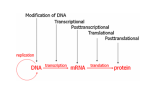
* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download DNA Structure
Mitochondrial DNA wikipedia , lookup
Zinc finger nuclease wikipedia , lookup
Epigenetic clock wikipedia , lookup
Human genome wikipedia , lookup
Microevolution wikipedia , lookup
DNA profiling wikipedia , lookup
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Behavioral epigenetics wikipedia , lookup
Nucleic acid tertiary structure wikipedia , lookup
Genomic library wikipedia , lookup
SNP genotyping wikipedia , lookup
DNA polymerase wikipedia , lookup
DNA damage theory of aging wikipedia , lookup
DNA methylation wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Point mutation wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
United Kingdom National DNA Database wikipedia , lookup
Molecular cloning wikipedia , lookup
DNA vaccination wikipedia , lookup
Genealogical DNA test wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Epigenetics of depression wikipedia , lookup
Non-coding DNA wikipedia , lookup
Primary transcript wikipedia , lookup
History of genetic engineering wikipedia , lookup
Extrachromosomal DNA wikipedia , lookup
Cre-Lox recombination wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Epigenetics of human development wikipedia , lookup
DNA supercoil wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Helitron (biology) wikipedia , lookup
Bisulfite sequencing wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Epigenetics wikipedia , lookup
Epigenetics in stem-cell differentiation wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Nucleic acid double helix wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Histone acetyltransferase wikipedia , lookup
Epigenomics wikipedia , lookup
1- B DNA 2- Double helix 3- Antiparallel 4- Major groove 5- Minor groove 6- Sugar puckering 7- C2 endo 8- C3 Exo 9- Syn, Anti --Right handed 10- A DNA, C3 endo position 11- Z DNA -- Left handed, G and A in syn position -- G and (A) in C3 endo position -- Base stacking --Hydrogen bond -- Phosphate position -- Hoogsteen Base pair -- Triple stranded helix -- Genome -- Relaxed form -- Super coil -- Histone like proteins -- Nucleosome -- Nucleosome core --Chromatin -- Linker -- Sscaffold -- Solenoid --Melting point Hypochromism. (A) Single-stranded DNA absorbs light more effectively than does double-helical DNA. (B) The absorbance of a DNA solution at a wavelength of 260 nm increases when the double helix is melted into single strands. Step Melting ΔG -1 /Kcal mol TA -0.12 T G or C A -0.78 CG -1.44 A G or C T -1.29 A A or T T -1.04 AT -1.27 G A or T C -1.66 C C or G G -1.97 A C or G T -2.04 GC -2.70 Structural Parameter A-DNA B-DNA Z-DNA Direction of helix rotation Right handed Right handed Left handed Residue per helical turn 11 10 12 Pitch (length) of the helix 28.2 Å 34 Å 44.4 Å Base pair tilt 20° -1° 7° Rotation per residue 32.7° 34.3° -30° Diameter of helix 23 Å 20 Å 18 Å Configuration dA, dT, dC anti anti anti of glycosidic bond dG anti anti syn Sugar Pucker dA, dT, dC C3' endo C2' endo C2' endo dG C3' endo C2' endo C3' endo Topology of major groove Narrow, deep Wide, deep Flat Topology of minor groove Broad, shallow Narrow, shallow Narrow, deep Major- and Minor-Groove Sides. Because the two glycosidic bonds are not diametrically opposite each other, each base pair has a larger side that defines the major groove and a smaller side that defines the minor groove. The grooves are lined by potential hydrogen-bond donors (blue) and acceptors (red). Propeller Twist. The bases of a DNA base pair are often not precisely coplanar. They are twisted with respect to each other, like the blades of a propeller. Axial View of DNA. Base pairs are stacked nearly one on top of another in the double helix Step Stacking ΔG -1 /kcal mol TA -0.19 T G or C A -0.55 CG -0.91 A G or C T -1.06 A A or T T -1.11 AT -1.34 G A or T C -1.43 C C or G G -1.44 A C or G T -1.81 GC -2.17 Histonea Molecular weight Number of amino acids Percentage Lysine + Arginine H1 22,500 244 30.8 H2A 13,960 129 20.2 H2B 13,774 125 22.4 H3 15,273 135 22.9 H4 11,236 102 24.5 a Data are for rabbit (H1) and bovine histones. Structure of Histone Acetyltransferase. The amino-terminal tail of histone H3 extends into a pocket in which a lysine side chain can accept an acetyl group from acetyl CoA bound in an adjacent site Histone modifications Histone proteins can undergo various types of modification, the best studied of these being histone acetylation. – the attachment of acetyl groups to lysine amino acids in the N-terminal regions of each of the core molecules. These N termini form tails that protrude from the nucleosome core octamer and their acetylation reduces the affinity of the histones for DNA and possibly also reduces the interaction between individual nucleosomes that leads to formation of the 30 nm chromatin fiber Histone acetyltransferases (HATs) – the enzymes that add acetyl groups to histones. Histone acetylation plays a prominent role in regulating genome expression. The tails of the core histones also have attachment sites for methyl and phosphate groups and for the common (‘ubiquitous') protein called ubiquitin. Ubiquitination of histone H2B is part of the general role that ubiquitin plays in control of the cell cycle. Phosphorylation of histone H3 and of the linker histone has been associated with formation of metaphase chromosomes Methylation of a pair of lysine amino acids at the fourth and ninth positions from the Nterminus of histone H3. Methylation of lysine-9 forms a binding site for the HP1 protein which induces chromatin packaging and silences gene expression Methylation of lysine-4 has the opposite effect and promotes an open chromatin structure Lysine-4 methylation is closely correlated with acetylation of histone H3 Two types of modification may work hand in hand to activate regions of chromatin. Remodeling is induced by an energy-dependent process that weakens the contact between the nucleosome and the DNA with which it is associated. Three distinct types of change can occur Remodeling, in the strict sense, involves a change in the structure of the nucleosome, but no change in its position. Sliding, or cis-displacement, physically moves the nucleosome along the DNA Transfer, or trans-displacement, results in the nucleosome being transferred to a second DNA molecule the proteins responsible for nucleosome remodeling work together in large complexes. One of these is Swi/Snf, made up of at least 11 proteins, which is present in many eukaryotes Direct methylation of DNA also has a silencing effect Silencing can be implemented is by removing acetyl groups from histone tails This is the role of the histone deacetylases (HDACs). HDACs are contained in multiprotein complexes. One of these is the mammalian Sin3 complex, which comprises at least seven proteins, including HDAC1 and HDAC2 along with others that do not have deacetylase activity but which provide ancillary functions essential to the process Both Sin3 and NuRD contain proteins that bind to methylated DNA.