* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Metal-catalysed approaches to amide bond formation
Bottromycin wikipedia , lookup
Marcus theory wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Kinetic resolution wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Asymmetric induction wikipedia , lookup
George S. Hammond wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aza-Cope rearrangement wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Elias James Corey wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Discodermolide wikipedia , lookup
Ene reaction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Petasis reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
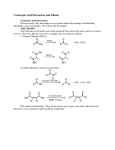
View Online Chem Soc Rev Dynamic Article Links Cite this: Chem. Soc. Rev., 2011, 40, 3405–3415 TUTORIAL REVIEW www.rsc.org/csr Metal-catalysed approaches to amide bond formation C. Liana Allen and Jonathan M. J. Williams* Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A Received 30th November 2010 DOI: 10.1039/c0cs00196a Amongst the many ways of constructing the amide bond, there has been a growing interest in the use of metal-catalysed methods for preparing this important functional group. In this tutorial review, highlights of the recent literature have been presented covering the key areas where metal catalysts have been used in amide bond formation. Acids and esters have been used in coupling reactions with amines, but aldehydes and alcohols have also been used in oxidative couplings. The use of nitriles and oximes as starting materials for amide formation are also emerging areas of interest. The use of carbon monoxide in the transition metal catalysed coupling of amines has led to a powerful methodology for amide bond formation and this is complemented by the addition of an aryl or alkenyl group to an amide typically using palladium or copper catalysts. 1. Introduction The amide bond is one of the most important functional groups in contemporary chemistry. It is essential to sustain life, making up the peptide bonds in proteins such as enzymes. It is found in numerous natural products and it is also one of the most prolific moieties in modern pharmaceutical molecules.1 Despite their obvious importance, the majority of amide bond syntheses involve the use of stoichiometric amounts of coupling reagents, making them generally expensive and wasteful procedures.2 This discrepancy has encouraged efforts towards the identification and development of more atom-efficient, catalytic methods for amide bond formation, as evidenced Department of Chemistry, University of Bath, Claverton Down, Bath, BA2 7AY, UK. E-mail: [email protected]; Fax: +44 (0)1225 386231; Tel: +44 (0)1225 383942 C. Liana Allen This journal is c Liana Allen was born near Manchester, UK. She received her Masters in Chemistry for Drug Discovery at the University of Bath and is currently a PhD candidate at the same university under the supervision of Prof. Jonathan Williams. Liana’s current research interests are developing novel, efficient, Lewis acid catalysed syntheses of amide bonds and applying them to the synthesis of pharmaceutical molecules. The Royal Society of Chemistry 2011 by the increasing number of publications in the area in recent years. Currently, the most popular industrial methods of amide synthesis rely on activation of a carboxylic acid (using a coupling reagent such as a carbodiimide) and subsequent coupling of the activated species with an amine (Scheme 1). Although a huge amount of development has been devoted to fine tuning these coupling reagents for more efficient amide synthesis, this methodology still suffers from the inherent drawback of producing a stoichiometric amount of waste product along with the desired amide.3 With this come the Scheme 1 Activation of a carboxylic acid. Jonathan Williams was born in Stourbridge, England in 1964. He received a BSc from University of York, a DPhil. from University of Oxford (with Prof. S G Davies), and was then a post-doctoral fellow at Harvard with Prof. D. A. Evans (1989–1991). He was appointed to a Lectureship in Organic Chemistry at Loughborough University in 1991, and was then appointed as a Professor of Organic Chemistry at the University Jonathan M. J. Williams of Bath in 1996, where his research has mainly involved the use of transition metals for the catalysis of organic reactions. Chem. Soc. Rev., 2011, 40, 3405–3415 3405 Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A View Online further problems of increased costs and significant waste by-products which need to be removed from the reaction mixture and cannot be recycled. Enzymatic methods are also available, although high isolation costs and somewhat limited substrate ranges can be problematic.4 In the search for an alternative to coupling reagents and enzymes in amide bond synthesis, non-metal catalysts such as organocatalysts and boron reagents5 have been reported, although these still often suffer from low atom efficiency and difficult isolations. A possible solution to these drawbacks lies with metal catalysis. Increasing attention is now being devoted to developing such amide bond syntheses which are not only atom-economical but also low cost and more environmentally friendly. Employing metal catalysis in amide syntheses also creates the possibility to start from substrates other than carboxylic acids, opening up previously unavailable synthetic routes to target molecules. 2. Amides from carboxylic acids With the abundance of coupling reagents available for the formation of amide bonds from carboxylic acids and amines, there has been little development in the area of metal catalysts for this reaction. The natural equilibrium of a carboxylic acid and an amine is heavily towards the salt formation (except at high temperatures), making a catalytic reaction between them challenging. However, the N-formylation of amines using formic acid as the formylating agent has recently been reported to proceed under catalytic conditions. Hosseini-Sarvari and Sharghi published the first example of this in their highly efficient reaction using ZnO as a catalyst under solvent-free conditions at 70 1C, achieving some excellent yields in short reaction times (Scheme 2).6 They also demonstrated the reusability of the ZnO catalyst, incurring only a small decrease in yield of amide after the third use. Rao and co-workers later published their investigation into a range of Lewis acid catalysts for the same reaction, reporting dichloride complexes of zinc, tin, lanthanum, iron, aluminium and nickel to give yields in the range of 80–100%.7 They found the best results were obtained when using ZnCl2 as a catalyst under the same solvent-free, 70 1C conditions that HosseiniSarvari and Sharghi had used. Although the catalyst loading could now be reduced from 50 mol% (ZnO) to just 10 mol% (ZnCl2), there was no report of possible recovery and reuse of Rao’s catalyst. Kim and Jang have reported the use of indium metal as a catalyst for the N-formylation of amines with formic acid, again under solvent-free conditions at 70 1C.8 They found the reaction to be efficiently catalysed by 10 mol% of the indium metal, which they presume reacts with the formic acid to form In(O2CH)3 and acts as a Lewis acid in the reaction. Scheme 2 N-Formylation of an amine with formic acid. 3406 Chem. Soc. Rev., 2011, 40, 3405–3415 3. Amides from esters As the preparation of amides from carboxylic acids is difficult to achieve in a catalytic manner, their derivatives, particularly esters, have been explored as an alternative in catalytic amide forming reactions. In 2003, a simple procedure was published by Ranu and Dutta, using a catalytic amount of indium triiodide and an excess of the amine.9 The elimination of toxic reagents and operational simplicity made this reaction a good alternative to the methods known at that time. Several excellent yields were reported for a range of amides containing functional groups using their conditions, but the reaction was not successful with secondary amines, making it unsuitable for the synthesis of tertiary amides. The same transformation was reported in 2005 by Gupta et al., using zinc dust as a reusable catalyst under either microwave or conventional heating.10 A modest range of amides was synthesised, using only aromatic esters and amines and again no demonstration of a tertiary amide synthesis. Despite the reported substrate range being limited, their procedure had the advantages of the zinc dust being able to be reused up to six times (after washing with dilute HCl) with only slight decreases in yield and a very short reaction time (2–8 minutes) when microwave heating was used. The same year, Porco and co-workers reported their findings of group (IV) metal alkoxide complexes which, in conjunction with an activator, could be used for the formation of amides from esters and amines (Scheme 3).11 They demonstrated an impressively varied substrate range, including the formation of amides 1–3 along with an intramolecular example giving 4. The wide range of structurally diverse amines and esters successfully coupled and the excellent yields attained under their reaction conditions make this a valuable methodology. A detailed mechanistic study was also carried out by the group. Using X-ray crystallography and NMR studies, they were able to determine the structures of key intermediates Scheme 3 Zirconium catalysed coupling of esters and amines with selected examples (HOAt = 1-hydroxy-7-azabenzotriazole, HOBt = 1-hydroxy-1H-benzotriazole, HYP = L-hydroxyproline). This journal is c The Royal Society of Chemistry 2011 View Online Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A Scheme 6 Scheme 4 Formation of key intermediate dimeric zirconium complexes. along the reaction pathway and deduce the active catalyst to be a dimeric zirconium complex (Scheme 4). Dimeric species 5 in which both zirconium centres are hexacoordinate is formed in the presence of amines. Coordination of an ester to one of the zirconium centres results in formation of either 6 or 7 by breaking of one bridging Zr–O bonds. Nucleophilic attack of the amine onto the ester then proceeds via a six-membered (path A) or four-membered (path B) transition state. 4. Amides from aldehydes or alcohols Aldehydes and alcohols are desirable starting materials in amide synthesis due to their ready availability and non-toxic nature. In the last ten years, catalytic systems to effect this transformation have been substantially developed into useful organic reactions. 4.1 Amides from aldehydes [not via an oxime] Oxidative amidation of aldehydes into amides has been known since the early 1980s. The general mechanism of this process is based on the reaction of an aldehyde with an amine to form a hemiaminal intermediate and subsequent oxidation to the amide product (Scheme 5). Loss of water from the hemiaminal to form the imine, then hydrogenation of the imine to form an amine is a potential side reaction in this process, one which has been exploited in the ‘borrowing hydrogen’ reaction where alcohols are used as alkylating agents for amines.12 The first catalyst system to be used for this transformation was Pd(OAc)2 (5 mol%), triphenylphosphine (15 mol%), potassium carbonate and an aryl bromide as the oxidant.13 Several aldehydes were successfully coupled with morpholine to form the corresponding amides under these conditions. Scheme 5 General mechanism of amide formation from aldehydes and amines. This journal is c The Royal Society of Chemistry 2011 Copper catalysed oxidative amidation. Improved reaction conditions using another palladium catalyst were reported by Torisawa and co-workers in 2008.14 Their use of H2O2 in urea as the oxidant combined with PdCl2 (2.5 mol%) and xantphos (2.5 mol%) allowed the reaction temperature and required time to be reduced, as well as greatly expanding the range of amides that could be synthesised in this reaction. A copper catalysed oxidative amidation has been reported (Scheme 6).13 tert-Butyl hydroperoxide solution in water serves as the oxidising agent and the use of amine hydrochloride salts minimises the competing reaction, oxidation of the amine. Their yields decreased when an aliphatic or electron-poor arylaldehyde was used, but when the reaction was applied to an enantiomerically pure amine, no racemisation occurred. Recently, several lanthanide catalysts have been reported to catalyse the oxidative coupling of aldehydes and amines. These catalysts are generally capable of facilitating the reaction at room temperature, which is a useful advantage. Additionally, no external oxidant is required, as the aldehyde is presumed to act as a hydrogen acceptor in the proposed catalytic cycle. Marks and Seos’ lanthanide-amido complex La[N(TMS)2]3 achieved varied yields of amides at room temperature in deuterated benzene.13 Due to the aldehyde substrate also acting as a hydrogen acceptor, a threefold excess was required. Shen and co-workers have reported several bimetallic lanthanide complexes used to catalyse this reaction. Their 2010 paper15 details the first structurally characterised complex of lanthanide and lithium metals with dianionic guanidinate ligands, with the Nd demonstrating its effectiveness for amidation of aldehydes with amines at just 1 mol% catalyst loading at room temperature. 4.2 Amides from alcohols [via an aldehyde] The direct catalytic conversion of alcohols and amines into amides and dihydrogen is a particularly desirable reaction due to its high atom efficiency and widely available starting materials. Murahasi and Naota reported the synthesis of lactams from an intramolecular reaction of amino alcohols in 1991,16 but the intermolecular reaction was realised by Milstein and co-workers in their breakthrough report from 2007.17 Catalysed by a ruthenium pincer complex with molecular hydrogen being the only by-product, this exceptionally clean and simple reaction has been emulated by several other groups since Milstein’s original publication (Scheme 8). The general mechanism of this reaction is the same as that of oxidative amidation involving aldehydes and amine, but with an additional oxidation step at the start to convert the alcohol into the aldehyde (Scheme 7). Milstein’s PNN pincer complex 8 undergoes an aromatisation/ dearomatisation catalytic cycle, where initial addition of the Chem. Soc. Rev., 2011, 40, 3405–3415 3407 View Online Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A Scheme 7 General mechanism of amide formation from alcohols and amines. An example of primary amide formation in this reaction was demonstrated by the group of Grützmacher.21 Using a rhodium catalyst and ammonia as the nitrogen source, primary amides were obtained in excellent yields in just four hours and at a temperature of 30 1C to 25 1C. These catalysts, though efficient and highly chemoselective in favour of amide formation, can be expensive, difficult to handle and do not tolerate secondary amines well. Some of these issues were addressed by Satsuma and co-workers in their reported g-alumina supported silver cluster.22 This re-usable, heterogeneous, easily prepared catalyst is a more economic alternative to the homogenous ruthenium and rhodium catalysts reported. In addition to this, secondary amines could be used in the reaction, giving tertiary amides in very good yields. 5. Amides from nitriles Scheme 8 Milstein’s catalyst for conversion of alcohols into amides. alcohol leads to aromatisation giving the pyridine complex 9. Subsequent loss of an aldehyde generates the known trans ruthenium dihydride complex 10. Elimination of dihydrogen regenerates the catalyst 8 and enables the catalytic cycle to continue (Scheme 9). The aldehyde forms an aminol by reaction with the amine and a similar cycle oxidises this to the amide product. Alternative ruthenium catalyst systems employing ruthenium precursors in combination with N-heterocyclic carbenes have been found to give exclusively the amide product (as opposed to the amine) as have been reported by the groups of Madsen18 and Hong.19 Although this was the first step towards the use of a simple, commercially available catalyst in this reaction, there was no real improvement in terms of yields or scope of reagents on those reported by Milstein. The first commercially available catalyst system for formation of amides from alcohols and amines was reported by our own group (although Milstein’s catalyst is now commercially available).20 The use of [Ru(p-cymene)Cl2]2 in combination with dppb, a base and a hydrogen acceptor produced the amide products in reasonable to good yields, however required increased reaction times compared with alternative catalysts. Scheme 9 Catalytic cycle for Milstein’s catalyst. 3408 Chem. Soc. Rev., 2011, 40, 3405–3415 Nitriles are well recognised as important substrates in organic chemistry due to their chemical versatility, allowing addition to the CRN triple bond by nucleophiles or electrophiles to lead to new C–N, C–C and C–O bonds. Their use in the synthesis of amides has, however, been somewhat limited to the three reactions discussed below. 5.1 Hydration of nitriles into primary amides A simple and efficient method of synthesising primary amides is the hydration of nitriles. Many metal catalysts have been reported to facilitate this transformation efficiently, an excellent and in depth review of which was pub lished by Kukushkin and Pombeiro in 2005.23 More recently, catalysts which allow the hydration of organonitriles under ambient conditions24 or in reaction times as short as 2 hours using microwave radiation25 have been published (Scheme 10). 5.2 Coupling of nitriles with amines A little known reaction which yields amides is the hydrolytic amidation of nitriles with amines. This was first published in 1986 by Murahashi and co-workers using the ruthenium catalyst RuH2(PPh3)4 (Scheme 11).26 They demonstrated the wide scope of this reaction with the efficient synthesis of several drug precursors (including 11) as well as lactams from the intramolecular reaction and diamide 12 from cinnamonitrile and a triamine. Development of this reaction has been sporadic; in 2000 a platinum catalyst was found by de Vries and co-workers to Scheme 10 Rhodium and gold catalysed nitrile hydration. This journal is c The Royal Society of Chemistry 2011 View Online Scheme 13 Iron-catalysed Ritter reaction. previously reported, but the reaction conditions are less desirable (Scheme 13).30 Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A 6. Amides from oximes Scheme 11 Murahashi’s ruthenium-catalysed hydrolytic coupling of nitriles with amines. perform the coupling,27 then in 2009 an iron catalysed version was published by our group.28 The former decreased the catalyst loading from 3 mol% with the ruthenium catalyst to levels as low as 0.1 mol% with the platinum(II) complex, but the reaction temperature remained high at 160 1C and the yields were moderate. Our own iron catalysed reaction allowed the reaction to be performed at 125 1C under solvent-free conditions, but a higher catalyst loading (10 mol%) was required as well as an excess of one of the reagents. Brief mechanistic investigations suggest that the involvement of an amidine intermediate formed from nucleophilic attack of the amine on the nitrile seems likely, as opposed to initial hydration of the nitrile to the primary amide then N-alkylation (Scheme 12). De Vries and co-workers found that when the reaction is run in the absence of water, the major product isolated is the amidine. The possibility of the reaction proceeding through initial hydration of the nitrile to the primary amide then subsequent N-acylation of the amine was ruled out after we found only a 21% conversion into secondary amide in the reaction between butyramide and benzylamine. 5.3 Coupling of nitriles with alcohols Nitriles can also be coupled with alcohols to form amides in the Ritter reaction. As an alternative to sulfuric acid, the Ritter reaction can be catalysed by metal complexes. One of the first examples of this was the use of bismuth triflate by Barrett and co-workers who published a wide range of amides synthesised from coupling various nitriles and tertiary alcohols.29 A later example by Cossy’s group described an iron-catalysed Ritter reaction with a wider substrate range than those Scheme 12 Proposed mechanism pathways for the addition of amines to nitriles. This journal is c The Royal Society of Chemistry 2011 Oximes have been used in organic synthesis since the 19th century in a diverse range of reactions. Their applications in metal catalysed amide bond synthesis have been well documented in the areas of aldoxime rearrangement into a primary amide and the Beckmann rearrangement of ketoximes. Recently, aldoximes have also been shown to react with amines to form secondary and tertiary amides. 6.1 Rearrangement of aldoximes into primary amides The rearrangement of aldoximes into primary amides has been shown to be catalysed by several metal complexes. This highly atom-efficient reaction also offers the option to start from an aldehyde and hydroxylamine (which forms the aldoxime in situ) (Scheme 14), or even the alcohol oxidation state. The examples of catalysts suitable for this rearrangement are largely based on precious metals. Chang31 and Mizuno32 have independently published reports of rhodium complexes, the latter being an example of a supported, reusable catalyst. Ruthenium catalysts have also been reported by both Crabtree33 and our group to perform this rearrangement.34 Another report by our group demonstrates the potential to start from the alcohol with the first step of the mechanism then being oxidation of the alcohol to the aldehyde, then condensation with hydroxylamine and subsequent rearrangement to the primary amide (Scheme 15).35 The oxidation step was conveniently catalysed by the same iridium catalyst used for the rearrangement and use of styrene as a sacrificial hydrogen acceptor. These current conditions are not particularly desirable, Scheme 14 Rearrangement of aldoximes into primary amides starting from an aldehyde. Scheme 15 Formation of primary amides from alcohols via aldoximes. Chem. Soc. Rev., 2011, 40, 3405–3415 3409 View Online but the novel concept of transformation of an alcohol into a primary amide via an oxime is clearly demonstrated. The most recent advances on this reaction have introduced more catalysts; a gold/silver co-catalysed system (Nolan et al.),36 a palladium acetate complex (Ali and Punniyamurthy)37 and most recently the use of simple metal salts InCl3 or Zn(NO3)2 at low catalyst loadings (0.1 mol% and 10 mol% respectively) have been published by our own group, representing the most cost-efficient catalysts reported so far.38 Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A 6.2 Beckmann rearrangement of ketoximes into secondary amides The Beckmann rearrangement of ketoximes into amides is a powerful methodology in organic synthesis. Traditionally this reaction requires harsh conditions such as high temperatures and strong acids, although recently, metal catalysed Beckmann rearrangements have been published, including conditions such as using an ionic liquid medium, or performing the reaction in the vapour phase. A notable metal catalysed varient has been reported by Ramalingan and Park and details a mercury(II) chloride catalysed reaction, run in acetonitrile at 80 1C (Scheme 16).39 A wide range of ketoximes was transformed into the corresponding secondary amides under their conditions, including several halogen substituted amides including 13 and a cyclic ketoxime yielding caprolactam 14 as the product. A further example of a metal catalysed Beckmann rearrangement uses a novel heterobimetallic catalyst with a cobalt centre and Lewis acidic zinc site on the periphery.40 Reasonable yields were achieved in a reaction time of 2 hours at 5 mol% catalyst loading. 6.3 Scheme 17 Nickel-catalysed formation of amides from aldehydes via aldoximes. Coupling of aldoximes and amines Our own recent paper on amide bond syntheses from aldoximes and amines describes the novel synthesis of amides from the coupling of aldoximes and amines.41 This reaction can also be run with the aldehyde and hydroxylamine forming the oxime in situ, catalysed by the simple metal salt NiCl2, present at 5 mol% in the reaction mixture (Scheme 17). As an additional oxidising agent is not required, this method of coupling aldehydes and amines is a more atom-efficient method compared with other conditions reported for this coupling. Based on the knowledge that a nitrile is an intermediate in the rearrangement of aldoximes into primary amides and amines and nitriles can couple to form secondary and tertiary amides, we proposed a reaction mechanism whereby the nitrile intermediate was intercepted by an amine. Scheme 18 Proposed mechanism of aldoxime rearrangement into primary amide. Further mechanistic investigations using 18O labelled oximes suggested that a bimolecular mechanism was operating (Scheme 18). A nickel metallocycle has been suggested to be a key intermediate in the mechanism, susceptible to nucleophilic attack in the presence of an amine. 7. Aminocarbonylation Scheme 16 Mercury ketoximes. 3410 catalysed Beckmann Chem. Soc. Rev., 2011, 40, 3405–3415 rearrangement of The combination of amines with carbon monoxide has been used in the catalytic aminocarbonylation of a variety of substrates to give amide products.42 Schoenberg and Heck published the first examples of the palladium-catalysed aminocarbonylation of aryl halides and vinyl halides over 35 years ago.43 Murahashi and co-workers later reported a This journal is c The Royal Society of Chemistry 2011 Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A View Online palladium catalysed carbonylation of allylamines to give b,g-unsaturated amides, avoiding the use of a halide species in the reaction. They proposed a p-allylpalladium complex, formed from oxidative addition of allylamines to a coordinatively unsaturated palladium–phosphine species, as a key intermediate in the mechanism of their reaction.44 More recent reports include the use of a Pd(OAc)2/xantphos catalyst developed by Buchwald and co-workers.45 For example, aryl bromides such as 1-bromonaphthalene 15 and 3-bromothiophene 16 were converted into the corresponding amide 17 or Weinreb amide 18 using the Pd(OAc)2/xantphos catalyst (Scheme 19). These coupling reactions involve the oxidative addition of the active Pd(0) catalyst into the aryl halide, ligand combination with CO to give an acylated palladium species which is then converted into amide by the addition of amine either directly or by prior co-ordination to the metal. Amongst the many other reports of aminocarbonylation of aryl halides, Beller and co-workers have prepared CNS active amphetamine derivatives 19 by the three component coupling of unprotected 5-bromoindole, carbon monoxide and a piperazine (Scheme 20).46 Aminocarbonylation reactions have also been performed in water using Pd(OAc)2 under ligand-free conditions.47 The direct synthesis of primary amides by aminocarbonylation can be problematic due to the handling difficulties of ammonia, its low nucleophilicity and ability to complex strongly with palladium. Several indirect approaches for the formation of primary amides have been developed, including the use of tert-butylamine as an ammonia equivalent followed by deprotection.48 Using this procedure, iodobenzene was converted into benzamide after removal of the t-Bu group with TBDMSOTf (tert-butyldimethylsilyl triflate) as shown in Scheme 21. Formamide has also been used as an ammonia equivalent for the formation of primary aromatic amides from aryl halides.49 However, Beller and co-workers have demonstrated that careful selection of a phosphine ligand does provide a catalytic system capable of converting aryl bromides with ammonia and CO directly into the corresponding primary amides.50 The use of Pd(OAc)2 in combination with dppf was effective for a range of aryl bromides and, under more forcing conditions, aryl chlorides. For example, 4-bromotoluene was converted into toluamide in good yield (Scheme 21). Aryl chlorides are usually cheaper alternatives to other aryl halides although they require more forcing conditions to undergo aminocarbonylation. Milstein and co-workers reported the first examples of the use of aryl chlorides in aminocarbonylation reactions where electron-rich bidentate phosphines were found to improve the reactivity of the palladium catalyst.51 Buchwald and co-workers have developed an interesting and practical approach to the aminocarbonylation of aryl chlorides which proceeds via an intermediate phenyl ester.52 The intermediate palladium acyl complex 20 reacts with sodium phenoxide which is a better nucleophile than the amine. The so-formed phenyl ester 21 then reacts with the amine in a subsequent step which does not involve the palladium catalyst. This approach was used for the conversion of a range of aryl chlorides into secondary and tertiary amines. In one example, the reaction of aryl chloride 22 with morpholine gave the tertiary amide 23 (Scheme 22). Mo(CO)6 has been used as a solid source of CO for the aminocarbonylation reactions of aryl triflates using a palladium catalyst, with DMAP (N,N-dimethylaminopyridine) as a co-catalyst which is believed to catalyse acyl transfer from the intermediate acylpalladium species.53 N,N-Dimethylformamide has also been used as a substrate to allow aminoformylation reactions to take place without the need for CO.54 The N,N-dimethylformamide is activated by the addition of POCl3 and then used to convert aryl iodides into the N,N-dimethylamide products (Scheme 23). Scheme 19 Pd-catalysed aminocarbonylation of aryl bromides. Scheme 20 Aminocarbonylation of an unprotected indole. This journal is c The Royal Society of Chemistry 2011 Scheme 21 Synthesis of primary amides. Chem. Soc. Rev., 2011, 40, 3405–3415 3411 View Online Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A Scheme 26 Cobalt and rhodium catalysed aminocarbonylation. Scheme 22 Aminocarbonylation of aryl chlorides. Scheme 27 Aminocarbonylation using an isocyanate, IPr = 1,3bis(2,6-diisopropylphenyl)imidazol-2-ylidene. Scheme 23 Use of DMF in aminocarbonylation. Aryl substrates which do not contain a halide or other suitable leaving group are able to undergo palladiumcatalysed coupling reactions by C–H insertion, but usually require the presence of an oxidant in order to allow for catalyst recycling. Orito and co-workers have demonstrated that substrates such as the amine-containing arene 24 can undergo direct aromatic carbonylation to give the benzolactam product 25.55 The reaction is catalysed by Pd(OAc)2 in the presence of Cu(OAc)2 and air which re-oxidises the catalyst (Scheme 24). Alkynes can also undergo aminocarbonylation reactions and this methodology is completely atom-efficient. In one example, Matteoli and co-workers have coupled phenylacetylene with aniline and CO to give the acrylamide product 26 with almost complete regioselectivity for the branched product (Scheme 25).56 Lu and Alper have reported a related example using a supported palladium catalyst to cyclise an aniline onto a pendant alkyne.57 The aminocarbonylation of alkenes has been less widely explored, although the three component coupling of an alkene, an amine and CO has considerable potential as an atom efficient synthesis of saturated amides. Chung and co-workers have used a heterogeneous cobalt catalyst for the aminocarbonylation of 1-pentene with aniline to give the amide 27 (Scheme 26).58 However, high temperatures and long reaction times were required and some alkenes led to lower conversions. An interesting double aminocarbonylation of alkynes using a rhodium catalyst has been reported by Huang and Hua.59 Using phenylacetylene as the substrate, the reaction was believed to occur by initial aminocarbonylation of the alkyne with pyrrolidine to give a branched acrylamide followed by a second aminocarbonylation leading to the 1,4-diamide 28 shown in Scheme 26. The reaction was also successful for aliphatic alkynes. Using isocyanates in place of the amine and CO, Schleicher and Jamison used a nickel catalyst complexed to the N-heterocyclic carbene ligand IPr to convert simple alkenes into the corresponding acrylamides.60 Although some alkene/ isocyanate combinations led to mixtures of branched and unbranched products, the use of vinylcyclohexane with tert-butylisocyanate gave the branched acrylamide 29 with complete selectivity and good isolated yield (Scheme 27). 8. N-Arylation and N-alkenylation of amides Scheme 24 Direct aromatic carbonylation. The cross coupling of amides with aryl and alkenyl halides is an important process that has been widely used for the preparation of amides of pharmaceutical interest.61 These reactions have mainly been achieved using palladium or copper catalysts, although other metals have been shown to be effective, including simple iron salts.62 8.1 Palladium-catalysed reactions Scheme 25 Aminocarbonylation of an alkyne. 3412 Chem. Soc. Rev., 2011, 40, 3405–3415 Buchwald and co-workers have developed a palladium catalyst that has shown broad scope for the N-arylation of amides wth aryl chlorides, aryl tosylates and aryl nonaflates (ArONf = ArOSO2CF2CF2CF2CF3).63 Typical examples This journal is c The Royal Society of Chemistry 2011 View Online Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A Scheme 30 Synthesis of potential anti-bacterial agents. Scheme 31 Amide formation by amination/hydrolysis. Scheme 28 Palladium-catalysed N-arylation reactions. include the use of [Pd(allyl)Cl]2 with JackiePhos for the coupling of aryl nonaflate 30 with amide 31 and the aryl chloride 33 with amide 34 (Scheme 28). The electron-withdrawing nature of the trifluoromethyl groups in JackiePhos was important for achieving full conversion under the reaction conditions. The more readily available ligand, xantphos (see Scheme 19), has also been shown to be useful for other palladium-catalysed cross coupling reactions of amides. Wallace and co-workers have used the Pd(0)/xantphos combination for the coupling of vinyl triflates with amides.64 Vinyl triflate 36 was coupled with acetamide to give the product 37 with good isolated yield (Scheme 29). An interesting tandem process involving N-alkenylation and N-arylation was designed by Willis and co-workers for the synthesis of indoles.65 When substrate 38 was reacted with benzamide, a double cross coupling led to the formation of the N-benzoylindole 39 (Scheme 29). The functional group tolerance of the palladium-catalysed arylation of amides is apparent from the work of a team of chemists from GlaxoSmithKline who reported the coupling of Scheme 29 Use of palladium/xantphos for N-arylation. This journal is c The Royal Society of Chemistry 2011 amide 40 with a range of heterocyclic triflates and bromides including triflate 41 to give the arylated product 42 (Scheme 30).66 An alternative approach to the formation of amides by palladium-catalysed cross coupling involving the use of anilines was developed by Xu and co-workers.67 The gemdibromoalkene 43 underwent palladium-catalysed amination where the initially-formed bromoenamine is readily hydrolysed to liberate the amide 44 which was isolated in good yield (Scheme 31). 8.2 Copper-catalysed reactions The copper-catalysed arylation of amides offers a cheaper alternative to the palladium-catalysed reactions. Typically, a copper(I) salt is used in combination with a diamine, such as 45 or 46, or a related ligand.68 Buchwald and co-workers have reported several related CuI/diamine catalysts which are effective for the N-arylation of amides with aryl halides. Representative examples for the formation of amides 47 and 48 are given in Scheme 32 Copper-catalysed N-arylation of aryl halides. Chem. Soc. Rev., 2011, 40, 3405–3415 3413 View Online Scheme 33 Copper-catalysed N-arylation of aryl halides. Scheme 36 Oxidative coupling of an amide and alkene. Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A 52. The selective formation of the (Z)-enamide was attributed to hydrogen bonding between the ester carbonyl oxygen and the amide NH in the intermediate (Scheme 36). 10. Scheme 34 Copper-catalysed N-arylation of aryl halides. Scheme 32. The N-arylation of amides could also be achieved using aryl chlorides, although a four-fold excess of the chloride was required under solvent-free conditions. The copper-catalysed coupling of vinyl halides with amides is also known,69,70 which has been exploited in an interesting example reported by the group of Li.71 N-Arylpyrroles were formed by the tandem coupling of an amide with dienyldiiodide 49. Using pentanamide, the acylpyrrole 50 was isolated in 95% yield (Scheme 33), although yields were lower in other reported examples. Potassium alkenyltrifluoroborate salts have also been used for the N-alkenylation of amides, as reported by Bolshan and Batey.72 These reactions occur under oxidative conditions with lower temperatures than those needed by vinyl halides. For example, various amides undergo copper-catalysed coupling with potassium hexenyltrifluoroborate at 40 1C (Scheme 34). 9. Enamide formation by addition reactions The transition metal catalysed addition of amides to alkynes provides a useful approach to the preparation of enamides. Gooßen and co-workers identified Ru(methallyl)(cod) with n-Bu3P and DMAP as an efficient catalyst for the selective formation of the (E)-enamide 51 from the coupling partners 1-hexyne and N-methylformamide.73 None of the branched product arising from Markovnikov addition to the alkyne was observed. The reaction was mainly used for the addition of lactams to alkynes. RuCl3 could be used as an alternative ruthenium source under otherwise similar reaction conditions,74 while the use of the rhenium catalyst Re2(CO)10 was also found to be highly regio- and stereoselective (Scheme 35).75 The oxidative coupling of amides with conjugated alkenes under Wacker-type conditions has been found to lead to the selective formation of (Z)-enamides.76 Using a palladium catalyst which required a copper co-catalyst for re-oxidation, benzamide reacted with ethyl acrylate to give the (Z)-enamide Scheme 35 Ru-catalysed addition of an amide to an alkyne. 3414 Chem. Soc. Rev., 2011, 40, 3405–3415 Conclusions In conclusion, there has been impressive progress in the use of metal catalysts for the formation of amides. The range of available methods allows the synthetic chemist to choose from a variety of starting materials for the construction of the amide bond. Acknowledgements We thank the Engineering and Physical Sciences Research Council for financial support (for C. L. A.) through the Doctoral Training Account. References 1 J. S. Carey, D. Laffan, C. Thomson and M. T. Williams, Org. Biomol. Chem., 2006, 4, 2337–2347. 2 D. J. C. Constable, P. J. Dunn, J. D. Hayler, G. R. Humphrey, J. L. Leazer, R. J. Linderman, K. Lorenz, J. Manley, B. A. Pearlman, A. Wells, A. Zaks and T. Y. Zhang, Green Chem., 2007, 9, 411–420. 3 E. Valeur and M. Bradley, Chem. Soc. Rev., 2009, 38, 606–631. 4 M. X. Wang, Top. Catal., 2005, 35, 117–130. 5 J. E. Anderson, R. Davis, R. N. Fitzgerald and J. M. Haberman, Synth. Commun., 2006, 36, 2129–2133. 6 M. Hosseini-Sarvari and H. Sharghi, J. Org. Chem., 2006, 71, 6652–6654. 7 A. C. Shekhar, A. R. Kumar, G. Sathaiah, V. L. Paul, M. Sridhar and P. S. Rao, Tetrahedron Lett., 2009, 50, 7099–7101. 8 J.-G. Kim and D. O. Jang, Synlett, 2000, 1231–1234. 9 B. C. Ranu and P. Dutta, Synth. Commun., 2003, 33, 297–301. 10 R. Arora, S. Paul and R. Gupta, Can. J. Chem., 2005, 83, 1137–1140. 11 C. Han, J. P. Lee, E. Lobkovsky and J. A. Porco, J. Am. Chem. Soc., 2005, 127, 10039–10044. 12 M. Hamid, C. L. Allen, G. W. Lamb, A. C. Maxwell, H. C. Maytum, A. J. A. Watson and J. M. J. Williams, J. Am. Chem. Soc., 2009, 131, 1766–1774. 13 K. Ekoue-Kovi and C. Wolf, Chem.–Eur. J., 2008, 14, 6302–6315 and references therein. 14 Y. Suto, N. Yamagiwa and Y. Torisawa, Tetrahedron Lett., 2008, 49, 5732–5735. 15 C. Qian, X. Zhang, Y. Zhang and Q. Shen, J. Organomet. Chem., 2010, 695, 747–752 and references therein. 16 T. Naota and S.-I. Murahasi, Synlett, 1991, 693–694. 17 C. Gunanathan, Y. Ben-David and D. Milstein, Science, 2007, 317, 790–792. 18 L. U. Nordstrøm, H. Vogt and R. Madsen, J. Am. Chem. Soc., 2008, 130, 17672–17674. 19 Y. Zhang, C. Chen, S. C. Ghosh, Y. Li and S. H. Hong, Organometallics, 2010, 29, 1374–1378. 20 A. J. A. Watson, A. C. Maxwell and J. M. J. Williams, Org. Lett., 2009, 11, 2667–2670. 21 T. Zweifel, J.-V. Naubron and H. Grützmacher, Angew. Chem., Int. Ed., 2009, 48, 559–563. This journal is c The Royal Society of Chemistry 2011 Downloaded by University of Oxford on 22 June 2011 Published on 17 March 2011 on http://pubs.rsc.org | doi:10.1039/C0CS00196A View Online 22 K. Shimizu, K. Ohshima and A. Satsuma, Chem.–Eur. J., 2009, 15, 9977–9980. 23 V. Y. Kukushkin and A. J. L. Pombeiro, Inorg. Chim. Acta, 2005, 358, 1–21. 24 A. Goto, K. Endo and S. Saito, Angew. Chem., Int. Ed., 2008, 47, 3607–3609. 25 R. S. Ramón, N. Marion and S. P. Nolan, Chem.–Eur. J., 2009, 15, 8695–8697 and ref. 8–18 therein. 26 S.-I. Murahashi, T. Naota and E. Saito, J. Am. Chem. Soc., 1986, 108, 7846–7847. 27 C. J. Cobley, M. van den Heuvel, A. Abbadi and J. G. de Vries, Tetrahedron Lett., 2000, 41, 2467–2470. 28 C. L. Allen, A. A. Lapkin and J. M. J. Williams, Tetrahedron Lett., 2009, 50, 4262–4264. 29 E. Callens, A. J. Burton and A. G. M. Barrett, Tetrahedron Lett., 2006, 47, 8699–8701. 30 B. Anxionnat, A. Guérinot, S. Reymond and J. Cossy, Tetrahedron Lett., 2009, 50, 3470–3473. 31 M. Kim, J. Lee, H.-Y. Lee and S. Chang, Adv. Synth. Catal., 2009, 351, 1807–1812. 32 H. Fujiwara, Y. Ogasawara, M. Kotani, K. Yamaguchi and N. Mizuno, Chem.–Asian J., 2008, 3, 1715–1721. 33 D. Gnanamgari and R. H. Crabtree, Organometallics, 2009, 28, 922–924. 34 N. A. Owston, A. J. Parker and J. M. J. Williams, Org. Lett., 2007, 9, 3599–3601. 35 N. A. Owston, A. J. Parker and J. M. J. Williams, Org. Lett., 2007, 9, 73–75. 36 R. S. Ramón, J. Bosson, S. Diez-González, N. Marion and S. P. Nolan, J. Org. Chem., 2010, 75, 1197–1202. 37 M. A. Ali and T. Punniyamurthy, Adv. Synth. Catal., 2010, 352, 288–292. 38 C. L. Allen, C. Burel and J. M. J. Williams, Tetrahedron Lett., 2010, 51, 2724–2726. 39 C. Ramalingan and Y.-T. Park, J. Org. Chem., 2007, 72, 4536–4538. 40 A. Mishra, A. Ali, S. Upreti and R. Gupta, Inorg. Chem., 2008, 47, 154–161 and ref. 26,27 therein. 41 C. L. Allen, S. Davulcu and J. M. J. Williams, Org. Lett., 2010, 12, 5096–5099. 42 A. Brennführer, H. Neumann and M. Beller, Angew. Chem., Int. Ed., 2009, 48, 4114–4133. 43 A. Schoenberg and R. F. Heck, J. Org. Chem., 1974, 39, 3327–3331. 44 S.-I. Murahashi, Y. Imada and K. Nishimura, J. Chem. Soc., Chem. Commun., 1988, 1578–1579. 45 J. R. Martinelli, D. A. Watson, D. M. M. Freckmann, T. E. Barder and S. L. Buchwald, J. Org. Chem., 2008, 73, 7102–7107. 46 K. Kumar, A. Zapf, D. Michalik, A. Tillack, T. Heinrich, H. Böttcher, M. Arlt and M. Beller, Org. Lett., 2004, 6, 7–10. 47 P. J. Tambade, Y. P. Patil, M. J. Bhanushali and B. M. Bhanage, Synthesis, 2008, 2347. 48 E. Takács, C. Varga, R. Skoda-Földes and L. Kollár, Tetrahedron Lett., 2007, 48, 2453–2456. This journal is c The Royal Society of Chemistry 2011 49 A. Schnyder, M. Beller, G. Mehltretter, T. Nsenda, M. Studer and A. F. Indolese, J. Org. Chem., 2001, 66, 4311–4315. 50 X.-F. Wu, H. Neumann and M. Beller, Chem.–Asian J., 2010, 5, 2168–2172. 51 Y. Ben-David, M. Portnoy and D. Milstein, J. Am. Chem. Soc., 1989, 111, 8742–8744. 52 J. R. Martinelli, T. P. Clark, D. A. Watson, R. H. Munday and S. L. Buchwald, Angew. Chem., Int. Ed., 2007, 46, 8460–8463. 53 L. R. Odell, J. Sävmaker and M. Larhed, Tetrahedron Lett., 2008, 49, 6115–6118. 54 K. Hosoi, K. Nozaki and T. Hiyama, Org. Lett., 2002, 4, 2849–2851. 55 K. Orito, A. Horibata, T. Nakamura, H. Ushito, H. Nagasaki, M. Yuguchi, S. Yamashita and M. Tokuda, J. Am. Chem. Soc., 2004, 126, 14342–14343. 56 U. Matteoli, A. Scrivanti and V. Beghetto, J. Mol. Catal. A: Chem., 2004, 213, 183–186. 57 S.-M. Lu and H. Alper, J. Am. Chem. Soc., 2008, 130, 6451–6455. 58 S. I. Lee, S. U. Son and Y. K. Chung, Chem. Commun., 2002, 1310–1311. 59 Q. Huang and R. Hua, Adv. Synth. Catal., 2007, 349, 849–852. 60 K. D. Schleicher and T. F. Jamison, Org. Lett., 2007, 9, 875–878. 61 B. P. Fors, K. Dooleweerdt, Q. Zeng and S. L. Buchwald, Tetrahedron, 2009, 65, 6576–6583 and references therein. 62 A. Correa, S. Elmore and C. Bolm, Chem.–Eur. J., 2008, 14, 3527–3529. 63 J. D. Hicks, A. M. Hyde, A. Martinez Cuezva and S. L. Buchwald, J. Am. Chem. Soc., 2009, 131, 16720–16734. 64 D. J. Wallace, D. J. Klauber, C.-y. Chen and R. P. Volante, Org. Lett., 2003, 5, 4749–4752. 65 M. C. Willis, G. N. Brace and I. P. Holmes, Angew. Chem., Int. Ed., 2005, 44, 403–406. 66 C. Barfoot, G. Brooks, P. Brown, S. Dabbs, D. T. Davies, I. Giordano, A. Hennessy, G. Jones, R. Markwell, T. Miles, N. Pearson and C. A. Smethurst, Tetrahedron Lett., 2010, 51, 2685–2689. 67 W. Ye, J. Mo, T. Zhao and B. Xu, Chem. Commun., 2009, 3246–3248. 68 D. S. Surry and S. L. Buchwald, Chem. Sci., 2010, 1, 13–31 and references therein. 69 X. Pan, Q. Cai and D. Ma, Org. Lett., 2004, 6, 1809–1812. 70 L. Jiang, G. E. Job, A. Klapars and S. L. Buchwald, Org. Lett., 2003, 5, 3667–3669. 71 X. Yuan, X. Xu, X. Zhou, J. Yuan, L. Mai and Y. Li, J. Org. Chem., 2007, 72, 1510–1513. 72 Y. Bolshan and R. A. Batey, Tetrahedron, 2010, 66, 5283–5294. 73 L. J. Gooßen, J. E. Rauhaus and G. Deng, Angew. Chem., Int. Ed., 2005, 44, 4042–4045. 74 L. J. Gooßen, M. Arndt, M. Blanchot, F. Rudolphi, F. Menges and G. Nieder-Schatteburg, Adv. Synth. Catal., 2008, 350, 2701–2707. 75 S. Yudha, Y. Kuninobu and K. Takai, Org. Lett., 2007, 9, 5609–5611. 76 J. M. Lee, D.-S. Ahn, D. Y. Jung, J. Lee, Y. Do, S. K. Kim and S. Chang, J. Am. Chem. Soc., 2006, 128, 12954–12962. Chem. Soc. Rev., 2011, 40, 3405–3415 3415