. Krabec, Medial vs. lateral frontal lobe lesions and differential impairment of central-gaze fixation maintenance in man, Brain 114 Ž1991. 2051–2067. w191x T. Paus T, S. Marrett, K.J. Worsley, A.C. Evans, Extra-retinal modulation of cerebral blood flow in the human visual cortex: implications for saccadic suppression, J. Neurophysiol. 74 Ž1995. 2179–2184. w192x T. Paus, M. Petrides, A.C. Evans, E. Meyer, Role of human anterior cingulate cortex in the control of oculomotor, manual and speech responses: a positron emission tomography study, J. Neurophysiol. 70 Ž1993. 453–469. w193x W. Penfield, E. Boldrey, Somatic motor and sensory representation in the cerebral cortex of man as studied by electrical stimulation, Brain 60 Ž1937. 389–443. w194x W. Penfield, H. Jasper, Epilepsy and the Functional Anatomy of the Human Brain, Little, Brown, and Co., Boston, 1954. w195x W. Penfield, T. Rasmussen, The Cerebral Cortex of Man. A Clinical Study of Localization of Function, MacMillan, New York, 1950. w196x W. Penfield, K. Welch, The supplementary motor area of the cerebral cortex, Arch. Neurol. Psychiatry 66 Ž1951. 289–317. w197x L. Petit, V.P. Clark, J. Ingehom, J.V. Haxby, Dissociation of saccade-related and pursuit-related activation in human frontal eye fields as revealed by fMRI, J. Neurophysiol. 77 Ž1997. 3386–3390. w198x L. Petit, C. Orssaud, N. Tzourio, F. Crivelli, A. Berthoz, B. Mazoyer, Functional anatomy of a prelearned sequence of horizontal saccades in humans, J. Neurosci. 16 Ž1996. 3714–3728. w199x L. Petit, C. Orssaud, N. Tzourio, G. Salamon, B. Mazoyer, A. Berthoz, PET study of voluntary saccadic eye movements in humans: basal ganglia–thalamocortical system and cingulate cortex involvement, J. Neurophysiol. 69 Ž1993. 1009–1017. w200x M. Petrides, B. Alivisatos, A.C. Evans, E. Meyer, Dissociation of human mid-dorsolateral from posterior dorsolateral frontal cortex in memory processing, Proc. Natl. Acad. Sci. U.S.A. 90 Ž1993. 873–877. w201x M. Petrides, B. Alivisatos, A.C. Evans, E. Meyer, Functional activation of the human frontal cortex during performance of verbal working memory tasks, Proc. Natl. Acad. Sci. U.S.A. 90 Ž1993. 873–882. w202x M. Petrides, D.N. Pandya, Projections to the frontal cortex from the posterior parietal region in the rhesus monkey, J. Comp. Neurol. 228 Ž1984. 105–116. w203x M. Petrides, D.N. Pandya, Comparative architectonic analysis of the human and the macaque frontal cortex, in: F. Boller, J. Grafman ŽEds.., Handbook of Neuropsychology, Vol. 9, Elsevier, Amsterdam, 1994, pp. 17–58. w204x N. Picard, P.L. Strick, Motor areas of the medial wall: a review of their location and functional activation, Cereb. Cortex 6 Ž1996. 342–353. w205x C. Pierrot-Deseilligny, I. Israel, ¨ A. Berthoz, S. Rivaud, B. Gaymard, Role of the difference frontal lobe areas in the control of the horizontal component of memory-guided saccades in man, Exp. Brain Res. 95 Ž1993. 166–171. w206x C. Pierrot-Deseilligny, S. Rivaud, B. Gaymard, Y. Agid, Cortical control of memory-guided saccades in man, Exp. Brain Res. 83 Ž1991. 607–617. w207x C. Pierrot-Deseilligny, S. Rivaud, B. Gaymard, Y. Agid, Cortical control of reflexive visually guided saccades, Brain 114 Ž1991. 1473–1485. w208x C. Pierrot-Deseilligny, S. Rivaud, B. Gaymard, R. Muri, A.-I. Vermersch, Cortical control of saccades, Ann. Neurol. 37 Ž1995. 557–567. w209x E.D. Playford, I.H. Jenkins, R.E. Passingham, J. Nutt, R.S.J. Frackowiak, D.J. Brooks, Impaired medial frontal and putamen w210x w211x w212x w213x w214x w215x w216x w217x w218x w219x w220x w221x w222x w223x w224x w225x w226x w227x w228x w229x activation in Parkinson’s disease: a positron emission tomography study, Ann. Neurol. 32 Ž1992. 151–161. L.J. Porrino, P.S. Goldman-Rakic, Brainstem innervation of prefrontal and anterior cingulate cortex in the rhesus monkey revealed by retrograde transport of HRP, J. Comp. Neurol. 205 Ž1982. 63–76. T.M. Preuss, P.S. Goldman-Rakic, Myelo- and cytoarchitecture of the granular frontal cortex and surrounding regions in the strepsirhine primate, Galago and the anthropoid primate, Macaca, J. Comp. Neurol. 310 Ž1991. 429–474. T. Rasmussen, W. Penfield, Movement of the head and eyes from stimulation of human frontal cortex, Res. Publ. -Assoc. Res. Nerv. Ment. Dis. 27 Ž1948. 346–361. S. Rivaud, R.M. Muri, B. Gaymard, A.I. Vermersch, C. Pierrot-Deseilligny, Eye movement disorders after frontal eye field lesions in humans, Exp. Brain Res. 102 Ž1994. 110–120. G. Rizzolatti, M. Matelli, G. Pavesi, Deficits in attention and movement following the removal of postarcuate Žarea 6. and prearcuate Žarea 8. cortex in macaque monkeys, Brain 106 Ž1983. 655–673. T. Ro, S. Cheifet, H. Ingle, R. Shoup, R. Rafal, Localization of the humans frontal eye fields and motor hand area with transcranial magnetic stimulation and magnetic resonance imaging, Neuropsychologia 37 Ž1999. 225–231. D.A. Robinson, A.F. Fuchs, Eye movements evoked by stimulation of frontal eye fields, J. Neurophyiol. 32 Ž1969. 637–648. A.J. Rockel, R.W. Hiorns, T.P.S. Powell, The basic uniformity in structure of the neocortex, Brain 103 Ž1980. 221–244. P.E. Roland, B. Larsen, N.A. Lassen, E. Skinhoj, Supplementary motor area and other cortical areas in organizing voluntary movements in man, J. Neurophysiol. 43 Ž1980. 118–136. P.E. Roland, K. Zilles, Functions and structures of the motor cortices in humans, Curr. Opin. Neurobiol. 6 Ž1996. 773–781. R. Romo, H. Merchant, A. Zainos, A. Hernandez, Categorical perception of somesthetic stimuli: psychophysical measurements correlated with neural events in primate medial premotor cortex, Cereb. Cortex 7 Ž1997. 317–326. E.M. Rouiller, A. Babalian, O. Kazennikov, V. Moret, X.-H. Yu, M. Wiesendanger, Transcallosal connections of the distal forelimb representations of the primary and supplementary motor cortical areas in macaque monkeys, Exp. Brain Res. 102 Ž1994. 227–243. G.S. Russo, C.J. Bruce, Effect of eye position within the orbit on electrically elicited saccadic eye movements: a comparison of the macaque monkey’s frontal and supplementary eye fields, J. Neurophysiol. 69 Ž1993. 800–818. G.S. Russo, C.J. Bruce, Neurons in the supplementary eye field of rhesus monkeys code visual targets and saccadic eye movements in an oculocentric coordinate system, J. Neurophysiol. 76 Ž1996. 825–848. K. Sakai, O. Hikosaka, S. Miyauchi, R. Takino, Y. Sasaki, B. Putz, Transition of brain activation from frontal to parietal areas in visuomotor sequence learning, J. Neurosci. 18 Ž1998. 1827–1840. K. Sasaki, H. Gemba, Development and change of cortical field potentials during learning of visually initiated hand movements in monkey, Exp. Brain Res. 48 Ž1982. 429–437. K. Sasaki, H. Gemba H, Compensatory motor function of the somatosensory cortex for the motor cortex temporarily impaired by cooling in the monkey, Exp. Brain Res. 55 Ž1984. 60–68. K. Sasaki, H. Gemba, Effects of premotor cortex cooling upon visually initiated hand movements in the monkey, Brain Res. 374 Ž1986. 278–286. J.D. Schall, Neural activity related to visually guided saccadic eye movements in the supplementary motor area of rhesus monkeys, J. Neurophysiol. 66 Ž1991. 530–558. J.D. Schall, Neuronal activity related to visually guided saccades in E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w230x w231x w232x w233x w234x w235x w236x w237x w238x w239x w240x w241x w242x w243x w244x w245x w246x w247x w248x w249x w250x the frontal eye fields of rhesus monkeys: comparison with supplementary eye fields, J. Neurophysiol. 66 Ž1991. 559–579. Schall, J.D., Visuomotor areas of the frontal lobe, in: K.S. Rockland, J.H. Kaas, A. Peters ŽEds.., Cerebral Cortex: Extrastriate Cortex in Primates, Vol. 12, 1997, pp. 527–637. J.D. Schall, A. Morel, J.H. Kaas, Topography of supplementary eye field afferents to frontal eye field in macaque: implications for mapping between saccade coordinate systems, Visual Neurosci. 10 Ž1993. 385–393. J.D. Schall, A. Morel, D.J. King, J. Bullier, Topography of visual cortex connections with frontal eye field in macaque: convergence and segregation of processing streams, J. Neurosci. 15 Ž1995. 4464–4487. P.H. Schiller, The effect of superior colliculus ablation on saccades elicited by cortical stimulation, Brain Res. 122 Ž1977. 154–156. P.H. Schiller, I. Chou, The effects of frontal eye field and dorsomedial frontal cortex lesions on visually guided eye movements, Nat. Neurosci. 1 Ž1998. 248–253. P.H. Schiller, I. Chou, The effects of anterior arcuate and dorsomedial frontal cortex lesions on visually guided eye movements in the rhesus monkey: I. Single and sequential targets, submitted. P.H. Schiller, I. Chou, The effects of anterior arcuate and dorsomedial frontal cortex lesions on visually guided eye movements in the rhesus monkey: I. Paired and multiple targets, submitted. P.H. Schiller, J.H. Sandell, Interactions between visually and electrically elicited saccades before and after superior colliculus and frontal eye field ablations in the rhesus monkey, Exp. Brain Res. 49 Ž1983. 381–392. P.H. Schiller, J.H. Sandell, J.H.R. Maunsell, The effect of frontal eye field and superior colliculus lesions on saccadic latencies in the rhesus monkey, J. Neurophysiol. 57 Ž1987. 1033–1049. P.H. Schiller, S.D. True, J.L. Conway, Paired stimulation of the frontal eye fields and the superior colliculus of the rhesus monkey, Brain Res. 179 Ž1979. 162–164. P.H. Schiller, S.D. True, J.L. Conway, Deficits in eye movements following frontal eye field and superior colliculus ablations, J. Neurophysiol. 44 Ž1980. 1175–1189. M. Schlag-Rey, N. Amador, H. Sanchez, J. Schlag, Antisaccade performance predicted by neural activity in the supplementary eye field, Nature 390 Ž1997. 398–401. J. Schlag, M. Schlag-Rey, Unit activity related to spontaneous saccades in frontal dorsomedial cortex of monkey, Exp. Brain Res. 58 Ž1985. 208–211. J. Schlag, M. Schlag-Rey, Evidence for a supplementary eye field, J. Neurophysiol. 57 Ž1987. 179–200. J. Schlag, M. Schlag-Rey, I. Pigarev, Supplementary eye field: influence of eye position on neural signals of fixation, Exp. Brain Res. 90 Ž1992. 302–306. G. Schlaug, U. Knorr, R.J. Seitz, Intersubject variability of cerebral activations in acquiring a motor skill: a study with positron emission tomography, Exp. Brain Res. 98 Ž1994. 523–534. W. Schultz, P. Apicella, T. Ljungberg, Responses of monkey dopamine neurons to reward and conditioned stimuli during successive steps of learning a delayed response task, J. Neurosci. 13 Ž1993. 900–913. M.A. Segraves, Activity of monkey frontal eye field neurons projecting to oculomotor regions of the pons, J. Neurophysiol. 68 Ž1992. 1967–1985. M.A. Segraves, M.E. Goldberg, Functional properties of corticotectal neurons in the monkey’s frontal eye field, J. Neurophysiol. 58 Ž1987. 1387–1419. M.A. Segraves, K. Park, The relationship of monkey frontal eye field activity to saccadic dynamics, J. Neurophysiol. 69 Ž1993. 1880–1889. J. Sergent, E. Zuck, S. Terriah, B. MacDonald, Distributed neural network underlying musical sight-reading and keyboard performance, Science 257 Ž1992. 106–109. 447 w251x D. Shi, H.R. Friedman, C.J. Bruce, Deficits in smooth pursuit eye movements after muscimol inactivation within the primate’s frontal eye field, J. Neurophysiol. 80 Ž1998. 458–464. w252x H. Shibasaki, G. Barrett, E. Halliday, A.M. Halliday, Components of the movement-related cortical potential and their scalp topography, Electroencephalogr. Clin. Neurophysiol. 49 Ž1980. 213–226. w253x H. Shibutani, H. Sadata, J. Hyvarinen, Saccade and blinking evoked microstimulation of the posterior parietal association cortex of the monkey, Exp. Brain Res. 55 Ž1984. 1–88. w254x K. Shima, H. Mushiake, N. Saito, J. Tanji, Role for cells in the presupplementary motor area in updating motor plans, Proc. Natl. Acad. Sci. U.S.A. 93 Ž1996. 8694–8698. w255x K. Shima, J. Tanji, Both supplementary and presupplementary motor areas are crucial for the temporal organization of multiple movements, J. Neurophysiol. 80 Ž1998. 3247–3260. w256x B.L. Shook, M. Schlag-Rey, J. Schlag, Direct projection from the supplementary eye field to the nucleus raphe interpositus, Exp. Brain Res. 73 Ž1988. 215–218. w257x B.L. Shook, M. Schlag-Rey, J. Schlag, Primate supplementary eye field: I. Comparative aspects of mesencephalic and pontine connections, J. Comp. Neurol. 301 Ž1990. 618–642. w258x B.L. Shook, M. Schlag-Rey, J. Schlag, Primate supplementary eye field: II. Comparative aspects of connections with the thalamus, corpus striatum, and related forebrain nuclei, J. Comp. Neurol. 307 Ž1991. 562–583. w259x J.J. Sloper, T.P.S. Powell, A study of the axon initial segment and proximal axon of neurons in the primate motor and somatic sensory cortices, Philos. Trans. R. Soc. London, Ser. B 285 Ž1979. 123–139. w260x E.E. Smith, J. Jonides, R.A. Keoppe, E. Awh, E.H. Schumacher, S. Minoshima, Spatial versus object working memory: PET investigations, J. Cognit. Neurosci. 7 Ž1995. 337–356. w261x E.E. Smith, J. Jonides, R.A. Koeppe, Dissociating verbal and spatial working memory using PET, Cereb. Cortex 6 Ž1996. 11–20. w262x W.K. Smith, The frontal eye fields, in: P.C. Bucy ŽEd.., The Precentral Motor Cortex, University of Illinois Press, Urbana, IL, USA, 1949, pp. 307–342. w263x M.A. Sommer, E.J. Tehovnik, Reversible inactivation of macaque frontal eye field, Exp. Brain Res. 116 Ž1997. 229–249. w264x M.A. Sommer, E.J. Tehovnik, Reversible inactivation of macaque dorsomedial frontal cortex, Exp. Brain Res. 124 Ž1999. 429–446. w265x M.A. Sommer, R.H. Wurtz, Frontal eye field neurons orthodromically activated from the superior colliculus, J. Neurophysiol. 80 Ž1998. 3331–3333. w266x D.L. Sparks, L.E. Mays, Spatial localization of saccade targets: I. Compensation for stimulation-induced perturbations in eye position, J. Neurophysiol. 49 Ž1983. 45–63. w267x G.B. Stanton, S.-Y. Deng, M.E. Goldberg, N.T. McMullen, Cytoarchitectural characteristics of the frontal eye fields in macaque monkeys, J. Comp. Neurol. 282 Ž1989. 415–427. w268x G.B. Stanton, M.E. Goldberg, C.J. Bruce, Frontal eye field efferents in the macaque monkey: I. Subcortical pathways and topography of striatal and thalamic terminal fields, J. Comp. Neurol. 271 Ž1988. 473–492. w269x K.M. Stephan, G.R. Fink, R.E. Passingham, D. Sibersweig, A.O. Ceballos-Bbaumann, C.D. Frith, R.S.J. Frackowaik, Functional anatomy of the mental representation of upper extremity movements in healthy subjects, J. Neurophysiol. 73 Ž1995. 373–386. w270x H. Suzuki, A. Azuma, H. Yumiya, Stimulus and behavioural factors contributing to the activation of monkey prefrontal neurons during gazing, Jpn. J. Physiol. 29 Ž1979. 471–489. w271x H. Suzuki, M. Azuma, Topographic studies on visual neurons in the dorsomedial prefrontal cortex of the monkey, Exp. Brain Res. 53 Ž1983. 47–58. w272x J.A. Sweeney, M.A. Mintun, S. Kwee, M.B. Wiseman, D.L. Brown, D.R. Rosenberg, J.R. Carl, Positron emission tomography study of voluntary saccadic eye movements and spatial working memory, J. Neurophysiol. 75 Ž1996. 454–468. 448 E.J. TehoÕnik et al.r Brain Research ReÕiews 32 (2000) 413–448 w273x J. Talairach, P. Tournoux, Co-Planar Stereotaxic Atlas of the Brain, Thieme Medical Publishers, New York, 1988. w274x J. Tanji, New concepts of the supplementary motor area, Curr. Opin. Neurobiol. 6 Ž1996. 782–787. w275x J. Tanji, K. Kurata, Comparison of movement-related activity in two cortical motor areas of primates, J. Neurophysiol. 48 Ž1982. 633–653. w276x J. Tanji, K. Kurata, Contrasting neuronal activity in supplementary and precentral motor cortex of monkeys: I. Responses to instructions determining motor responses to forthcoming signals of different modalities, J. Neurophysiol. 53 Ž1985. 129–141. w277x J. Tanji, H. Mushiake, Comparison of neural activity in the supplementary motor area and primary motor cortex, Cognit. Brain Res. 3 Ž1996. 143–150. w278x J. Tanji, K. Okano, K. Sato, Neuronal activity in cortical motor areas related to ipsilateral, contralateral, and bilateral digit movements of the monkey, J. Neurophysiol. 60 Ž1988. 325–343. w279x J. Tanji, K. Shima, Role for supplementary motor area cells in planning several movements ahead, Nature 371 Ž1994. 413–416. w280x E.J. Tehovnik, The dorsomedial frontal cortex: eye and forelimb fields, Behav. Brain Res. 67 Ž1995. 147–163. w281x E.J. Tehovnik, Electrical stimulation of neural tissue to evoke behavioural responses, J. Neurosci. Methods 65 Ž1996. 1–17. w282x E.J. Tehovnik, K.-M. Lee, The dorsomedial frontal cortex of the rhesus monkey. Topographic representation of saccades evoked by electrical stimulation, Exp. Brain Res. 96 Ž1993. 430–442. w283x E.J. Tehovnik, K.-M. Lee, P.H. Schiller, Stimulation-evoked saccades from the dorsomedial frontal cortex of the rhesus monkey following lesions of the frontal eye fields and superior colliculus, Exp. Brain Res. 98 Ž1994. 179–190. w284x E.J. Tehovnik, W.M. Slocum, P.H. Schiller, Behavioural conditions affecting saccadic eye movements elicited electrically from the frontal lobes of primates, Eur. J. Neurosci. 11 Ž1999. 2431–2443. w285x E.J. Tehovnik, W.M. Slocum, A.S. Tolias, P.H. Schiller, Saccades induced electrically from the dorsomedial frontal cortex: evidence for a head-centered representation, Brain Res. 795 Ž1998. 287–291. w286x E.J. Tehovnik, M.A. Sommer, Electrically evoked saccades from the dorsomedial frontal cortex and frontal eye fields: a parametric evaluation reveals differences between areas, Exp. Brain Res. 117 Ž1997. 369–378. w287x E.J. Tehovnik, M.S. Sommer, Effective spread and time-course of neural inactivation caused by lidocaine injection in monkey cerebral cortex, J. Neurosci. Methods 74 Ž1997. 17–26. w288x D. Thaler, Y.C. Chen, P.D. Nixon, C.E. Stern, The functions of the medial premotor cortex: I. Simple learned movements, Exp. Brain Res. 102 Ž1995. 445–460. w289x H. Tokuno, M. Takada, A. Nambu, M. Inase, Somatotopical projections from the supplementary motor area to the red nucleus in the macaque monkey, Exp. Brain Res. 106 Ž1995. 351–355. w290x A.S. Tolias, T. Moore, S.M. Smirnakis, E.J. Tehovnik, A.G. Siapas, P.H. Schiller, Modulation of spatial sensitivity of V4 neurons during eye movements, submitted. w291x J. van der Steen, I.S. Russell, G.O. James, Effects of unilateral frontal eye field lesions on eye–head coordination in monkey, J. Neurophysiol. 55 Ž1986. 696–714. w292x H. van Mier, L.W. Tempel, J.S. Perlmutter, M.E. Raichle, S.E. Petersen, Changes in brain activity during motor learning measured with PET: effects of hand on performance and practice, J. Neurophysiol. 80 Ž1998. 2177–2199. w293x F. Viallet, J. Massion, R. Massarino, R. Khalil, Coordination w294x w295x w296x w297x w298x w299x w300x w301x w302x w303x w304x w305x w306x w307x w308x w309x w310x w311x w312x between posture and movements in a bimanual load-lifting task: putative role of a medial frontal region including the supplementary motor area, Exp. Brain Res. 88 Ž1992. 674–684. C. Vogt, O. Vogt, Zur Kenntnis der elektrisch erregbaren hirnrindengebiete bei den saugetieren, Journal fur ¨ ¨ Psychologie und Neurologie Leipzig 8 Ž1907. 277–456. C. Vogt, O. Vogt, Allgemeinere ergebnisse unserer hirnforschung, Journal fur ¨ Psychologie und Neurologie Leipzig 25 Ž1919. 279–462. C. Vogt, O. Vogt, Die vergleichend-architektonische und die vergleichend-reizphysiologische felderung der groahirnrinde unter ˆ besonderer berucksichtigung der menschlichen, Naturwis¨ senschaften 14 Ž1926. 1190–1194. G. von Bonin, P. Bailey, The Neocortex of Macaca Mulatta, University of Illinois Press, Urbana, 1947, p. 136. ¨ M. von Cranach, R. Schmid, M.W. Vogel, Uber einige bedingungen des zusammenhanges von lidschlag und blickwendung, Psychologische Forschung 33 Ž1969. 68–78. C.F. von Economo, G.N. Koskinas, Die Cytoarchitektonik der Hirnrinde des erwachsenen Menschen, Springer, Berlin, 1925. A.E. Walker, The Primate Thalamus, University of Chicago Press, Chicago, 1938. A.E. Walker, A cytoarchitectural study of the prefrontal area of the macaque monkey, J. Comp. Neurol. 73 Ž1940. 59–86. A.E. Walker, Stimulation and ablation. Their role in the history of cerebral physiology, J. Neurophysiol. 20 Ž1957. 435–449. Y. Watanabe, T. Fujita, J. Gyoba, Investigation of the blinking contingent upon saccadic eye movement, Tohuku Psychologia Folia 39 Ž1980. 121–129. K. Welch, P. Stuteville, Experimental production of unilateral neglect in monkeys, Brain 81 Ž1958. 341–347. M. Wiesendanger, Recent developments in studies of the supplementary motor areas of primates, Rev. Physiol. Biochem. Pharmacol. 103 Ž1986. 1–59. M. Wiesendanger, The riddle of supplementary motor area function, in: N. Mano, I. Hamada, M.R. DeLong ŽEds.., Role of the Cerebellum and Basal Ganglia in Voluntary Movement, Elsevier, 1993, pp. 253–266. R.H. Wilkins, The electrical excitability of the cerebrum, J. Neurosurg. 20 Ž1963. 904–916, Žtranslation of original paper by Fritsch and Hitzig, published in 1870.. S.P. Wise, J. Tanji, Supplementary and precentral motor cortex: contrast in responsiveness to peripheral input in the hindlimb area of the anaesthetized monkey, J. Comp. Neurol. 195 Ž1981. 433–442. C.N. Woolsey, Organization of somatic sensory and motor areas of the cerebral cortex, in: H.F. Harlow, C.N. Woolsey ŽEds.., Biological and Biochemical Bases of Behavior, University of Wisconsin Press, Madison, WI, USA, 1958, pp. 63–81. C.N. Woolsey, T.C. Erickson, W.E. Gilson, Localization in somatic sensory and motor areas of human cerebral cortex as determined by direct recording of evoked potentials and electrical stimulation, J. Neurosurg. 51 Ž1979. 476–506. C.N. Woolsey, P.H. Settlage, D.R. Meyer, W. Spencer, T.P. Hamuy, A.M. Travis, Patterns of localization in precentral and ‘‘supplementary’’ motor areas and their relation to the concept of a premotor area, Res. Publ. Assoc. Nerv. Ment. Dis. 30 Ž1952. 238–264. T.A. Yousry, U.D. Schmid, H. Alkadhi, D. Schmid, A. Peraud, A. Buettner, P. Winkler, Localization of the motor hand area to a knob on the precentral gyrus. A new landmark, Brain 120 Ž1997. 141– 157.