* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Poster
Endomembrane system wikipedia , lookup
Magnesium transporter wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Cell growth wikipedia , lookup
Protein moonlighting wikipedia , lookup
Protein structure prediction wikipedia , lookup
Protein (nutrient) wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Signal transduction wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Cytokinesis wikipedia , lookup
Paracrine signalling wikipedia , lookup
Protein–protein interaction wikipedia , lookup
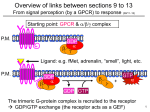
H-Ras GTPase Key to Understanding Cancer? Marquette University High School SMART Team: Mohammed Ayesh, Wesley Borden, Andrew Bray, Brian Digiacinto, Patrick Jordan, David Moldenhauer, Thomas Niswonger, Joseph Radke, Amit Singh, Alex Vincent, and Caleb Vogt Teachers: Keith Klestinski and David Vogt Mentor: Evgenii Kovrigin, Ph.D., Medical College of Wisconsin Abstract Cell Cycle Control H-ras is activated late in the G1 phase. Once H-ras is activated, the cell advances past the G1 checkpoint and is compelled to complete mitosis. The protein known as H-Ras GTPase is essential to proper biological functioning in the entire web of life. The main function of this protein is giving the "stop" signal to the process of cell reproduction. Unfortunately, this protein is not perfect and severe consequences, such as cancer, can arise when H-Ras GTPase malfunctions. H-Ras GTPase is a protein from the large family of enzymes that bind and split GTP. H-Ras GTPase is vital in processes like cell-to-cell communication, protein translation in ribosomes, and programmed cell death (apoptosis). Its main fields of operation are determining stem cell into specific functioning cells, as well as replicating preexisting "specialized" cells. All G domain based proteins have a universal structure and two universal switch mechanisms, which consist of a mixed, six-stranded beta sheet and five alpha helices. H-Ras GTPase works by first dissociating from GDP and binding to GTP, activating the protein. Then it binds to another Gprotein to enact a cell response. After interaction, the enzyme GAP hydrolyzes GTP to GDP, turning off the Hras switch. Ras GTPase Ras GDPase Active Inactive Controlling the “Switch” between Active and Inactive States In the graphics (above and below), H-ras is shown in both active and inactive states. In its active form, the enzyme is bound to GTP. In this state H-ras helps direct mitosis in eukaryotes. When H-ras is inactive it is bound with GDP, ceasing cell replication. H-Ras GTPase has a high affinity for both GDP and GTP. This affinity for GDP impairs the switch from turning off, which can lead to serious problems, such as cancer. When GTPase bonds to GTP, the molecular switches change shape. This newly shaped GTPase now bonds to a protein to transmit instructions. Since H-Ras GTPase is central to cell division, slight mutations in the protein cause the switch to be "stuck-on," resulting in hyperactive cell growth and division. These oncogenic mutations in HRas GTPase are responsible for nearly 30% of all humanform cancers. © 2008, Physiomics, Corp. http://www.physiomicsplc.com/cell%20cycle%20model.htm Connection to Cancer Surprisingly, H-ras is responsible for an estimated 30% of human cancers. The trigger for the cancer is often UV light or spontaneous DNA mutation. In the worst case scenario Hras’s active site is damaged, and it cannot release GTP. This mutation locks H-ras in the active position. If H-ras is not inactivated, unchecked cell division and tumor formation may occur. Directing Intracellular Communication H-ras is part of a G protein pathway. A G protein pathway begins when a signal from outside the cell binds to a surface protein. This causes a second protein to bind to GTP, which activates the secondary protein. H-ras qualifies as the secondary protein. The activated protein binds to a third protein called a G protein, which causes further signals that complete the intended cell activity. © 2008, Pearson Education, Campbell Biology, 7th Ed http://users.rcn.com/jkimball.ma.ultranet/BiologyPages/C/Cancer.html A SMART Team project supported by the National Institutes of Health (NIH) – National Center for Research Resources Science Education Partnership Award (NCRR-SEPA)