* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Genome evolution wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
RNA interference wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Secreted frizzled-related protein 1 wikipedia , lookup
RNA silencing wikipedia , lookup
Community fingerprinting wikipedia , lookup
Polyadenylation wikipedia , lookup
Gene expression profiling wikipedia , lookup
Molecular evolution wikipedia , lookup
List of types of proteins wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Non-coding DNA wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Messenger RNA wikipedia , lookup
Point mutation wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Non-coding RNA wikipedia , lookup
Histone acetylation and deacetylation wikipedia , lookup
Transcription factor wikipedia , lookup
Endogenous retrovirus wikipedia , lookup
Epitranscriptome wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Gene regulatory network wikipedia , lookup
Eukaryotic transcription wikipedia , lookup
RNA polymerase II holoenzyme wikipedia , lookup
Gene expression wikipedia , lookup
Promoter (genetics) wikipedia , lookup
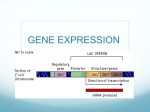
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 18 Regulation of Gene Expression Lectures by Erin Barley Kathleen Fitzpatrick © 2011 Pearson Education, Inc. Gene expression: Bacteria vs. Eukaryotes • prokaryotes and eukaryotes alter gene expression in response to their changing environment – gene expression = refers to the entire process whereby genetic information is decoded into a protein • prokaryotes and eukaryotes carry out gene expression in similar ways – transcription using an RNA polymerase – translation using ribosomes • but there are some differences: – – – – – 1. RNA polymerases differ – only one in prokaryotes; 3 in eukaryotes 2. transcription factors used by eukaryotes 3. transcription is terminated differently in prokaryotes vs. eukaryotes 4. ribosomes – bacterial ones are smaller 5. lack of compartmentalization in bacteria – transcribe and translate at the same time So what is a gene? • • • • • unit of inheritance located on chromosomes region of specific nucleotide sequence located along the length of DNA DNA sequence that codes for a specific sequence of amino acids BUT: some DNA sequences are NEVER translated – e.g. rRNA and tRNA are transcribed but not translated into anything • so a gene is a region of DNA that is either – 1. translated into a sequence of amino acids (polypeptide) functional protein – 2. transcribed into a RNA molecule So what is a gene? • molecular components of a gene: – – – – – A. coding sequences - eukaryotes have introns within their coding sequence B. promoter C. enhancers – found in eukaryotes D. UTRs – found in eukaryotes E. poly-adenylation sequence – found within the eukaryotic 3’ UTR Overview: Conducting the Genetic Orchestra • genetic and biochemical work in bacteria identified two things – 1. protein-binding regulatory sequences associated with genes – 2. proteins that can bind these regulatory sequences – either activating or repressing gene expression • these two components underlie the ability of both prokaryotic and eukaryotic cells to turn genes on and off Bacteria often respond to environmental change by regulating transcription • natural selection has favored bacteria that produce only the products needed by that cell • bacteria regulate the production of enzymes by feedback inhibition or by gene regulation • gene expression in bacteria is controlled by the operon model Precursor Feedback inhibition trpE gene Enzyme 1 trpD gene Enzyme 2 Regulation of gene expression trpC gene trpB gene Enzyme 3 trpA gene Tryptophan (a) Regulation of enzyme activity (b) Regulation of enzyme production Operons: Definitions & Concepts • bacteria group functionally related genes so they can be under coordinated control by a single “on-off regulatory switch” • the regulatory “switch” is a segment of DNA called an operator – binding sites for transcription factors that help RNA polymerase II bind the nearby promoter • the operator can be controlled by proteins or nutrients – e.g. can be switched off by a protein called a repressor – repressor prevents gene transcription - binds to the operator and blocks RNA polymerase binding to the promoter – repressor is the product of a separate regulatory gene – repressor can be in an active or inactive form, depending on the presence of other molecules • co-repressor is a molecule that cooperates with a repressor protein to switch an operon off – e.g. the amino acid tryptophan Operons: Definitions & Concepts • operon = the entire stretch of DNA that includes the operator, the promoter, and the genes that the promoter controls – the transcription of the downstream genes is polycistronic – produces one long piece of mRNA containing multiple transcription units • two kinds – “On” and “Off” operons trp operon Promoter trpE Operator Start codon mRNA 5 Genes of operon trpD trpC trpB trpA B A Stop codon E D C Polypeptide subunits that make up enzymes for tryptophan synthesis Repressible and Inducible Operons: Two Types of Negative Gene Regulation • OFF operon = repressible operon is one that is usually on but is turned OFF by a repressor – e.g. the trp operon is a repressible operon • ON operon = inducible operon is one that is usually off but is turned ON by an inducer – e.g. lac operon is an inducible operon The trp Operon: Repressible Operons • • • E. coli can synthesize the amino acid tryptophan when it is absent from the growth media by default the trp operon is on and the genes for tryptophan synthesis are transcribed comprised of the: – 1. operator – capable of binding a repressor protein – 2. genes of the operon – for synthesizing tryptophan when it is missing from the growth media • plus a regulatory gene = trpR – expressed whether tryptophan is absent or present The trp Operon: Repressible Operons trp operon Promoter Promoter Genes of operon DNA trpE trpR Regulatory gene mRNA 3 RNA polymerase Operator Start codon trpD trpC trpB trpA C B A Stop codon mRNA 5 5 E Protein Inactive repressor (a) Tryptophan absent, repressor inactive, operon on D Polypeptide subunits that make up enzymes for tryptophan synthesis •when tryptophan is absent – the operon needs to function to make tryptophan •the repressor protein is made but it is inactive & is incapable of binding the operator •RNA polymerase can bind the promoter and the downstream genes are expressed The trp Operon: Repressible Operons DNA No RNA made mRNA Protein Active repressor Tryptophan (corepressor) (b) Tryptophan present, repressor active, operon off •when tryptophan is present – the operon does not need to be functional •tryptophan acts as a co-repressor & binds the repressor protein •this allows the repressor to bind and repress the function of the operator •MUCH lower downstream gene expression vs. when the operon is ON The lac Operon: Inducible Operons • proposed by Francois Jacob and Jacques Monod - 1960s • E.coli can use glucose and other sugars (such as lactose) as their sole source of carbon and energy • the normal situation is for the bacteria to use glucose – levels of a bacterial enzyme called beta-galactosidase (lactose breakdown) are very low • when lactose is given to the bacteria – b-Gal levels increase – said to be induced • the lac operon is an inducible operon – contains genes that code for enzymes used in the hydrolysis and metabolism of lactose • when E. coli are grown with glucose – no need for the enzymes of the lac operon since there is no lactose in the medium – so the operon is turned OFF • but with media containing lactose – need to turn the operon ON to make the enzymes for metabolizing and using lactose The lac Operon: Inducible Operons • genes of the lac-operon: – 1. lacZ gene = beta-galactosidase – splits the lactose into glucose and galactose – 2. lacY gene – 3. lacA gene – 4. lacI gene = codes for a lac repressor – 5. operator = binds transcription factors – 6. promoter = binds RNA polymerase II • THIS IS IMPORTANT!!! - without any outside control - the lac repressor gene lacI is constitutively active and acts to eventually switch the lac operon OFF – through the constitutive production of a lac repressor protein • a molecule called an inducer is needed to inactivate the repressor to turn the lac operon ON Regulatory gene Promoter Operator lacZ lacI DNA No RNA made 3 mRNA 5 Protein RNA polymerase Active repressor (a) Lactose absent, repressor active, operon off lac repressor protein •when lactose is absent – an active repressor is made •the genes metabolizing lactose are NOT needed •repressor gene lacI is constitutively active – makes a lactose repressor •repressor binds the operator and hinders the binding of the RNA polymerase to the promoter •downstream genes are transcribed AT A VERY LOW LEVEL •when lactose is present – an inducer is required to turn the operon ON •metabolizing and using lactose is now needed •ALLOLACTOSE ACTS AS AN INDUCER •allolactose – form of lactose that can enter bacterial cells •the inducer binds the repressor and prevents it from binding to the operator •the downstream genes are expressed AT A HIGH LEVEL •lactose binding to the repressor shifts the repressor to its non-DNA binding conformation lac operon lacI DNA lacZ lacY -Galactosidase Permease lacA RNA polymerase 3 mRNA 5 mRNA 5 Protein Allolactose (inducer) Inactive repressor (b) Lactose present, repressor inactive, operon on Transacetylase • in nature – the inducer of the lab operon is a lactose derivative • in the lab – other inducers can be used to turn the operon on – e.g. IPTG = isopropyl-b-D-thiogalactoside – IPTG is NOT a substrate of -Gal • we can also give the bacteria a specific b-Gal substrate that will turn colors – X-Gal – turns blue with broken down by b-gal enzyme – used to identify bacteria containing cloned genes – can insert additional genes into plasmids containing the b-gal gene • insertion of your desired gene INTO the plasmid disrupts -gal expression • inability to breakdown X-Gal – colonies are white • inducible enzymes usually function in catabolic pathways – their synthesis is induced by a chemical signal • repressible enzymes usually function in anabolic pathways – their synthesis is repressed by high levels of the end product • regulation of the trp and lac operons involves negative control of genes because operons are switched off by the active form of the repressor Positive Gene Regulation: CAP proteins • some operons are also subject to positive control • when bacteria are given both lactose AND glucose - the bacteria will use glucose – the enzymes for glycolysis are continually present in bacteria • when lactose is present and glucose is short supply – it makes the enzymes for lactose metabolism • how does the bacteria sense the low levels of glucose?? Promoter DNA lacI lacZ CAP-binding site cAMP Operator RNA polymerase Active binds and transcribes CAP Inactive CAP Allolactose Inactive lac repressor (a) Lactose present, glucose scarce (cAMP level high): abundant lac mRNA synthesized -when glucose is scarce accumulation of a small molecule called cyclic AMP (cAMP) -cAMP functions as a “2nd messanger” to signal that glucose levels are low in the growth medium - high levels of cAMP activate a regulatory protein called catabolite activator protein (CAP) -cAMP binds CAP and activates it - activated CAP attaches to the lac operon promoter and accelerates transcription (functions as a transcription factor) - enhances the affinity of RNA polymerase for the promoter • CAP helps regulate other operons that encode enzymes used in catabolic pathways • when glucose levels are low and lactose levels are high – 1. lactose binds the lactose repressor and prevents it from binding the operator and inhibiting gene transcription = genes for lactose metabolism are made – 2. cAMP activation of CAP and its binding to the lac promoter increases transcription = lactose genes are made at a higher rate • when glucose levels increase and lactose levels decrease – 1. CAP activation will eventually decrease and so will its enhancement of transcription – 2. the lactose repressor is now able to bind the operator and inhibit transcription • so the lac operon is actually under dual control as lactose increases and glucose decreases: – positive – as levels of cAMP rise – so does CAP activation and the activity of the lac operon – negative – as repressor activity decreases & the activity of the lac operon increases – THEREFORE: it is the allosteric state of the lac repressor that determines if transcription happens – it is the presence of CAP that controls the rate at which transcription will happen Eukaryotic gene expression is regulated at many stages Signal NUCLEUS Chromatin • • • • • all organisms must regulate which genes are expressed at any given time in the same organism – the genomes are identical from cell to cell so why do different cells express different genes/proteins?? differences result from differential gene expression = the expression of different genes by cells with the same genome several steps along the replication/transcription/translation path are control points for differential gene expression – control of DNA transcription – modification of DNA-histone interaction – post-transcriptional control – post-translational control DNA Chromatin modification: DNA unpacking involving histone acetylation and DNA demethylation Gene available for transcription Gene Transcription RNA Cap Exon Primary transcript Intron RNA processing Tail mRNA in nucleus Transport to cytoplasm CYTOPLASM mRNA in cytoplasm Degradation of mRNA Translation Polypeptide Protein processing, such as cleavage and chemical modification Degradation of protein Active protein Transport to cellular destination Cellular function (such as enzymatic activity, structural support) Control of DNA Transcription: Histone Acetylation • each of the histone proteins (H2A, H2B, H3, H4) contain flexible extensions of 20 to 40 amino acids called “tails” • these histones can be modified posttranslationally by the addition of functional groups • at the end of these tails are several positively charged lysine amino acids • some of these lysines undergo reversible chemical modification called acetylation – important for transcription, resistance against DNA degradation Histones Histones Acetylation and Deacetylation of DNA • numerous post-translational modifications can be done to histone proteins – affects how the DNA-histone interacts and ultimately affects the transcription of the DNA • some histone lysines undergo reversible chemical modifications called acetylation and deacteylation • acetylation = transfer of an acetyl group onto the NH2 terminus of an amino acid – for histones – performed by a family of enzymes called histone acteyltransferases (HATs) • acetylation neutralizes the +ve charge of these lysines – its interaction with the DNA is eliminated – the DNA becomes less tightly associated with the histone – results in better access for the transcriptional machinery to the DNA acetyl coA “donor” lysine R-group Acetylation and Deacetylation of DNA • deacetylation = removal of this acetyl group from the histone by a family of enzymes called histone deacetylases (HDACs) – increases the interaction between DNA and the histone by removing the acetyl group and increasing the “positivity” of the lysine residues – 11 eukaryotic HDACs !!! acetyl coA “donor” lysine R-group Control of DNA Transcription: Acetylation and Deactylation of DNA • acetylation/deacetylation is a transient histone modification that affects transcription – euchromatin – higher HAT activity more transcriptionally active form of chromatin – heterochromatin – higher HDAC activity less transcriptionally active form of chromatin heterochromatin euchromatin Increased binding of transcription factors and RNA Pol II to “opened” acetylated chromatin Protein Control of DNA Transcription: Acetylation and Deacetylation of DNA • the HAT/HDAC enzymes are part of a large complex of proteins that binds the DNA – includes transcription factors, other regulatory proteins, RNA polymerase II • it is now thought that HAT and HDAC enzymes are recruited into this complex – once there – they modify the DNA and give the rest of the transcription machine better “access” to the DNA helix • non-histone proteins can also be acetylated!! – e.g. transcription factors are also acetylated/deacetylated – changes their activity level and therefore transcription Histone Methylation • histone methylation = the addition of methyl groups (CH3) to certain amino acids on histone tails – lysines or arginines – usually lysines – is associated with reduced transcription in cases, increased transcription in others – usually results in increased association between the histone and the DNA and a decrease in transcription in that area – histone methylation is considered an epigenetic modification • alteration of gene expression by mechanisms outside of DNA structure • performed by a family of enzymes called histone methyltransferases DNA Methylation • in addition to histones – methyl groups can be attached to certain DNA bases = DNA methylation – – – – usually cytosine done by a different set of enzymes than those that methylate histones is associated with reduced transcription in some species i.e. the more methylated, the more inactive the gene • DNA methylation essential for long-term inactivation of genes during cellular differentiation – DNA methylation can last through several rounds of replication – when a methylated DNA sequence is replicated – the daughter strand is methylated too – can affect transcription rates over several rounds of replication Regulation of Transcription Initiation • chromatin-modifying enzymes provide initial control of gene expression by making a region of DNA either more or less able to bind the transcription machinery • additional transcriptional levels are also found – enhancers – promoters Organization of a Typical Eukaryotic Gene Enhancer (distal control elements) DNA Upstream Proximal control elements Transcription start site Exon Promoter Intron Exon Intron Poly-A signal sequence Exon Transcription termination region Downstream • most eukaryotic genes are associated with multiple control elements – segments of noncoding DNA that serve as binding sites for transcription factors that help regulate transcription – distal elements– known as enhancers – proximal elements – associated with promoters • many of these control elements and the transcription factors they bind are responsible for the differential gene expression seen in different cell types Transcription Factors • proteins that bind sequences of DNA to control transcription • can act as activators or repressors to transcription – activating TFs - proteins that recruit the RNA polymerase to a promoter region – repressing TFs – proteins that prevent transcription in many ways • must contain a DNA binding domain to be a transcription factor • not always one protein – can be multiple subunits together in a complex • two broad categories: – 1. general transcription factors – 2. specific transcription factors Transcription Factors • two broad categories: – 1. general transcription factors are essential for the transcription of all protein-coding genes • assist the RNA polymerase in binding the promoter region – only give a low level of transcription!! • activity is enhanced by specific transcription factors – 2. specific transcription factors control the high-level, differential expression of specific genes within a specific cell type • • • • bind the promoter and enhancer regions of a gene can function to activate or repress transcription e.g. Runx-2 – transcription factor that is found in osteoblasts directs the expression of several osteogenic genes involved in making bone Transcription Factors bind DNA • binding of a TF to DNA is easy to see experimentally using a DNA mobility shift assay • Step #1 – radioactively label your DNA • Step #2 – mix your labelled DNA with your possible TFs • Step #3 – Southern blot - run the DNA on a gel and transfer it to a filter paper -control – DNA not mixed with the possible TF • Step #4 – expose the filter to film to develop the radioactive signal DNA footprinting assay Promoters • • • • sequence of DNA located immediately upstream of the transcription start site promotes transcription of DNA into RNA site of RNA polymerase binding in both prokaryotes and eukaryotes contain sequences for the binding of RNA polymerase and sequences for the binding of transcription factors Promoters • initial work done in bacteria – found two kinds of DNA sequences in the promoter • 1. those that are found in the promoters of all bacterial genes • 2. those that are found in a more limited number of genes that respond to a specific signal • core bacterial promoter: binds RNA polymerase and an associated sigma factor (part of the RNA polymerase complex) – two key DNA sequences located 10 and 35 bp upstream from the transcription start site (TSS) – for the binding of the RNA polymerase – -10 site = TATA box (consensus sequence TATAAT) – -35 site - TTGACA Promoters • eukaryotic promoters: 10 classes of promoters known – DEFINITION: DNA sequence that binds the RNA polymerase II (plus 7 additional factors) – BUT – more complicated than prokaryotic promoters – are additional upstream DNA sequences that regulate transcription – three well-known regions studied • -30, -75 and -90 sites – two kinds of DNA sequences associated with transcription of a eukaryotic gene: – 1. part of the promoter (i.e. control elements) involved in the basic process of transcription – 2. control elements active in a particular tissue type or in response to a specific signal – regulated transcription Promoters • promoter sequences involved in basic transcription – TATA box – conserved from the bacterial TATA box • • • • • • • A/T rich sequence - 30 base pairs upstream of start site in most genes but not all – not found in housekeeping genes together with the transcription start site – considered to be the core promoter accurately positions the RNA polymerase at the start site also binds general transcription factors actually provides a very low level of transcription needs upstream promoter elements (UPEs) to increase transcriptional control – UPEs – also essential for transcription • • • • increase the efficiency of transcription e.g. SP1 sequence &/or CCAAT box some genes can have an SP1 and CCAAT box – e.g. hsp70 gene others may only have one – e.g. metallothionein gene • promoters sequences involved in regulated transcription – promoters also have control elements that are shared with a more limited number of genes – interspersed among the UPEs – provide cell-type specific or signal-specific transcription – some well-known sequences are known as Response Elements – e.g. metallothionein gene – metal response elements (MREs) • gene binds metals such as zinc – acts to decrease oxidative stress – many hormones act through these response elements • e.g. estrogen Histone 2A gene promoter Metallothionein 1 gene promoter Promoters – functions of promoter were discovered through • mutation or deletion of specific regions • putting these regions in front of a reporter gene is the reporter gene transcribed? • e.g. insertion of the metallothionein gene promoter into a reporter plasmid containing the firefly luciferase gene (reporter gene) • transcription of the luciferase gene will be driven by the inserted promoter • put in the entire promoter and measure luciferase activity • put in “pieces” of the promoter with specific regions deleted – measure luciferase activity Activation of Metallothionein Gene Expression by Hypoxia Involves Metal Response Elements and Metal Transcription Factor-1 Cancer Res March 15, 1999 vol. 59 no. 6 pp1315-1322 Enhancers • distal control elements of a gene • DNA sequences that act to enhance eukaryotic transcription • can be found either: – – – – upstream of the gene downstream of the gene within the gene even on a different chromosome!!! • act to increase the activity of the promoter – DO NOT have promoter activity themselves • some enhancers are active in all tissues and increase promoter activity constitutively • others are only expressed in specific cells • made up of several DNA sequences (sequence elements) that bind transcription factors which interact together • many of these sequence elements are also found in the promoter Enhancer (distal control elements) DNA Upstream Proximal control elements Transcription start site Exon Promoter Intron Exon Intron Poly-A signal sequence Exon Transcription termination region Downstream Enhancers & their Transcription Factors • transcription factors than bind enhancers are called activators – positively acting transcription factors • • activators = proteins that bind to DNA sequences and stimulate/activate transcription of a gene activators have two domains – 1. DNA binding domain – 2. activation domain - site that activates transcription by helping to form the transcription initiation complex Activation domain DNA-binding domain DNA Eukaryotic gene elements: a summary • so the typical eukaryotic gene consists of up to 4 distinct control elements – 1. core promoter itself – upstream of the transcription start site – 2. upstream promoter elements (UPEs) located close to the promoter – required for efficient transcription in any cell – 3. elements that intersperse among the UPEs and activate transcription of genes in specific tissues or in response to specific stimuli – regulated transcription elements – 4. distant elements called enhancers Transcription Initiation • transcription can happen as long as the core promoter is present – but transcription rates will be very low • • • so efficient transcription of eukaryotic genes requires the activity of the promoter, enhancers and a multitude of transcription factors together with the RNA polymerase II these components come together to form a transcription initiation complex stepwise assembly – 1. binding of three general transcription factors at the TATA box– TFIIA, TFIID TFIIB – 2. recruitment and binding of the RNA polymerase II at the TF/TATA box complex • in some organisms – polymerase is bound to these 3 TFs before binding DNA first = RNA polymerase holoenzyme – 3. additional general TFs join – 4. binding of gene-specific TFs + interaction with enhancer/activators Promoter Activators Gene DNA Distal control Enhancer element -activators bind to the DNA of the enhancer via their DNA-binding domains -the activators bind to regions called distal control elements TATA box General transcription factors DNAbending protein Group of mediator proteins a DNA bending protein “bends” the distal enhancer region – bringing it close to the the promoter RNA polymerase II general transcription factors, promoterspecific TFs, mediator proteins and RNA polymerase II form a transcription initiation complex with the enhancer and its activators RNA polymerase II Transcription initiation complex RNA synthesis Transcription Initiation activator-enhancer Mediator protein = co-activator -multi-subunit protein complex found in the transcription initiation complex of all eukaryotes -31 proteins!!!! -functions as a bridge between transcription factors promoter-specific transcription factors Repressors • some transcription factors can also function as repressors or silencers – inhibiting expression of a particular gene by a variety of methods – some repressors bind activators and prevent their binding to enhancers – some bind the distal control elements in the enhancer directly – others bind proximal control elements or the promoter Cell-Type Specific Transcription Enhancer Promoter Control elements LIVER CELL NUCLEUS Albumin gene Crystallin gene Available activators • both liver and lens cells have the same genome • so why does a liver cell make albumin and a lens cell make crystallin????? • it’s the transcription factors and control elements • liver cell has a unique complement of transcription factors that activate albumin transcription Albumin gene expressed Crystallin gene not expressed (a) Liver cell Enhancer Control elements Promoter LENS CELL NUCLEUS Albumin gene Crystallin gene Available activators • the lens cell has a different set of TFs that activates crystallin transcription • these transcription factors may only be made within a lens or liver cell at a precise time in development or in response to an extracellular signal (e.g. growth factor or hormone) or even an environmental cue Albumin gene not expressed Crystallin gene expressed (b) Lens cell Coordinately Controlled Genes in Eukaryotes • unlike the genes of a prokaryotic operon, each coexpressed eukaryotic gene has a core promoter and several other control elements • these genes can be scattered over different chromosomes, but each has the same combination of control elements as one another • multiple copies of activators recognize these control elements on each gene and promote their simultaneous transcription Mechanisms of Post-Transcriptional Regulation • transcription alone does not account for gene expression • regulatory mechanisms can operate at various stages after transcription • allow a cell to fine-tune gene expression rapidly in response to environmental changes • post-transcriptional processing: – 1. mRNA structure – cap and tail; UTRs – 2. mRNA splicing – 3. mRNA half life and degradation Post-Transcriptional Regulation: mRNA structure • pre-RNA processing to mRNA involves the addition of the 5’ methylated cap and 3’ poly-A tail • cap is added shortly after transcription initiation – by a capping enzyme which is associated with the RNA polymerase II – cap - 7-methylguanosine – function of the cap • 1. protection against mRNA degradation • 2. export out into the cytoplasm • 3. binding of the small subunit for translation Post-Transcriptional Regulation: mRNA structure • cap is followed by the 5’UTR region (untranslated region) – found between the transcription start site and ends one nucleotide before the ATG/start codon of the coding sequence – ontains elements for controlling gene expression and mRNA export – contains sequences that are involved in translation initiation – in bacteria – contains a sequence for docking of the ribosome = Shine Delgarno Sequence TSS Post-Transcriptional Regulation: mRNA structure • poly A tail – in animal cells, all mRNAs (except histone mRNAs) have polyA tails – prevent degradation of the mRNA and induces export from the nucleus – two special mRNA sequences are needed – located in the 3’UTR • 1. Poly Adenylation signal – AAUAAA • 2. Poly A site – downstream from the signal – area rich in Gs ands Us – area where the mRNA is cut and the poly-A tail is added -complex of proteins binds the poly-Adenylation signal -a Poly(A) polymerase is recruited – cleaves the mRNA at the poly A site and adds hundreds of “A” nucleotides Post-Transcriptional Regulation: mRNA structure • 3’UTR – second of the two UTRs that flank a transcription unit’s coding sequence – length is important – longer the UTR, the lower level of gene expression – can be an area of variability – controls expression levels and localization of a protein within a cell – contains numerous regulatory regions for • 1. poly –adenylation – contains the polyA signal and polyA site • 2. mRNA creation – silencer regions to repress transcription of mRNA • 3. mRNA stability – contain AU-rich elements (AREs) that increase the stability of the mRNA • 4. mRNA export – contains sequences that attract nuclear export proteins • 5. translation efficiency – also affected by AREs Post-transcriptional control: mRNA degradation • the life span of mRNA molecules in the cytoplasm is a key to determining protein synthesis • eukaryotic mRNA is more long lived than prokaryotic mRNA • numerous enzymes (RNases) can breakdown mRNA • the binding of small RNAs called microRNAs (miRNAs) to the mRNA can target it for degredation Post-transcriptional Regulation: Splicing • • removal of introns and the joining of exons performed in the nucleus by the spliceosome – small nuclear RNAs – U1, U2, U4, U5 and U6 – associated with protein subunits = snRNPs – snRNPs form the core of the spliceosome • • spliceosome recognizes conserved sequences at the start and end of an intron = splice sites introns are removed as a lariat structure – a 5’ G at the end of the intron is joined in an unusual phosphodiester bond (2’ to 5’) to the A at the end of the intron – “A” nucleotide is called a branch point Post-transcriptional Regulation: Splicing • • • • • • • the snRNPs are numbered based on their “entrance” into the splicing reaction 1. first is U1 – base pairs with the 5’ G at the start of the intron 2. next is U2 which binds the branch point A 3. U4/U6 and U5 enter next – formation of the completed spliceosome 4. rearrangements among these snRNAs “loops out” the intron and cuts the intron at the 5’ end 5. departure of U1 and U4 + joining of the 5’ end of the intron to the A (completes the lariat) 6. U6 snRNA cuts at the 3’ end of the intron and joins the two exon sequences Splicing • In alternative RNA splicing, different mRNA molecules are produced from the same primary transcript, depending on which RNA segments are treated as exons and which as introns Exons DNA 1 4 3 2 5 Troponin T gene Primary RNA transcript 3 2 1 5 4 RNA splicing mRNA 1 2 3 5 or 1 2 4 5 Initiation of Translation • initiation of translation of selected mRNAs can be blocked by regulatory proteins that bind to sequences or structures of the mRNA • alternatively, translation of all mRNAs in a cell may be regulated simultaneously • for example, translation initiation factors are simultaneously activated in an egg following fertilization Protein Processing • after translation - various types of protein processing, including folding, cleavage and the addition of chemical groups take place • known as post-translational processing • numerous kinds of chemical additions Post-translational modifications of Proteins • usually involves enzymes adding – – – – – – 1. hydrophobic groups (fatty acids) for membrane localization of protein 2. cofactors to enhance enzymatic activity 3. additional peptides or proteins – e.g. ubiquitin 4. structural changes – disulfide bridges between cysteines 5. proteolytic cleavage 6. chemical groups • numerous chemical groups can be added – – – – – a. phosphorylation b. methylation c. acetylation d. glycosylation e. iodination • non enzymatic modifications – changes the chemistry of an amino acid without the use of an enzyme – e.g. chemical addition of biotin (vitamin B7) as a cofactor Protein Folding: Chaperones • protein function is completely dependent upon 3D structure • the information for folding is contained within the amino acid sequence of the polypeptide chain – the hydrophobic residues are “buried” within the center of the folding protein – spontaneous process – large numbers of interactions between the R groups of the AAs form • ionic • van der waals – between hydrophobic groups • disulfide bridges • folding can begin the minute the PP chain emerges from the ribosome • but most protein don’t • these proteins are met at the ribosome by a class of proteins called molecular chaperones Protein Folding: Chaperones • molecular chaperones – best described class of chaperones – heat shock proteins or HSPs – numerous families – e.g. HSP40, HSP60, HSP70 – work by interacting with exposed hydrophobic AAs – hydrophobic residues are dangerous – distinct mechanisms for each chaperone – e.g. HSP60 forms a “barrel-like” structure that “isolates” folding proteins AFTER they are made = known as chaperonin – bind to the hydrophobic residues and ensures correct folding • some chaperones bind to misfolded proteins and sequester them until they are destroyed Proteasome and ubiquitin to be recycled Ubiquitin Proteasome Protein to be degraded • • • • Protein entering a proteasome Protein fragments (peptides) if the protein is not folded properly – will have to be degraded Proteasomes are giant protein complexes that bind protein molecules and degrade them consists of a protein complex that forms a hollow cylinder top and bottom are additional protein complexes that feed the abnormal protein into the core – – – • Ubiquitinated protein protein is unfolded as it is fed in exposed to proteases within the core keeps the protein in the core until the entire protein is cleaved into peptides signal to enter the proteosome is the chemical attachment of a poly-ubiquitin chain – ubiquitin is prepared by an enzyme (Ubiquitin activating enzyme) – attached to lysines by enzymes (Ubiquitin ligase) – done over and over poly-ubiquitin chain Noncoding RNAs play multiple roles in controlling gene expression • only a small fraction of DNA codes for proteins – 30,000 to 100,000 genes • a fraction of the non-protein-coding DNA pieces are genes for RNAs such as rRNA and tRNA • but most are transcribed into noncoding RNAs (ncRNAs) – e.g. miRNA – e.g. siRNA • noncoding RNAs regulate gene expression at two points: mRNA translation and chromatin configuration MicroRNAs • • • • • • • • MicroRNAs (miRNAs) are small single-stranded RNA molecules that can bind to mRNA Hairpin Hydrogen miRNA can degrade mRNA or block its translation bond miRNAs are made by RNA polymerase II & are capped Dicer and poly-adenylated exported out to the cytoplasm (a) Primary miRNA transcript miRNA miRNAprotein a protein called Dicer cleaves the primary miRNA into the complex mature miRNA one strand of miRNA associates with proteins RNAinduced silencing complex (RISC) then the RISC base pairs with it complementary mRNA nucleotides – usually in the 3’UTR mRNA degraded Translation blocked (b) Generation and function of miRNAs if base pairing is extensive cleavage of mRNA 5 3 – • happens in plant cells if base pairing is limited repression of translation – happens in animal cells Small Interfering RNAs • • • through RNA interference (RNAi) viruses can target and destroy host mRNAs RNAi is done through the production of small interfering RNAs (siRNAs) – target and destroy mRNA just like miRNA siRNAs and miRNAs are similar but form from Dicer cutting up different RNA precursors – miRNA made from single stranded RNA made by the cell – siRNA made from double stranded RNA (shRNA) made by viruses • siRNA is now used in the lab to target specific mRNAs – introduce artificial shRNA into cells siRNA that will bind and degrade your desired piece of mRNA 1. viral production of dsRNA (shRNA) 2. complexing with DICER siRNA 3. integration of siRNA with RISC 4. binding to target mRNA 5. DESTRUCTION of mRNA The Evolutionary Significance of Small ncRNAs • Small ncRNAs can regulate gene expression at multiple steps • An increase in the number of miRNAs in a species may have allowed morphological complexity to increase over evolutionary time • siRNAs may have evolved first, followed by miRNAs