* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Neurochemistry of Dementias
Optogenetics wikipedia , lookup
Alzheimer's disease wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Premovement neuronal activity wikipedia , lookup
Time perception wikipedia , lookup
Biochemistry of Alzheimer's disease wikipedia , lookup
Synaptogenesis wikipedia , lookup
Environmental enrichment wikipedia , lookup
End-plate potential wikipedia , lookup
Feature detection (nervous system) wikipedia , lookup
Eyeblink conditioning wikipedia , lookup
Long-term depression wikipedia , lookup
Biology of depression wikipedia , lookup
Neuroeconomics wikipedia , lookup
Synaptic gating wikipedia , lookup
Neural correlates of consciousness wikipedia , lookup
Neuromuscular junction wikipedia , lookup
Visual selective attention in dementia wikipedia , lookup
Neurotransmitter wikipedia , lookup
NMDA receptor wikipedia , lookup
Spike-and-wave wikipedia , lookup
Signal transduction wikipedia , lookup
Aging brain wikipedia , lookup
Stimulus (physiology) wikipedia , lookup
Endocannabinoid system wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
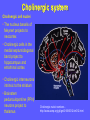
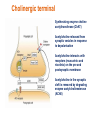
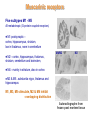
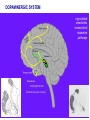
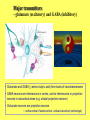
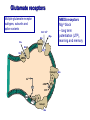
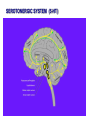
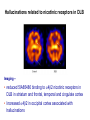
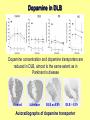
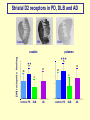
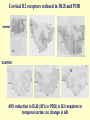
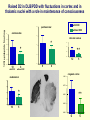
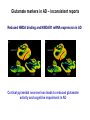
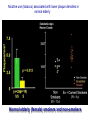
Neurochemistry of the Dementias and transmitter-based therapies Dr Margaret Piggott [email protected] [email protected] Examining neurotransmitter mechanisms is important because • Different dementias have different neurochemical profiles with implications for treatment • Neurochemical changes underlie symptoms • Antipsychotic, anxiolytic, pro-cognitive and antidepressant drugs all Modulate Transmitter Systems You will have varying familiarity with neuroscience Apologies for fact-laden stuff How much you know already? Covering things that may be in MCQ NEUROCHEMISTRY OF THE DEMENTIAS transmitter therapies THE OXFORD TEXTBOOK OF OLD AGE PSYCHIATRY (Psychiatry in the Elderly 4th edition) Chapter 6 Neurochemical pathology of neurodegenerative disorders of old age Piggott MA and Court JA (2008) (in revision) Parkinson’s Disease Dementia, edited by Professor Murat Emre Chapter 13 - Neurochemistry of Parkinson’s disease dementia Piggott MA and Perry EK (2010) Early-Onset Dementia, edited by Professor John R Hodges Chapter 9 – Neurochemical pathology in degenerative dementias Elaine Perry, Rose Goodchild and Margaret Piggott (2001) Neurotransmitter types Amino acids glutamate, aspartate, D-serine, glycine, amino butyric acid (GABA), Biogenic amines dopamine, serotonin, norepinephrine, epinephrine, histamine Others acetylcholine, adenosine, anandamide, nitric oxide Peptides over 50 peptide neurotransmitters, somatostatin, substance P, endorphin Neurotransmitters activate one or more types of receptors. The effect on the postsynaptic cell depends on the properties of those receptors Cholinergic system Cholinergic cell nuclei • The nucleus basalis of Meynert projects to neocortex • Cholinergic cells in the medial septum/diagonal band project to hippocampus and entorhinal cortex • Cholinergic interneurons intrinsic to the striatum • Brainstem pedunculopontine (PPN) neurons project to thalamus Cholinergic nuclei numbers http://www.acnp.org/g4/gn401000012/ch012.html Cholinergic terminal Synthesising enzyme choline acetyltransferase (ChAT) Acetylcholine released from synaptic vesicles in response to depolarisation Acetylcholine interacts with receptors (muscarinic and nicotinic) on the pre and postsynaptic membrane Acetylcholine in the synaptic cleft is removed by degrading enzyme acetylcholinesterase (AChE) Muscarinic receptors Five subtypes M1 - M5 M1 M1 All metabotropic (G-protein coupled receptors) M1 postsynaptic – cortex, hippocampus, striatum, low in thalamus, none in cerebellum M4/M2 M2 M2 - cortex, hippocampus, thalamus, striatum, cerebellum and brainstem, M4 - mainly in striatum, also in cortex M3 & M5 – substantia nigra, thalamus and hippocampus M2 M1 M1, M3, M5 stimulate, M2 & M4 inhibit - overlapping distribution Autoradiographs from frozen post mortem tissue Neuronal Nicotinic Receptors (nAChR) Ligand-gated ion channels (ionotropic) a4 2 a3ß2ß4a5 a7 a5 ß2 a4 2 ACh Binding Sites ß a3 2 ACh Binding Sites a7 5 ACh Binding Sites 11 different subunits a2- a 9, and ß2-ß4 • (Ca2 +, Na+) • rapid signalling • local changes presynaptic activation of nicotinic receptors leads to transmitter release from several different neuronal types – heteroreceptor (Metabotropic receptors slower, longer lasting changes) Neuronal nicotinic receptor (a42) distribution temporal cortex striatum cerebellum thalamus occipital cortex midbrain DOPAMINERGIC SYSTEM nigrostriatal mesolimbic mesocortical dopamine pathways Thalamus Dopamine receptors (all GPCR) D2, D3, D4 inhibitory, D1 & D5 stimulatory D2 and D1 in striatum > thalamus > cortex D3 is limbic, in nucleus accumbens, ventral pallidum, limbic thalamus (not cortex) D4 - despite high affinity for clozapine, & links to ADHD, receptor protein has very low density in human - many polymorphisms, and 48bp repeat (2x 4x or 7x) in third intracytoplasmic loop - D4 variants not linked to disease (except ADHD, 7x repeats) - D4 variants not associated with clinical response -defective gene ~2% population → low sensitivity to dopamine and clozapine D5 low density – cholinergic neurons, sub-thalamic nucleus antipsychotic drug potencies correlate with their ability to block D2 Major transmitters – glutamate (excitatory) and GABA (inhibitory) • Glutamate and GABA (-amino butyric acid) form basis of neurotransmission • GABA neurons are interneurons in cortex, can be interneurons or projection neurons in subcortical areas (e.g. striatal projection neurons) • Glutamate neurons are projection neurons – corticocortical, thalamocortical, cortical-subcortical (corticofugal) Glutamate receptors Multiple glutamate receptor subtypes, subunits and splice variants NMDA receptors Mg2+ block – long term potentiation (LTP), learning and memory Na+/ Ca2+ Asp 2+ Mg2+ Glu (Ca2+) Na+ H+ NMDA PCP Glu 2+ Mg2+ Mg Glu AMPA Group II G Ca2+ cAMP IP3 ATP DAG PI-PLC PIP2 G Group I Glu AC Glu Glutamate neurotransmission • Glutamate has role in cognition at normal concentrations (LTP) • Reduced glutamate affects learning and memory • Excess glutamate leads to excitotoxic cell death (Ca++) • Alzheimer’s disease - both too much and too little glutamate at different times • Glutamatergic pyramidal neurones in entorhinal cortex and hippocampus are particularly vulnerable to tangle formation and cell loss GABA receptors GABAA chloride ion channel, post-synaptic Different combinations of subunits have different pharmacology and cellular and regional distributions diverse pharmacological properties of GABAA drugs GABAB metabotropic G-protein coupled receptor (GPCR) Many drug development programmes target GABA and glutamate Benzodiazepines positively modulate GABAA and increase chloride conductance Negative GABA modulators could enhance cognition Modafinil –decreased GABA transmission and increased glutamate SEROTONERGIC SYSTEM (5-HT) SEROTONIN Receptors 7 classes of serotonin receptors, 5HT1 - 7 All GPCR (except 5HT3 - ligand-gated ion channel) 5HT4 - presynaptic, stimulate release of transmitters This array of receptor subtypes provides huge signalling possibilities • alternate splicing increases the number of proteins • oligomerisation increases the number of complexes • multiple G-proteins allow crosstalk between receptor families NORADRENERGIC SYSTEM multiple a- and -adrenergic receptors all metabotropic GPCR HISTAMINE SYSTEM . 4 Histamine Receptor types all GPCR Any more neurotransmitters? Adenosine, Cannabinoid Neuropeptide Transmitters (Substance P, Orexin, Neurotensin, Somatostatin, Substance Y, Opioids etc) human genome shows more than 300 potential GPCR About half remain ‘orphan receptors’, endogenous ligands unknown Receptor heteromers and oligomers A2A, D2, mGluR5 and M1 receptors form ‘raft’ of receptors GPCR e.g. histamine H3, can have constitutive spontaneous activity where G-protein coupled in absence of agonist Agonist or Antagonist? If it causes a response, it's an agonist If it causes a response that is relatively smaller than the response to another agonist, it's a partial agonist If it inhibits the response caused by an agonist, it's an antagonist If there is some baseline level of activity in the absence of agonist and the drug inhibits that, it's an inverse agonist AD, DLB Alzheimer’s DLB Global cognitive impairment Memory impairment plus impaired language (aphasia) impaired movement (apraxia) impaired recognition (agnosia) or disturbed executive functioning Gradual decline No disturbance of consciousness Progressive cognitive decline plus two out of three Core Features • Cognitive fluctuation of with variation in attention and alertness • Recurrent visual hallucinations • Spontaneous features of parkinsonism Additional features anxiety, wandering, depression, psychosis REM sleep behaviour disorder, neuroleptic sensitivity, low DaTSCAN, falls and syncope, transient loss of consciousness, severe autonomic dysfunction, hallucinations in other modalities, delusions, depression Dementia with Lewy bodies and Parkinson’s disease dementia • • • • • spectrum very similar clinically pathologically probably indistinguishable movement disorder before dementia by >one year PDD movement disorder within one year of dementia, or later, or not at all DLB • 20% of DLB no EPS, while PDD begins with levodopa responsive Parkinsonism • Some dopaminergic and cholinergic receptor differences (compensatory changes in PD esp. D2 up-regulation in PD) Cortical cholinergic markers in AD Post-mortem % loss ChAT activity Choline uptake AChE activity Nicotinic binding 35-50 60 40-60 30-70 Muscarinic M1 receptor reduced efficiency of coupling to G-protein as disease progresses, reduced receptor density late in disease In vivo imaging – loss of AChE, vesicular ACh transporter, M1 and nicotinic receptor Biopsy – 3.5 yrs disease, ACh markers reduced up to 50% Cholinergic Changes in DLB post-mortem neurochemistry More extensive cholinergic loss than AD (cortex and brainstem rather than hippocampus) In vivo PET – loss of cortical acetylcholinesterase (AChE) in DLB exceeds AD Cortical ChAT loss greater than in AD Striatal ChAT loss Retained cortical M1 receptors and G-protein coupling Reduced striatal M1 receptors Cortical a42 nicotinic receptors reduced as in AD, but much more reduced in striatum Clinical consequences of cholinergic losses Memory – hippocampus Learning – hippocampus, cortex Cholinergic transmission target frontal cortex Basal Ganglia intrinsic cholinergic neurons Cholinergic transmission target Thalamus, MD nucleus Attention – cortex, thalamus Consciousness, sleep, and dreaming brainstem, thalamus, cortex Movement, balance and motor regulation – striatum, brainstem, thalamus Visual function – cortex, thalamus Basal forebrain cholinergic nuclei - nbM Brain stem cholinergic nuclei - PPN and LDTg Cholinergic loss correlates with Cognitive Decline (dpm/mg prot/min) in AD ACh synthesis Reduced choline acetyltransferase (ChAT) in temporal and frontal cortex correlates with cognitive impairment 8 control value 7 6 p<0.001 5 4 3 2 1 0 0 1 2 3 4 5 6 7 8 9 Dementia rating and in DLB and PDD Visual Hallucinations Level of cholinergic activity Prevalence of recurrent complex VH in different disorders relates to the extent of cortical ChAT loss Controls PSP 100 90 PD 80 VaD 70 60 50 40 PDD AD DLB 30 20 10 0 0 10 20 30 40 50 Rate of hallucinations Inferior temporal cortex 60 picture of hallucination by artist with PD In DLB, more reduced ChAT is associated with visual hallucinations ChAT activity in temporal cortex ChAT nmol/hr/mg protein 4 3 Presence of VH is good predictor of response to ChEI p=0.02 2 1 12 5 +VH -VH 0 DLB with and without visual hallucinations Hallucinations related to nicotinic receptors in DLB Imaging – • reduced 5IA85480 binding to a42 nicotinic receptors in DLB in striatum and frontal, temporal and cingulate cortex • Increased a42 in occipital cortex associated with hallucinations Fluctuations related to nicotinic receptors in DLB • Temporal cortex nicotinic receptor a42 reduced in DLB/PDD • Greater reduction in cortex and thalamus in cases without fluctuations Temporal cortex In an environment of reduced cholinergic activity, a higher density of nicotinic receptors could amplify small transmitter changes leading to variations in consciousness and attention 3H epibatidine fmol/mg 4 3 2 1 16 6 0 +FC -FC Fluctuations impair ADL and are over seconds, minutes, hours, and days Dopamine in DLB Dopamine concentration and dopamine transporters are reduced in DLB, almost to the same extent as in Parkinson’s disease Control Alzheimer DLB no EPS DLB + EPS Autoradiographs of dopamine transporter Dopamine transporters in PD, PDD, DLB±EPS, and AD 125I PE2I binding fmol/mg posterior caudate posterior putamen 1.0 1.0 0.8 0.8 0.6 0.6 0.4 0.4 0.2 0.2 0.0 0.0 Control Significant loss even in DLB with no EPS – support for FP-CIT SPECT (DaTSCAN) in AD/DLB discrimination PD no dementia PDD DLB+EPS DLB no EPS AD Striatal D2 receptors in PD, DLB and AD Control PD DLB [3H] raclopride fmol/mg caudate 50 putamen 17 12 40 14 26 30 40 20 20 10 10 0 0 DLB 27 8 30 8 controls PD 15 50 AD controls PD DLB AD Cortical D2 receptors reduced in DLB and PDD 20/21 22 Ent cx normal 36 20 36 36 22 21 nsb 20 20 21 DLB/PDD 22 22 22 Ent cx Ent cx Ent cx 21 21 36 21 20 36 36 20 20 40% reduction in DLB (30% in PDD) in D2 receptors in temporal cortex; no change in AD 125I epidepride binding fmol/mg Temporal cortex D2 decline with MMSE 0.7 DLB and PDD, Ba 20 N=20, r=0.58, p=0.008 0.6 0.5 0.4 Consistent with 0.3 • Neuroleptics impair cognition • D2 PET in hippocampus 0.2 correlates with memory 0.1 DLB PDD 0.0 0 5 10 15 MMSE 20 25 30 Thalamic D2 receptors elevated in PD (~50%) compared to controls and other disease groups parafascicular ventral area centromedian 8 7 8 centromedian 6 6 4 4 6 5 reticular nucleus u 4 2.0 2 2 2 0 0 1.0 0.5 1 0 1.5 3 10 3 9 9 5 7 3 8 8 4 7 3 8 8 0.0 4 12 6 11 11 5 ventroposterior 8 laterodorsal nucleus 5 paraventricular nucleus 6 4 12 4 u 3 u control 2 2 10 PD no dementia 1 0 8 0 12 12 10 6 MD 7 6 5 4 3 2 1 0 0 11 6 8 9 4 6 7 3 PDD DLB - EPS 5 2 5 DLB + EPS 6 4 7 12 5 9 10 5 Raised D2 in DLB/PDD with fluctuations in cortex and in thalamic nuclei with a role in maintenance of consciousness 8 with DOC parafascicular 125I epidepride fmol/mg without DOC 6 centromedian 6 5 reticular nucleus 4 1.5 4 3 1.0 0.5 2 2 0.0 12 1 5 0 0 10 6 with DOC without DOC mediodorsal 9 6 D2 1.00 cingulate cortex MD 4 0.75 3 2 0.50 1 0.25 0 10 5 reticular CM/pf 0.00 6 5 Dopamine mechanisms • Elevated D2 receptors in PD - compensates for low dopamine • Reduced D2 receptors in DLB and PDD may correlate with poor levodopa response and neuroleptic sensitivity D2 receptors decline as PD progresses faster in cortex than striatum and thalamus D2 receptors are on GABA interneurons i.e. inhibiting inhibitory neurons - a higher density of D2 receptors will amplify small transmitter changes Glutamate markers in AD – inconsistent reports Reduced NMDA binding and NMDAR1 mRNA expression in AD Cortical pyramidal neurone loss leads to reduced glutamate activity and cognitive impairment in AD With reduced NMDA receptors in AD, odd that NMDA antagonist memantine effective - it blocks NMDA receptor better than Mg2+ But reduced membrane potential (due to pathology, reduced energy metabolism) leads to release voltage dependent Mg2+ block of NMDA → and excessive, neurotoxic entry of Ca2+ So Memantine efficacy in moderate-severe AD with heavier pathology • acting as uncompetitive, low-affinity, open-channel blocker • limiting excessive glutamate • reducing signal to noise Memantine is also a D2 agonist, 5HT3 antagonist Serotonergic abnormalities • neurone loss & tangles in raphe, reduced 5HT • relatively retained 5-HT function linked to more psychosis (AD and DLB) • 5-HT2A receptors more reduced with severe dementia • 5HT receptor polymorphisms linked to Aggression, Psychosis, Depression, Anxiety Noradrenergic Abnormalities • Extensive neuron loss locus coeruleus, reductions in noradrenaline, increased turnover in surviving neurons linked to upregulation of the noradrenaline transporter • In PD noradrenaline loss linked to → PDD • Noradrenaline changes may be related to Aggression, Psychosis, Depression Fronto-Temporal Dementia Younger onset (45 – 60 years) Pathology most apparent in the II and deep cortical layers, coinciding with location of D2 and 5HT1 receptors Neurotransmitter losses Serotonin – concentration and transporters reduced, 5HT1A and 5HT2A receptors reduced Compulsive behaviours, sweet and carbohydrate consumption Dopamine – concentration and transporters reduced, D2 receptors elevated in striatum Rigidity, flat facies, depression Norepinephrine and some neuropeptide transmitters – slight reduction Anxiety, suspiciousness, restlessness Acetylcholine – little or no reduction greater imbalance DA/ACh in striatum may exacerbate EPS GABA, glutamate - unchanged Cholinergic Therapy - Residual receptor availability Cholinesterase inhibitors delusions, hallucinations, agitation, aggression, anxiety, apathy, as well as cognition (implying cholinergic mechanisms) Galantamine (Reminyl, or Razadyne) AChEI and nicotinic receptor allosteric modulator Donepezil (Aricept) AChEI Rivastigmine (Exelon) AChEI and BuCHEI Why might DLB Patients respond to Cholinergic Treatment? • Cortical muscarinic receptors up-regulated • M1 receptors remain coupled to G-proteins (unlike AD) • ACh very reduced • Less neuron loss or cortical atrophy • Little or no tangle burden • Symptoms fluctuate 150 100 M1 potential for higher function to be restored 50 • Low M1 receptors in striatum 0 0 10 avoids worsening parkinsonism • AChEI only inhibit 30% AChE activity 20 30 40 50 Striatal D2 DLB PDD Control AD PD 60 Cholinergic and dopaminergic influence and consequences Neuronal survival Alzheimer pathology Cognitive impairment See table of anticholinergic medications – many regularly used by the elderly. Implications – Anticholinergic Medication Use and Cognitive Impairment in the Older Population: The MRC Cognitive Function in Ageing Study. Fox et al JAGS 2011 Smoking (and coffee drinking) inversely associated with PD, not with AD (most studies) Nicotine use (tobacco) associated with lower plaque densities in normal elderly Normal elderly (female) smokers and non-smokers CHOLINERGIC TRANSMISSION Reduces Alzheimer-type pathology • Muscarinic M1 Agonists reduce A levels in CSF in AD • In triple-Tg-AD mouse, M1 agonist AF267B rescued cognitive deficits and reduced A and tau pathology (dicyclomine M1 antagonist) • Cholinesterase inhibitors may reduce amyloid Reviews Fisher A., Neurotherapeutics: 5 2008, 433-442 Caccamo A., Current Alzheimer Research. 6 2009:112-7 Alzheimer pathology increased in PD in relation to antimuscarinic drugs SENILE PLAQUES NEUROFIBRILLARY TANGLES p=0.005 compared to no drug P=0.02 compared to no drug 4.5 4 1.6 3.5 1.4 3 1.2 2.5 1 2 0.8 1.5 0.6 1 0.4 0.5 0 21 15 NO DRUG ACUTE 18 CHRONIC 1.8 0.2 0 21 NO DRUG 15 ACUTE 18 CHRONIC acute <2y, chronic 2-18y Anticholinergics: benztropine, orphenadrine, trihexyphenidyl, oxybutynin Groups matched for age and PD duration NEUROLEPTIC MEDICATION IS ASSOCIATED WITH INCREASED TANGLE DENSITY IN DLB/PDD anterior cingulate cortex 5 1.4 4.5 Tangle density frontal cortex 1.2 4 3.5 1 3 0.8 *p=0.04 2.5 2 0.6 1.5 0.4 1 0.2 0.5 0 0 - NL (23) + NL (17) - NL (23) + NL (17) DLB/PDD matched for age, duration of PD, duration of dementia, MMSE, prevalence of delusions and visual hallucinations Cognitive and Neuropsychiatric Symptoms in dementia Can Cholinergic and Dopaminergic Mechanisms Explain All? Not quite – glutamate, serotonin and noradrenaline also important other influences need elucidation