* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download TBI Abstract - Stacey Lee, PhD
Psychoneuroimmunology wikipedia , lookup
Intracranial pressure wikipedia , lookup
Neuroeconomics wikipedia , lookup
Subventricular zone wikipedia , lookup
Nervous system network models wikipedia , lookup
Biochemistry of Alzheimer's disease wikipedia , lookup
Human brain wikipedia , lookup
Neuroinformatics wikipedia , lookup
Neurophilosophy wikipedia , lookup
Cognitive neuroscience wikipedia , lookup
Brain morphometry wikipedia , lookup
Molecular neuroscience wikipedia , lookup
Blood–brain barrier wikipedia , lookup
Holonomic brain theory wikipedia , lookup
Neurolinguistics wikipedia , lookup
Selfish brain theory wikipedia , lookup
History of neuroimaging wikipedia , lookup
Activity-dependent plasticity wikipedia , lookup
Neuroregeneration wikipedia , lookup
Brain Rules wikipedia , lookup
Aging brain wikipedia , lookup
Neuroanatomy wikipedia , lookup
Neuropsychology wikipedia , lookup
Clinical neurochemistry wikipedia , lookup
Neuroplasticity wikipedia , lookup
Haemodynamic response wikipedia , lookup
Metastability in the brain wikipedia , lookup
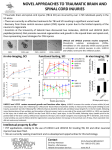
NEUROPROTECTIVE EFFICACY OF PURINERGIC RECEPTOR AGONISTS IN VITRO AND IN WHOLE ANIMALS AFTER TRAUMATIC BRAIN INJURY Stacey Lee, PhD and Jim Lechleiter, PhD Department of Cellular and Structural Biology, UTHSCSA Traumatic brain injuries (TBIs) are the cause of over 30% of injury-related deaths in the United States. Both civilians and military personnel are at risk for TBIs from blunt force or blast trauma. Side effects of TBI can range from dizziness, nausea, headaches, concentration problems, memory impairment, mood disorders, and seizures. On a molecular level, there are two phases of injury after a TBI. The primary phase is the initial physical trauma. The secondary phase occurs within the following days of the injury leading to edema, excitotoxicity, neuronal damage, cell death, inflammatory responses, and blood-brain barrier dysfunction. Currently, there is no FDA-approved treatment for TBI despite an urgent need for one. The goal of our work is to characterize novel therapeutic compounds to mitigate brain damage induced by TBI. The project aims to mitigate injury responses by improving astrocyte energy metabolism. Astrocytes are essential for maintaining neuronal function and homeostasis in the brain. They provide neurons with metabolic support, modulate synaptic transmission, and aid in repair after injury. Previous research from the lab has shown that enhancing mitochondrial metabolism in astrocytes protects neurons from oxidative stress. Specifically, treating astrocytes with a purinergic receptor agonist enhances their ATP production and decreases cytotoxic edema and reactive gliosis. We hypothesize that enhancing astrocyte metabolic activity with these agonists will protect the brain against TBI-induced damage. We have two mechanisms for generating TBIs: a pneumatic impact device that generates a closed controlled cortical impact, or blunt trauma, and an air-driven shock tube simulating an IED explosion, or blast trauma. Using these two methods, we will asses the ability of our compounds to reduce astrocyte injury response, injury size, edema, inflammatory response, behavior, and seizures. We predict agonist treatment after injury will prevent the secondary damage responses from exacerbating the injury and lead to an improved outcome on a molecular and behavioral level. This work is supported by the Department of Defense grant number W81XWH-15-1-0283.