* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Towards the Discovery of New Antimicrobials: the Bifunctional
Cell membrane wikipedia , lookup
Protein phosphorylation wikipedia , lookup
SNARE (protein) wikipedia , lookup
Magnesium transporter wikipedia , lookup
P-type ATPase wikipedia , lookup
Protein moonlighting wikipedia , lookup
Mechanosensitive channels wikipedia , lookup
Signal transduction wikipedia , lookup
Endomembrane system wikipedia , lookup
Protein domain wikipedia , lookup
List of types of proteins wikipedia , lookup
Type three secretion system wikipedia , lookup
Bacterial microcompartment wikipedia , lookup
Lipid bilayer wikipedia , lookup
Theories of general anaesthetic action wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Lipopolysaccharide wikipedia , lookup
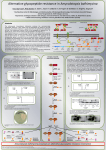
TowardstheDiscoveryofNewAnQmicrobials: theBifuncQonalPenicillinBindingProteins CarminaMicelli,AdrianJ.Lloyd,ChristopherG.DowsonandDavidI.Roper SchoolofLifeSciences,GibbetHillCampus,TheUniversityofWarwick,Coventry,CV47AL StructureandfuncQonofPBPs Results The major component of the bacterial cell wall is a cross-linked glycopep@de polymer calledpep@doglycan(PG).ThePGlayerisan important an@microbial target, due to its essen@alityanduniquenesstobacteria[1]. TPacQve site(S459) UB2H H2O D-Ala D-Ala D-Ala D-Ala m-Dap D-Ala m-Dap m-Dap D-Glu D-Glu D-Glu L-Ala L-Ala L-Ala 20 40 60 GTase 80 100 0 120 0 D-Glu GlcNAc-MurNAc The bifunc@onal penicillin-binding proteins (PBPs) func@on in the last stage of the PG biosynthe@c pathway. Here, the precursor Lipid II (undecaprenyl-pyrophosphorylM u r N A c ( p e n t a p e p @ d e ) - G l c N A c ) i s polymerizedandcross-linkedintoPGbythe glycosyltransferase(GT)andtranspep@dase (TP) domain, respec@vely. The TP domain can also catalyze DD-carboxypep@dase reac@ons, resul@ng in release of the terminalD-Ala[2]. 100 120 30000 PBP1a+10μMMoenomycin PBP1a+10μMMoenomycin 25000 20000 15000 PBP1a 10000 Fluorescence(521nm) PBP1b 80 35000 β-lactaman@bio@csbindtotheTPdomain, causing weakening of the cell wall and eventually bacteria lysis. To escape the ac@on of β-lactams bacteria have evolved resistance mechanisms, for instance the the produc@on of β-lactamases in Gramnega@vebacteria[3]. Escherichiacoli (PDBid:3VMA) 60 L-Ala Fluorescence(521nm) 5000 20000 15000 PBP1a 10000 5000 0 0 0 10 20 30 40 50 0 60 10 20 Time 30 40 50 60 Time Con@nuous fluorescent GTase assay of Pa PBP1a (leb) and Ab PBP1a (right) at 2μM concentra@on. MoenomycinisanMaffinityinhibitoroftheGTdomain,hereusedasacontrol. 0.5 0.5 PBP1a+20μMLipidIIm-DAP 0.4 Absorbance(555nm) 0.4 Aimoftheproject 0.3 0.2 PBP1a+0.1%TritonX-100 0.1 Absorbance(555nm) PBP1a Acinetobacterbaumannii (PDBid:3UDX) 40 Elu@onvolume 25000 GTacQve site(E233) 20 Purifica@onofPaPBP1a(leb,PDBid:4OON)andAbPBP1a(right,PDBid:3UDX).Crystalliza@onofthese proteinsrequiresapoorlycontrolledproteolysisstep[6-7]. m-Dap GlcNAc-MurNAcGlcNAc-MurNAcGlcNAc-MurNAc GTacQve site(E92) 100 Elu@onvolume 0 Post-proteolysis 150 50 100 D-Alam-Dap D-Ala 200 0 300 LipidIIprecursor D-AlaD-Glu D-Ala Absorbance(280) Absorbance(280) L-Ala L-Ala TPase CPase 200 400 Post-proteolysis ---GlcNAc-MurNAcGlcNAc-MurNAc--- Pre-proteolysis 250 500 TPacQve site(S510) ODD Pre-proteolysis 600 PBP1a+20μMLipidIIm-DAP 0.3 0.2 0.1 PBP1a+0.1%TritonX-100 0 0 -0.1 -0.1 0 Structural and kine@c characteriza@on of bifunc@onal PBPs in Gram-nega@ve bacteria, with a primary focus on Pseudomonas aeruginosa (Pa) and Acinetobacter baumannii (Ab), two pathogens responsible for the majority of nosocomialinfec@onsandwithreportedincreasingmul@-drugresistance[4]. This research will provide in vitro data that will help beier elucidate the mechanis@c basis of in vivo catalyzed reac@ons and regula@on of PBPs and, ul@mately, guide the op@miza@on of validated screening assays aimed at discoveringnewPBPinhibitors. 5 10 15 20 25 30 35 40 0 Time 5 10 15 20 25 30 35 40 Time Con@nuousspectrophotometricTPaseassayofPaPBP1a(leb)andAbPBP1a(right)at3μMconcentra@on. 0.1%TritonX-100isthedetergentusedtosolubilizeLipidIIm-DAP,hereusedasacontrol. Conclusionsandfuturework The published crystalliza@on condi@ons for Pseudomonas aeruginosa and AcinetobacterbaumanniiPBP1ahavebeentried,butnocrystalshavegrownsofar. Next,crystalliza@onaiemptswillbefocusedontheGTdomainoftheseproteins. The soluble constructs of PBP1a from Pseudomonas aeruginosa and Acinetobacter baumanniihaveshowntobeac@veinboththeGTaseandTPaseassays. Next,thefull-lengthversionsofPBP1awillbeinves@gated,andwhetherornotthe presence of the transmembrane helix affects the ac@vity of PBP1a in vitro will be evaluated. Methods Fragment-basedscreeningbyproteincrystallography Absorbance(280) References Elu@onvolume Screeningof fragmentlibraries Crystalliza@on Expressionandpurifica@on [1] Brown, L., Wolf, J., Prados-Rosales, R., and Casadevall, A. (2015). Through the wall: extracellular vesicles in Gram-posi@ve bacteria, mycobacteria and fungi. Nature Reviews Microbiology,13(10),pp.620-630. [2]Sauvage,E.,Kerff,F.,Terrak,M.,Ayala,J.andCharlier,P.(2008).Thepenicillin-bindingproteins:structureandroleinpep@doglycanbiosynthesis.FEMSMicrobiologyReviews, 32(2),pp.234-258. [3]Zapun,A.,Contreras-Martel,C.,andVernet,T.(2008).Penicillin-bindingproteinsandbeta-lactamresistance.FEMSMicrobiologyReviews,32(2),pp.361-385. [4]Zavascki,P.,Carvalhaes,G.,Picão,C.,andGales,C.(2010).Mul@drug-resistantPseudomonasaeruginosaandAcinetobacterbaumannii:resistancemechanismsandimplica@onsfor therapy.ExpertReviewofAnD-InfecDveTherapy,8(1),pp.71-93. [5]Schwartz,B.,Markwalder,J.,Seitz,S.,Wang,Y.,andStein,R.(2002).Akine@ccharacteriza@onoftheglycosyltransferaseac@vityofEschericiacoliPBP1banddevelopmentofa con@nuousfluorescenceassay.Biochemistry,41(41),pp.12552-12561. [6]Starr,J.,Brown,M.,Aschenbrenner,L.,Caspers,N.,Che,Y.,Gerstenberger,B.,Huband,M.,Knafels,J.,Lemmon,M.,Li,C.,McCurdy,S.,McElroy,E.,Rauckhorst,M.,Tomaras,A., Young,J.,Zaniewski,R.,Shanmugasundaram,V.,andHan,S.(2014).Siderophorereceptor-mediateduptakeoflac@[email protected] Chemistry,57(9),pp.3845-3855. [7] Han, S., Caspers, N., Zaniewski, R., Lacey, B., Tomaras, A., Feng, X., Geoghegan, K., and Shanmugasundaram, V. (2011). Dis@nc@ve aiributes of β-lactam target proteins in Acinetobacterbaumanniirelevanttodevelopmentofnewan@[email protected],133(50),pp.20536-20545. Hitiden@fica@on Medicinalchemistry InvitroPGsynthesisassays L-Ala m-Dap D m-Dap D D-Ala D-Ala D-Ala Highfluorescence Lowfluorescence D-Ala ---GlcNAc-MurNAc--- Noenzyme GTase C o n @ n u o u s G T a s e assay with dansylated LipidIIsubstrate[5]. Time ---GlcNAc-MurNAc--- L-Ala L-Ala TPase m-Dap D-Glu L-Ala ---GlcNAc-MurNAc--- D-Glu m-Dap D-Ala D-Ala m-Dap D-Glu Absorbance D-Ala m-Dap D-Ala D-Glu D-Ala L-Ala D-Ala D-Ala GlcNAc-MurNAc D-Glu m-Dap D muramidase PG chains D-Glu D-Glu GTase GlcNAc-MurNAc L-Ala Fluorescence ---GlcNAc-MurNAc CPase/TPase Nolipid L-Ala ---GlcNAc-MurNAc--- Time Con@nuous CPase/TPase assay with Lipid II mDAPsubstratedevelopedbyDr.AdrianJ.Lloyd. Acknowledgements ThisresearchisfundedbyBBSRC.ThanktoalltheC10labmembersfortheirsupport