* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Fast and Flexible Single Nucleotide Polymorphism (SNP) Detection
Vectors in gene therapy wikipedia , lookup
Gene therapy wikipedia , lookup
Genetic engineering wikipedia , lookup
Human genetic variation wikipedia , lookup
Genome evolution wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
Tay–Sachs disease wikipedia , lookup
Population genetics wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
History of genetic engineering wikipedia , lookup
Saethre–Chotzen syndrome wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Oncogenomics wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Genome (book) wikipedia , lookup
Genome editing wikipedia , lookup
Genome-wide association study wikipedia , lookup
Designer baby wikipedia , lookup
Neuronal ceroid lipofuscinosis wikipedia , lookup
Microsatellite wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Public health genomics wikipedia , lookup
Microevolution wikipedia , lookup
Molecular Inversion Probe wikipedia , lookup
Frameshift mutation wikipedia , lookup
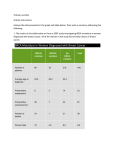
Fast and Flexible_1.qxd 28.08.2000 12:55 Uhr Seite 23 Fast and Flexible Single Nucleotide Polymorphism (SNP) Detection with the LightCycler System S. Lohmann, L. Lehmann, and K. Tabiti Roche Molecular Biochemicals, Penzberg, Germany ntroduction One of the major objectives of genetics is the association of sequence variations with heritable phenotypes. Traditional strategies, such as linkage analysis, in which pedigree analysis track transmission of a disease through a family, have been successfully applied to in the detection of Mendelian disorders. In recent years a more powerful approach involving the detection of single nucleotide polymorphisms (SNPs) has become increasingly popular. By convention, a nucleotide polymorphism must be present in at least one percent of the human population to be called an SNP. SNPs are the most common type of DNA sequence variations and occur once every 100-300 bases. Researchers looking for associations between a disease and specific sequence differences in a population, use this high degree of variation. As it is easier to obtain DNA samples from a random set of individuals in a population, than from every member of a family over several generations, it is conceivable that researchers may be able to increase the identification of genes responsible for pathological traits. Due to the high frequency of SNPs, the chances are high that the disease is predominantly caused by, or closely associated with, specific SNPs. SNPs present a potentially vast arena for the detection of genetic alterations that seem to relate to medically important differences in disease susceptibility and drug response. In the near future it may be possible to identify the genetic basis for complex diseases such as cancer, cardiovascular disease, mental illness, autoimmune states, and diabetes. The number of polymorphisms known to predispose for a disease is believed to increase exponentially. Yet even currently known disease-predisposing polymorphisms may benefit from our expanding knowledge: "old polymorphisms” may be relevant in a novel context. For example, the E4 genotype of the human apolipoprotein E gene has been traditionally associated with lipid disorders. Many recent reports have obser- ROCHE MOLECULAR BIOCHEMICALS ved a relationship between the Apo E genotype and the susceptibility to Alzheimer's disease. Even more recently, scientists have reported that polymorphisms of the apolipoprotein E gene may influence the outcome after traumatic brain injury and intracerebral hemorrhage [1]. Thus, one particular haplotype is informative for several traits. Besides the above-mentioned common diseases in humans, the response to medication in terms of both adverse reactions and beneficial response is closely regulated by a plenitude of genes. This understanding is fueling many pharmaceutical companies' interest in generating SNP libraries, with a view to detecting genes involved in drug metabolism. One of the most predominant examples of such an effort is the SNP Consortium (TSC), a joint nonprofit venture of ten pharmaceutical companies with the aim of creating and releasing to the public a library of some 300 000 SNPs [2]. LIGHTCYCLER I The possibility of using DNA sequence information to distinguish those individuals who are likely to benefit from a new medication from those who could suffer adverse side-effects, or to determine the optimal dosage, has rekindled the interest in the research area termed pharmacogenetics. The ideas underlying pharmacogenetics are not new: for decades it has been understood that genes affect the interindividual variation in drug response. The concerted actions in detecting and analyzing SNPs may now open new avenues for personalized medicine, which should improve therapeutic outcome, reduce overall healthcare costs and ultimately benefit the individual. Besides these SNPs involved in drug metabolism, specific SNPs are associated with a higher risk of disease (i.e. disease associated SNPs). Knowledge of these SNPs is important for the understanding of processes in disease development and the role and function of the genes involved. A future vision is to use this information for improved diagnosis, disease prevention and causal therapy. This article gives an overview describing how the LightCycler Instrument can be applied in genotyping BIOCHEMICA · No. 4 m 2000 23 Fast and Flexible_1.qxd 28.08.2000 12:55 Uhr Seite 24 SNPs in less than 40 min for 32 research samples and refers to the most recent publications and LightCycler Kits in these exciting research areas. S NP Detection using the LightCycler System The LightCycler Instrument enables both the amplification and the real-time, on-line detection of a PCR product, thus allowing accurate quantification. Furthermore, the system also provides a unique and innovative approach perfectly suited for the detection and genotyping of single nucleotide polymorphisms: the melting curve analysis feature. During the melting curve analysis the LightCycler Instrument monitors the temperature-dependent hybridization of the sequencespecific Hybridization Probes to single stranded DNA. hn LIGHTCYCLER A LC-Red 640 hn Fluorescein mutation probe 1 anchor probe 1 LC-Red 705 Fluorescein mutation probe 2 anchor probe 2 primer 5‘ 3‘ codon 112 mutation site codon 158 mutation site 3‘ 5‘ B primer The genotype of codon 112 is monitored in the F2 detection channel of the LightCycler Instrument. Fluorescence -d(F2)/dT 4 Homozygous (CGC) Heterozygous Homozygous (TGC) H2O 3 2 1 0 -0.5 45.0 50.0 55.0 60.0 Temperature (°C) 65.0 70.0 75.0 Fluorescence -d(F3)/dT The genotype of codon 158 is monitored in the F3 detection channel of the LightCycler Instrument. 3.2 2.8 2.4 2.0 1.6 1.2 0.8 0.4 0.0 Homozygous (CGC) Heterozygous Homozygous (TGC) H2O 45.0 Ý 50.0 55.0 60.0 Temperature (°C) 65.0 70.0 75.0 Figure 1: Dual color SNP genotyping example using the LightCycler-Apo E Mutation Detection Kit (Cat. No. 3 004 716). A. Schematic presentation of the PCR fragment, and the ori- entation of the PCR primers, the anchor probes, and the mutation probes.B. Discrimination of the different genotypes at codon 112 and 158 of the Apoliprotein E sequence with the melting curve analysis function. The first negative derivative of the fluorescence versus temperature [-d(F2)/dT] or [-d(F3/dT] graph shows peaks with different T m. The melting peaks indicate that the fully homologous sequence (homozygous CGC) has a higher Tm than the sequences that have a mismatch with the mutation probe (homozygous TGC). Samples containing both sequences (heterozygous genotypes) display two peaks at exactly the same temperatures as the respective homozygous samples. As a negative control, the template DNA was replaced with PCR-grade water. 24 BIOCHEMICA · No. 4 m 2000 No post-PCR processing is needed and the risk of contamination is minimized, as amplification and genotyping are performed in the same sealed capillary without any further handling steps. In brief, during the PCR, a DNA fragment of the respective gene is amplified with specific primers from human genomic DNA. The amplicon is detected by fluorescence using specific pairs of Hybridization Probes. Hybridization Probes consist of two different oligonucleotides that hybridize to an internal sequence of the amplified fragment during the annealing phase of PCR cycles. One probe is labeled at the 5’-end with a LightCycler–Red fluorophore (LightCycler-Red 640 or LightCycler-Red 705) and, to avoid extension, is modified at the 3’-end by phosphorylation. The other probe is labeled at the 3’-end with fluorescein. Only after hybridization to the template DNA do the two probes come in close proximity, resulting in fluorescence resonance energy transfer (FRET) between the two fluorophores. During FRET, fluorescein, the donor fluorophore, is excited by the light source of the LightCycler Instrument, and part of the excitation energy is transferred to LightCycler-Red, the acceptor fluorophore. The emitted fluorescence of the LightCycler-Red fluorophore is measured. These Hybridization Probes are also used to determine the genotype by means of a melting curve analysis implemented after the amplification cycles are completed and the amplicon is formed. The melting temperature (Tm) of the heteroduplex consisting of Hybridization Probe and single-stranded target DNA sequence is dependent on GC content, length, degree of homology and sequence order [3]. One of the two Hybridization Probes covers the region of the potential mutation (mutation probe) and has a lower Tm than the adjacent probe (anchor probe) (Figure 1). Hybridization Probe/DNA hybrids containing a mismatch melt at a lower Tm than perfectly matched probes. Hence, wildtype, mutant and heterozygous genotypes can be distinguished by different melting temperatures, displayed in the LightCycler software as melting peaks. The time required to genotype SNPs is reduced to less than 40 min for 32 research samples, while eliminating post PCR-processing and minimizing the risk of contamination (Figure 1). The principle of using Hybridization Probes and melting curving analysis to detect specific mutations and SNPs has been described in detail elsewhere [4,5]. For more detailed information please visit our website http://www.biochem.roche.com/lightcycler ROCHE MOLECULAR BIOCHEMICALS 28.08.2000 12:55 Uhr Seite 25 A Technical Note describing different concepts and strategies for mutation detection using the LightCycler Instrument is available on request (TN 14/2000: General Recommendations for LightCycler Mutation Analysis Using Dual Color and Melting Temperature). A pplication fields Pharmacogenetics SNPs and other DNA variations in drug metabolizing enzymes are related to side effects, incompatibility and variations in therapeutic efficacy. The human cytochrome P-450 (CYP) supergene family represents an important group of enzymes involved in the metabolite activation of procarcinogenesis to produce potentially carcinogenic metabolites. A rapid and reliable method has been established for genotyping the CYP1B1 codon 432-polymorphisms using the LightCycler Instrument [6]. Variability in drug acetylation is due to geneticallycontrolled expression of the N-acetyltransferase 2 enzyme (NAT2). Several polymorphisms in the NAT2 gene are responsible for a slow acetylator phenotype. The LightCycler-NAT2 Mutation Detection Kit (Cat. No. 3 113 914) from Roche Molecular Biochemicals (RMB) allows the simultaneous detection of the 4 major polymorphisms in the NAT2 gene, using two primer pairs to amplify and cover the different polymorphisms with four different Hybridization Probe pairs. Dihydropyrimidine dehydrogenase (DPD), thymidylate synthase (TS) and thymidine phosphorylase (TP) are involved in fluoropyrimidine metabolism and thus regulate levels of 5-FU, a drug used in chemotherapy. Several point mutations in these enzymes are associated with expression levels of these enzymes and are consequently related to therapeutic success. Dedicated LightCycler kits for analyzing these polymorphisms are under development with a view to supporting research in this field. Metabolic disorders The ability to use the LightCycler System for the rapid detection of deletion mutations in inherited metabolic diseases has recently been demonstrated in research studies for two different parameters [7]. Fabry disease is an X-linked recessive disorder caused by deficient activity of a-galactosidase. Using Hybridization Probes and melting curve analysis, a 2-bp deletion mutation in genomic DNA of an individual with Fabry disease has successfully been detected. In addition, the aut- ROCHE MOLECULAR BIOCHEMICALS hors were able to detect a 9-bp deletion in an individual with carbamyl-phophate synthase I (CPS1) deficiency using the SYBR Green I detection format and melting curve analysis. CPS1 deficiency is an autosomal recessive disorder caused by deficient activity of CPS1 in the liver. This study demonstrates, for the first time, the detection of a 9-bp deletion using the SYBR Green I detection format. With this latter format the Tm is influenced primarily by GC content, and to a lower extent by size. Two point mutations (Cys282Tyr, His63Asp) in the human hemochromatosis gene (HFE) are considered to be responsible for the development of hereditary hemochromatosis (HH), an autosomal recessive ironloading syndrome. The homozygous Cys282Tyr mutation is present in 80-100% of hemochromatosis cases [8]. The authors used a multiplex PCR protocol to amplify the relevant locations. Two sets of Hybridization Probes, both labeled with LC Red 640, were used for the genotyping of the SNPs. As shown by Bernhard et al., it is also possible to use a Dual Color approach to detect both mutations [9]. A recently described third mutation, S65C, is enriched in HH individuals with a mild form of the disease and with no mutations at C282 or H63. Bollhalder et al. published a LightCycler protocol for the simultaneous detection of the H63D and S65C mutations of the HFE gene [10]. One primer and probe set can be used to detect both sites, as the distance between them is just 6 nucleotides (A193T, C187G). One mutation probe was designed to be complementary to the 63D mutant and the S65 wildtype. Therefore, 65C mutant melts off at the lowest melting temperature (Tm), followed by the H63 wildtype and the 63D mutant allele showing the highest Tm. LIGHTCYCLER Fast and Flexible_1.qxd Protease inhibitor 1 (a1-antitrypsin; AT) is the main serum inhibitor of proteolytic enzymes. In AT deficiency, enzymes such as neurophil elastase can damage the lung tissues, leading to pulmonary emphysema. The 3 most important types are: type M (90% of the population), type S (Pi*S), and type Z (Pi*Z) [11]. A pair of primers flanking each of the mutations in the Pi*S and Pi*Z alleles was designed to amplify a 238 bp and a 253 bp product. Hybridization Probe pairs homologous to the wildtype sequence were used for genotyping [12]. Disease-associated applications The most common causes of inherited thrombophilia in the Caucasian population are point mutations in the Factor V gene and the Prothrombin gene (Factor II). BIOCHEMICA · No. 4 m 2000 25 Fast and Flexible_1.qxd 28.08.2000 12:55 Uhr Seite 26 LIGHTCYCLER The C677T point mutation in the methylenetetrahydrofolate reductase (MTHFR) gene is also associated with a higher risk. Over 90% of individuals with a resistance to activated protein C (APC-resistance), a condition associated with thrombophilia, are associated with hetero- or homozygosity for a single point mutation in the Factor V gene. This G to A substitution in exon 10 (G1691A), also known as Factor V Leiden, leads to an arginine substituted by glutamine (R506Q) and prevents cleavage by activated protein C that normally inactivates the coagulation factor. A respective LightCycler research kit is available from Roche Molecular Biochemicals: the LightCycler Factor V Leiden Mutation Detection Kit (Cat. No. 2 212 161). A methodological comparison between allele-specific PCR (PCR-SSP) and the LightCycler technology in the genotyping of the Factor V Leiden mutation in research samples was recently published [13]. The results generated with both methods showed 100% correlation. For prothromobin, a G to A substitution (G20210A) in the 3´untranslated region of the human prothrombin gene leads to a substitution of argine by glutamine. This polymorphism is associated with an elevated level of plasma prothrombin. The approach for detecting this SNP is identical to the Factor V assay. A dedicated RMB research kit is available for the LightCycler, i.e. the LightCycler Prothrombin Mutation Detection Kit (Cat. No 2 236 842). An additional independent risk factor for thrombophilia is hyperhomocysteinemia. A point mutation in the MTHFR gene results in reduced MTHFR activity. Elevated plasma homocysteine levels are the consequence. Research papers on the genotyping of the relevant point mutation (C677T) in the responsible MTHFR gene with the LightCycler Instrument have been published [14, 15]. The role of apolipoprotein E (Apo E) in lipid disorders has already been mentioned. The LightCycler – Apo E Mutation Detection Kit from RMB (Cat. No. 3 004 716) can be used for the simultaneous detection of two point mutations within codons 112 and 158 of the human apolipoprotein E gene using a dual color approach (Figure 1). The Hybridization Probe mix provided with the kit includes one LC Red 640 labeled Hybridization Probe for detecting the nucleotide polymorphism C3932T, while a second Hybridization Probe labeled with LC Red 705 specifically detects the possible C4070T substitution. A recently published evaluation report shows 100% concordance for the data obtained for 120 DNA samples with allele specific restriction enzyme analysis (ASRA) and genotyping with the LightCycler-Apo E Mutation Detection Kit, representing a genotype distribution typical for the Caucasian population [16]. 26 BIOCHEMICA · No. 4 m 2000 The respective LightCycler kit from RMB for human apolipoprotein B (Apo B) is intended as a research tool to help further understand the relevance of the Apo B gene in diseases like familial hypercholesterolemia, atherosclerosis, and ischemic heart disease (Cat. No. 3 004 708). Two relevant point mutations in the Apo B gene are located in directly adjacent positions: C9774T, G9775A. Only one Hybridization Probe can be used to detect these nucleotide polymorphisms since the C9774T mutation has a smaller impact on Tm than the G9775A, resulting in different Tms. A detailed description of the different detection principles used for prothrombin, apolipoprotein B and apolipoprotein E has been published [17]. Monogenic hereditary disease Cystic fibrosis (CF) is one of the most common autosomal recessive disorders among the Caucasian population, with a frequency of 0.05% and a carrier frequency of 5%. Various disease causing polymorphisms are associated with CF. The most common mutation is a 3bp deletion that removes a phenylalanine from the protein at codon 508 (F508del). This deletion accounts for 70% of all mutations, and 50% of individuals with CF are homozygous for this mutation. Amplification and genotyping with the LightCycler Instrument was achieved in less than 30 min. A total of 105 samples were genotyped for the deletion F508del. Furthermore, a single nucleotide polyphorphism leading to the F508C variant was detected [18]. Polygenic hereditary disease The majority of disorders such as allergies, diabetes or cancer are related to a distinct polygenic background. The individual genetic background may be associated with a higher risk of cancer development, but complex events occur during carcinogenesis and tumor progression. Point mutations in different members of the ras gene family are involved in a variety of human malignancies. N- ras mutations are frequently observed in hematological malignancies. A rapid and reliable strategy for detecting N- ras mutations in acute lymphoblastic leukemia (ALL) has recently been published [19]. The authors designed two sets of primers and Hybridization Probes to cover ‘hot spot’ regions for mutations within codons 12, 13, and 61. Amplicons exhibiting an abnormal melting characteristic were directly sequenced. 134 samples from individuals with childhood ALL were screened for N- ras mutations and 14 mutation-positive cases characterized in less than three days, revealing an incidence for N- ras mutations of 10%, i.e. compatible with previous reports. ROCHE MOLECULAR BIOCHEMICALS 28.08.2000 12:55 Uhr Seite 27 Infectious disease Helicobacter pylori colonizes the human stomach, inducing chronic gastritis and playing a major role in gastric and duodenal ulceration. Clarithromycin is frequently used in macrolide antibiotic therapies. Resistance to clarithromycin is associated with single base pair mutations within the peptidyltransferaseencoding region of the 23S rRNA gene. Gibson et al. use the SYBR Green I detection format in combination with a single labeled Hybridization Probe to monitor these polymorphisms using melting curve analysis. The authors were able to detect the three most relevant SNPs using this approach [20]. C onclusions There are numerous examples for SNP detection in research applications. In each case a LightCycler approach can be developed to perform these assays. Table 1 summarizes the LightCycler Kits currently available from RMB and those applications that have already been published. The renewed and extensive interest in genome polymorphism will have a major impact upon population genetics, drug development, forensic, cancer and genetic disease research. The optimal genotyping method should allow the allele determination of SNPs rapidly and economically. The LightCycler System fulfills these demands and is the perfect tool for reliable and accurate genotyping of single nucleotide polymorphisms. Application area Associated gene Reference / LightCycler Kit Pharmacogenetics Cytochrome P450: CYB1B1 [6] Cytochrome P450: CYP2D6 N-Acetyltransferase 2 (NAT2) [21] Cat.No. 3 113 914 Metabolic disorders Disease-associated Monogenic hereditary disease Polygenic hereditary disease Infectious disease others Thiopurine methyltransferase (TPMT*3) [22] Carbamyl-phosphate synthase (CPS1) [7] Fabry disease [7] Hemochromatosis (HFE) [8, 9, 10, 23] Protease inhibitor 1 (a1-antitrypsin; AT) [11, 12] Angiotensin converting enzyme (ACE) [24] Apolipoprotein B (Apo B) Cat. No. 3 004 708 Apolipoprotein E (Apo E) Cat. No. 3 004 716 Factor V Cat. No. 2 212 161 Methylenetetrahydrofolate reductase (MTHFR) [14, 15] Mitochondrial DNA (MELAS3243) [25] Plasma plasminogen activator inhibitor-1 (PAI-1) [26] Prothrombin (Factor II) Cat. No. 2 236 842 Cystic fibrosis (CF) [18] b-Globin (HbC, HbE, and HbS genotyping) [27] BRCA1 [28] N-ras [19] Glycoprotein Ia [29] Clarithromycin-resistance associated gene mutations [20] Lamivudine resistance associated gene mutations [30] Human platelet antigen-1 (HPA-1) [31] LIGHTCYCLER Fast and Flexible_1.qxd Note: LightCycler kits listed are for life science research only. Ý Table 1 Summary of research application areas and published literature or respective LightCycler Kit. ROCHE MOLECULAR BIOCHEMICALS BIOCHEMICA · No. 4 m 2000 27 Fast and Flexible_1.qxd 28.08.2000 12:55 Uhr Seite 28 References LIGHTCYCLER [1] Mc Carron, M.O., Muir, K. W., Weir, C. J., Dyker, A. G., Bone, I., Nicoll, J.A.R., and Lees, K.R. (1998) Stroke 29: 1882-1887. [2] Marshall, E. (1999) Science 284: 406-407. [3] SantaLucia, J. Jr., Allwai, H.T., and Seneviratne, P.A. (1996) Biochemistry 35: 3555-3562. [4] Reiser, A., Geyer, M., van Miltenburg, R., Nauck, M., and Tabiti, K.(1999) Biochemica 2: 12-15. [5] Bernard, P.S. and Wittwer, C.T. (2000) Clin. Chemistry 46: 147-148. [6] Brüning, T., Abel, J., Koch, B., Lorenzen, K., Harth, V., Donat, S., Sachinidis, A., Vetter, H., Bolt, H.M., and Ko, Y. (1999) Arch Toxicol 73: 427-430. [7] Aoshima, T., Sekido, Y., Miyazaki, T., Kajita, M., Mimura, S., Watanabe, K., Shimokata, K., and Niwa, T. (2000) Clin. Chemistry 46: 119-122. [8] Mangasser-Stephan, K., Tag, C., Reiser, A., and Gressner, A.M. (1999) Clin. Chemistry 45: 1875-1878. [9] Bernard, P.S., Ajioka, R.S., Kushner, J.P., and Wittwer, C.T. (1998) AJP 153: 1055-1061. [10] Bollhalder, M., Mura, C., Landt, O., and Maly, F.E. (1999) Clin. Chemistry 45: 2275-2278. [11] Aslanidis, C., Nauck, M., and Schmitz, G. (1999) Clin. Chemistry 45: 1872-1875. [12] Aslanidis, C. and Schmitz, G. (2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [13] Schlatterer, K., Orth, M., Fitzner, R., and Tauber, R. (2000) Biochemica 1: 31-34. [14] Bernard, P.S., Lay, M.J., and Wittwer, C.T. (1998) Anal. Biochem. 255: 101-107. [15] Aslanidis, C. and Schmitz, G. (2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [16] Nauck, M., Hoffmann, M.M., Wieland, H., and März, W. (2000) Clin. Chemistry 46: 722-724. [17] Biochemica 1 (2000): 29-30. [18] Gundry, C. N., Bernard, P.S., Herrmann M.G., Reed, G.H., and Wittwer, C.T. (1999) Genetic testing 3(4): 365-370. [19] Nakao, M., Janssen, J.W.G., Seriu, T., and Bartram, C.R. (2000) Leukemia 14(2): 312-315. [20] Gibson, J.R, Saunders, N.A., Burke, B., and Owen, R.J. (1999) J. Clin. Microbiol. 37: 3746-3748. [21] Bjerke, J., Schur, C., Wong, S., Nuwayhid, N., and Chang, C.C. (2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [22] Schütz, E. and von Ahsen, N. (2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [23] Kyger, E.M., Krevolin, M.D., and Powell, M.J. (1998) Anal. Biochem. 260: 142-148. [24] Sakai, E., Tajima, M., Mori, M., Inage, R., Fukumoto, M., and Nakagawara, K.(2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [25] Kleinle, S., and Liechti-Gallati, S. (2000) Rapid Cycle RealTime PCR Methods and Applications. Springer Verlag, in press. [26] Nauck, M., Wieland, H., and März, W. (1999) Clin. Chemistry 45: 1141-1147. [27] Hermann, M.G., Dobrowolski, S.F., and Wittwer, C.T. (2000) Clin. Chemistry 46: 425-428. 28 BIOCHEMICA · No. 4 m 2000 [28] Pals, G, Pinolda, K., and Worsham, M.J. (1999) Mol. Diagn. 4: 241-246. [29] Morita, H., Kurihara, H., Nagai, R., and Yazaki, Y.(2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. [30] Cane, P.A., Cook, P., Ratcliffe, D., Mutimer, D., and Pillay, D. (1999) Antimicrob. Agents Chemother. 43: 1600-1608. [31] Nauck, M. S., Gierens, H., Nauck, M.A., März, W., and Wieland, H. (2000) Rapid Cycle Real-Time PCR Methods and Applications. Springer Verlag, in press. Product Cat. No. Pack Size LightCycler- NAT2 Mutation Detection Kit 3 113 914 32 reactions LightCycler-Apo B Mutation Detection Kit (codon 3500) 3 004708 32 reactions LightCycler-Apo E Mutation Detection Kit (codon 112 and 158) 3 004716 32 reactions LightCycler Factor V Leiden 2 212161 Mutation Detection Kit 32 reactions LightCycler-Prothrombin (G20210A) Mutation Detection Kit 2 236842 32 reactions LightCycler Red 640 NHS ester 2 015 161 1 vial LightCycler Red 705 Phosphoramidite 2 157 594 1 vial ROCHE MOLECULAR BIOCHEMICALS