* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download capitulo primero
Cytokinesis wikipedia , lookup
SNARE (protein) wikipedia , lookup
Protein (nutrient) wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Signal transduction wikipedia , lookup
Protein phosphorylation wikipedia , lookup
Cytoplasmic streaming wikipedia , lookup
Cell membrane wikipedia , lookup
Protein moonlighting wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Chloroplast wikipedia , lookup
Magnesium transporter wikipedia , lookup
List of types of proteins wikipedia , lookup
Endomembrane system wikipedia , lookup
Western blot wikipedia , lookup
Photosynthesis wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
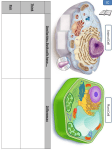
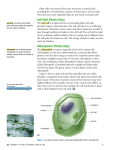
18th International Symposium on Iron Nutrition and Interaction in Plants Madrid – May 30-June 3 2016 PIC1-NICO-YGGT: AN IRON TRANSLOCON AT THE CHLOROPLAST ENVELOPE!? Peter Hagl, Roland Stübe, Cecilia Vasquez-Robinet, Katrin Philippar* DFG-Heisenberg group "Plastid fatty acid & iron transport", Department Biology I, Biocenter LMU Munich, Grosshaderner-Str. 2-4, D-82152 Planegg-Martinsried, Germany *[email protected] Chloroplasts originated about three billion years ago by endosymbiosis of an ancestor of today’s cyanobacteria with a mitochondria-containing host cell. During evolution, chloroplasts of higher plants established as the site for photosynthesis and thus became the basis for all life dependent on oxygen and carbohydrate supply. To fulfill this task, plastid organelles are loaded with the transition metals iron, copper, and manganese, which due to their redox capacity are essential for photosynthetic electron transport. In consequence, chloroplasts represent the Fe--richest system in plant cells. However, improvement of oxygenic photosynthesis in turn required adaptation of metal transport and homeostasis since metalcatalyzed generation of reactive oxygen species (ROS) causes oxidative damage. This is most acute in chloroplasts, where radicals and transition metals are side by side and ROS-production is a usual feature of photosynthetic electron transport. Thus, on the one hand when bound by proteins, chloroplast-intrinsic metals are a prerequisite for photoautotrophic life, but on the other hand become toxic when present in their highly reactive, radical generating, free ionic forms. In consequence, transport, storage and cofactorassembly of metal ions in plastids have to be tightly controlled and are crucial throughout plant growth and development (for overview see López-Millán et al., 2016). The protein PIC1 functions in Fe-uptake across the inner chloroplast envelope. Chlorotic and dwarfish pic1 knockout mutants are reminiscent of Fe-deficiency symptoms and show severely impaired plastid development (Duy et al., 2007). In contrast, PIC1 overexpressing lines display reactive oxygen stress, impaired seed yield and imbalanced Fe-homeostasis (Duy et al., 2011). Our search for PIC1 interaction partners by the yeast Split Ubiquitin system identified the protein NiCo, a putative Ni/Co transporter in the inner chloroplast envelope. To verify PIC1-NiCo protein interaction, we used an immuno-precipitation approach and found YGGTc, a 3rd inner envelope membrane protein. After separation of native inner envelope membrane complexes, we can show that all three proteins PIC1-NiCo-YGGTc run in the same fractions. Furthermore, the TonB box domain of PIC1 is mediating protein-protein interaction. Inducible PIC1and NiCo-RNAi knockdown mutants in Arabidopsis show very similar development of leaf chlorosis and a severely damaged chloroplast ultrastructure. Whereas YGGTc knockouts are embryo lethal, inducible YGGTc knockdown lines are chlorotic in young leaves. Thus, we conclude that these three proteins build an iron translocon complex in the inner chloroplast envelope membrane that might be similar to non-standard prokaryotic ABC transporter complexes. In our proposed model PIC1 represents the permease protein, whereas NiCo functions in metal binding/sensing and YGGTc is stabilizing the complex. chloroplast, Fe-transport, membrane protein complex REFERENCES Duy, D., Wanner, G., Meda, A.R., von Wiren, N., Soll, J., and Philippar, K. (2007). Plant Cell 19, 986-1006. Duy, D., Stübe, R., Wanner, G., and Philippar, K. (2011). Plant Physiol. 155, 1709-1722. López-Millán, A., Duy, D., and Philippar, K. (2016). Front. Plant Sci. 7:178. doi:10.3389/fpls.2016. 00178. ACKNOWLEDGEMENT: This work is supported by the Deutsche Forschungsgemeinschaft (DFG), grants no. PH73/3–3, PH73/6-1 (Heisenberg fellowship) to KP. Preferred Presentation format: POSTER Selected Session: • Fe acquisition, transport and distribution in plants 1