* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Hoxd1
Multielectrode array wikipedia , lookup
Caridoid escape reaction wikipedia , lookup
Proprioception wikipedia , lookup
Neurogenomics wikipedia , lookup
Neural modeling fields wikipedia , lookup
Recurrent neural network wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Synaptogenesis wikipedia , lookup
Neural coding wikipedia , lookup
Types of artificial neural networks wikipedia , lookup
Biology and consumer behaviour wikipedia , lookup
Biological neuron model wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Circumventricular organs wikipedia , lookup
Neuroanatomy wikipedia , lookup
Metastability in the brain wikipedia , lookup
Microneurography wikipedia , lookup
Synaptic gating wikipedia , lookup
Nervous system network models wikipedia , lookup
Optogenetics wikipedia , lookup
Neural engineering wikipedia , lookup
Central pattern generator wikipedia , lookup
Channelrhodopsin wikipedia , lookup
Development of the nervous system wikipedia , lookup
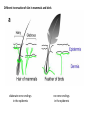
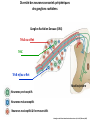
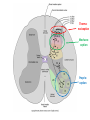
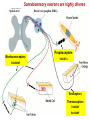
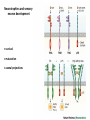
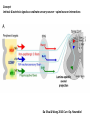
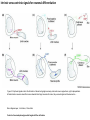
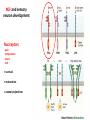
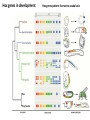
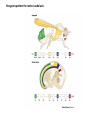
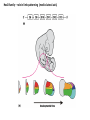
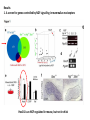
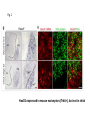
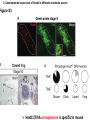
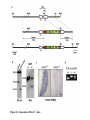
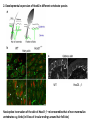
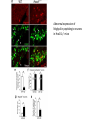
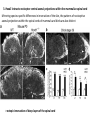
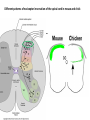
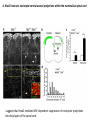
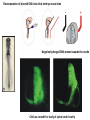
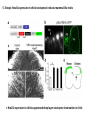
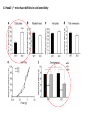
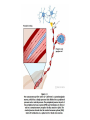
Summary so far ……. Somatosensory system > sense of body & environment on body > sensory modalities: touch, pain, temperature; proprioception > somatosensation integrated into spinal circuits > reflexes > somatosensory information sent to somatosensory cortex Development PNS nervous system > neural crest derived > somatosensory neurons of the DRG > specialized for sensory modalities The sense of touch Importance of touch > neural crest derived > somatosensory neurons of the DRG > specialized for touch > Transcription factors driave differentiation of touch sensing neurons (cMaf; MafA) Species-specific neuronal circuits directed by neurotrophic factor control of transcriptional programmes > novel neural circuits arise during evolution to encode unique behaviors among different animal species. How did somatosensory system evolve? What adaptations have been necessary in vertebrates? - Constant temperature (sea) > variable temperature (land) - Fins became limbs (circuits for coordinated movement) - Scales (fish), feathers (birds) & skin (mammals) Chick Mouse > hopping gait = sautiller > alternating locomotion > feathers > hairs, a cold adaptation necessary for the prevention of heat loss. Expect species specific differences in spinal circuitry Different innervation of skin in mammals and birds elaborate nerve endings in the epidermis no nerve endings in the epidermis Diversité des neurones sensoriels périphériques des ganglions rachidiens Ganglion Rachidien Dorsaux (DRG) TrkA ou c-Ret TrkC TrkB et/ou c-Ret Moelle épinière Neurones prorioceptifs Neurones mécanoceptifs Neurones nociceptifs & thermosensitifs Marmigère and Ernfors Nature Reviews Neuroscience 8, 114–127 (February 2007) Thermonociception Mechanoception Proprioception Somatosensory neurons are highly diverse Spinal cord Dorsal root ganglion (DRG) Proprioceptors Mechanoreceptors TrkC/NT-3 TrkB/BDNF Nociceptors Thermoceptors TrkA/NGF Ret/GDNF Neurotrophins and sensory neuron development > survival > maturation > axonal projections Intrinsic versus extrinsic signals for neuronal differentiation Concept: Intrinsic & extrinsic signals co-ordinate sensory neuron – spinal neuron interactions Da Silva & Wang 2010 Curr. Op. Neurobiol Intrinsic versus extrinsic signals for neuronal differentiation Figure 2. Peripheral signals control the formation of dorsal root ganglion sensory and motor neuron projections. (a,b) Subpopulations of brachial motor neurons extend their axons towards their target muscles. En route, they encounter glial-cell-line-derived ne... Simon Hippenmeyer, Ina Kramer, Silvia Arber Control of neuronal phenotype: what targets tell the cell bodies Concept: A species-specific signal controls nociceptive circuit formation NGF TrkA HoxD1 Skin Nociceptive neuron Spinal cord neuron DRG Spinal cord Summary The form of a scientific paper Hypothesis Species are endowed with unique sensory capabilities encoded by divergent neural circuits. One potential explanation for how divergent circuits have evolved is that conserved extrinsic signals are differentially interpreted by developing neurons of different species to yield unique patterns of axonal connections. Although NGF controls survival, maturation and axonal projections of nociceptors of different vertebrates, whether the NGF signal is differentially transduced in different species to yield unique features of nociceptor circuits is unclear. Results We identified a species-specific signaling module induced by NGF and mediated by a rapidly evolving Hox transcription factor, Hoxd1. Mice lacking Hoxd1 display altered nociceptor circuitry which resembles that normally found in chicks. Conversely, ectopic expression of Hoxd1 in developing chick nociceptors promotes a pattern of axonal projections reminiscent of the mouse. Conclusion We propose that conserved growth factors control divergent neuronal transcriptional events which mediate interspecies differences in neural circuits and the behaviors they control. NGF and sensory neuron development Nociceptors - pain - temperature - touch - itch > survival > maturation > axonal projections Hox genes in development Hox genes pattern the rostro-caudal axis Hox genes pattern the rostro-caudal axis HoxD family – role in limb patterning (medio-lateral axis) 1. Search for transcriptional targets of NGF different between birds and mammals i. Genes enriched in nociceptors ii. NGF-regulated genes expressed in nociceptors in vivo iii. mouse DRG explants grown in the presence or absence of NGF for identification of NGF-dependent genes expressed in nociceptors in vitro. > NGF dependent genes in mouse nociceptors Mouse DRG culture + NGF Chick DRG culture + NGF > differentially regulated genes detected by Q-PCR HoxD1 Results 1. A screen for genes controlled by NGF signalling in mammalian nociceptors HoxD1 is an NGF regulated in mouse, but not in chick Fig. 1 HoxD1 expressed in mouse nociceptors (TrkA+), but not in chick 2. Developmental expression of Hoxd1 in different vertebrate species > HoxD1/TrkA co-expression is specific to mouse 2. Developmental expression of Hoxd1 in different vertebrate species WT HoxD1 -/- Nociceptive innervation of the skin of Hoxd1−/− mice resembles that of non-mammalian vertebrates e.g. birds (in B loss of circular endings around hair follicles) Abnormal expression of Mrgbp4 in peptidergic neurons in HoxD1-/- mice 3. Hoxd1 instructs nociceptor central axonal projections within the mammalian spinal cord Mirroring species-specific differences in innervation of the skin, the patterns of nociceptive axonal projections within the spinal cords of mammals and birds are also distinct > ectopic innervation of deep layers of the spinal cord Different patterns of nociceptor innervation of the spinal cord in mouse and chick 4. Hoxd1 instructs nociceptor central axonal projections within the mammalian spinal cord - suggests that Hoxd1 mediates NGF-dependent suppression of nociceptor projections into deep layers of the spinal cord. In Mouse Loss of NGF or Loss of HoxD1 > “chick-type” skin & spinal cord innervation What is effect of expressing HoxD1 in chick DRG neurons? The gain-of-function experiment Electroporation of plasmid DNA into chick embryo neural tube Negatively charged DNA moves towards the anode Chick as a model for study of spinal cord circuitry 5. Ectopic Hoxd1 expression in chick nociceptors induces mammal-like traits > HoxD1 expression in chick suppressed deep layer nociceptor innervation in chick HoxD1 mutation changes nociceptor circuitry in the mouse spinal cord Physiological consequences? 6. Hoxd1−/− mice have deficits in cold sensitivity Conclusion > Hoxd1 instructs development of mammal-specific features of nociceptive neural circuitry. > behavioral sensitivity to extreme cold is markedly compromised in Hoxd1 mutant mice > suggests HoxD1 was co-opted by nociceptors in mammals for cold sensation The end