* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Supplementary Information (docx 341K)
Gene therapy wikipedia , lookup
Copy-number variation wikipedia , lookup
Non-coding DNA wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Cancer epigenetics wikipedia , lookup
Skewed X-inactivation wikipedia , lookup
Transposable element wikipedia , lookup
Genetic engineering wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
Y chromosome wikipedia , lookup
Human genome wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Oncogenomics wikipedia , lookup
Essential gene wikipedia , lookup
Gene desert wikipedia , lookup
Public health genomics wikipedia , lookup
Pathogenomics wikipedia , lookup
Long non-coding RNA wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Quantitative trait locus wikipedia , lookup
History of genetic engineering wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Polycomb Group Proteins and Cancer wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
X-inactivation wikipedia , lookup
Biology and consumer behaviour wikipedia , lookup
Microevolution wikipedia , lookup
Ridge (biology) wikipedia , lookup
Genomic imprinting wikipedia , lookup
Designer baby wikipedia , lookup
Gene expression programming wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Minimal genome wikipedia , lookup
Genome evolution wikipedia , lookup
Epigenetics of human development wikipedia , lookup
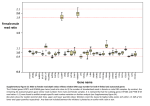
SUPPLEMENTARY INFORMATION SUPPLEMENTARY METHODS Topological Domains and Genes Predicted to Show Haploinsufficiency Bordering DGAP242 Translocation Breakpoints Six Mb windows surrounding the chromosomal breakpoints were analyzed for topologically associating domains (TADs), gene annotation, predicted haploinsufficiency (HI), OMIMassociated phenotypes and inner ear expression. TADs were predicted using human embryonic stem cell Hi-C domains from the Hi-C project (chromosome.sdsc.edu) and converted to hg19 for comparison to the breakpoints.1 Gene annotation was obtained from the University of California Santa Cruz Genome Browser (genome.ucsc.edu).2 If the breakpoint occurred within the open reading frame of any gene, that gene was considered disrupted. If a gene resided within the TAD, it was evaluated for potential dysregulation. To assess pathogenicity, HI prediction was downloaded from DECIPHER (decipher.sanger.ac.uk).3 Only disrupted or dysregulated genes with HI scores of ≤10% were considered as possibly pathogenic. Inner ear cell-type specific gene expression was determined using the Shared Harvard Inner Ear Laboratory Database (SHIELD) (shield.hms.harvard.edu).4,5 Possible effects of disrupted or dysregulated pathogenic genes were determined by assessment of OMIM phenotypes from the Online Mendelian Inheritance in Man Catalog of Human Genes and Genetic Disorders (www.omim.org) and mapping of phenotypes to genes implicated within TAD regions.6 1 SUPPLEMENTARY FIGURES Supplementary Figure 1. Topologically associating domains (TADs) disrupted by DGAP242’s chromosomal translocation and genes predicted to show haploinsufficiency (HI). The top row is the chromosome section, containing the banding patterns of the 6 Mb chromosome region surrounding the breakpoint (red arrowhead). The second row includes genes present in that section of the chromosome (green rectangles). The third row corresponds to predicted HI genes (red rectangles). The fourth row includes the topological domain data, with blue rectangles representing Hi-C domains, and black rectangles representing domain boundary regions, or regions where no contacts were detected. Units are in bp x 107. Panel A: Chromosome 1 (chr1) TAD and HI presence. Predicted HI genes are PROX1, USH2A (OMIM #276901), ESRRG and TGFB2 (OMIM #614816), which are located ~2.7, ~1.1, 0, and ~1.5 Mb from the translocation breakpoint, respectively. Note that ESRRG is directly disrupted by the translocation. Panel B: Chromosome 5 (chr5) TAD and HI presence. Predicted HI genes are NR2F1 (OMIM #615722), FAM172A, KIAA0825, ANKRD32 and MCTP1, which are located ~625, ~108, 0, ~519, and ~483 kb from the translocation breakpoint, respectively. Note that KIAA0825 is directly disrupted by the translocation. Gene transcript orientation for haploinsufficient genes is denoted by a forward arrow (encoded on the positive reference strand) or reverse arrow (encoded on the negative reference strand). 2 3 SUPPLEMENTARY TABLES Supplementary Table 1. Anchored primers for targeted locus amplification Sequence (5’-3’) Name Target region TLA1a chr1_downstream_fw1 AACATTTACAAATCCCCAACAC TLA1b chr1_downstream_rev1 GGATCAGTGGTGCAGAAATA TLA2a chr1_upstream_fw1 GGACAGATTTGCACTTGTTAC TLA2b chr1_upstream_rev1 CTTGAAATCGCCAGCATTTG TLA3a chr5_downstream_fw1 TCAAGGTAATGAAATTCAGGGA TLA3b chr5_downstream_rev1 GTTCTCACTACTGACACCTTAA TLA4a chr5_upstream_fw1 TCAAGGAGTAAGACCCAGAG TLA4b chr5_upstream_rev1 AGGGTCTGGGTGCATATATT 4 Supplementary Table 2. Expression of candidate genes in mouse inner ear hair cells in ascending order of false discovery rate (FDR). Genes that both reside within a TAD disrupted by the breakpoints and have a HI score ≤10% were assessed for enriched expression in hair cells, using the SHIELD database (shield.hms.harvard.edu).4,5 An asterisk (*) denotes significantly enriched expression levels in inner ear hair cells (FDR < 0.01). Gene Fold Enrichment in Hair Cells False Discovery Rate (FDR) USH2A* 24.76 2.91E-05 FAM172A* 2.93 2.18E-03 ESRRG* 4.55 8.20E-03 MCTP1 1.16 5.97E-02 PROX1 5.48 7.11E-02 KIAA0825 1.32 3.14E-01 ANKRD32 2.39 3.33E-01 NR2F1 1.23 6.80E-01 TGFB2 1.85 7.43E-01 5 SUPPLEMENTARY DISCUSSION Assessment of candidate genes in the breakpoint-containing TADs When we extended our analysis to the identification of possible dysregulated genes by examining the TADs disrupted by the breakpoints and identifying genes with a HI score ≤10%, several additional predicted HI genes were identified within the chr1 and chr5 TAD breakpoint regions: PROX1, USH2A, TGFB2, NR2F1, FAM172A, ANKRD32 and MCTP1 (Supplementary Figure 1). Of these candidates, only USH2A, TGFB2 and NR2F1 are associated with an OMIM phenotype (#276901 Usher Syndrome, Type IIA, USH2A; #614816 Loeys-Dietz Syndrome 4, LDS4; and #615722 Bosch-Boonstra-Schaaf Optic Atrophy Syndrome, BBSOAS; respectively). Of all three diseases, only Usher Syndrome has a phenotype of SNHL. When we analyzed the inner ear expression pattern of these genes using data from the SHIELD database, only USH2A and FAM172A have significantly enriched expression levels in inner ear hair cells, suggesting a potential role for these genes in HL (Supplementary Table 2).4,5 However, of these genes, FAM172A is not predicted to be associated with pathogenicity and USH2A is associated with an autosomal recessive disorder, which is inconsistent with the predicted HI score (%HI = 4.17) and any dysregulation that may have occurred by the heterozygous chromosomal rearrangement in DGAP242. Etiology of the t(1;5) The cause of DGAP242’s de novo translocation is unknown. One possibility is that the AT-rich nature of the sequences at the chromosome 1 and 5 breakpoints made them more susceptible to rearrangement. AT-rich sequences have been hypothesized to induce genome instability by forming vulnerable secondary structures. Lesions in these regions could then be repaired by the 6 non-homologous end-joining repair pathway, leading to improper ligation of non-homologous chromosomes.7 Another possibility is that this translocation occurred by a microhomologymediated repair mechanism, which has been reported to underpin many non-recurrent translocations.8 It has not escaped our attention that a 16 bp region of microhomology (TGACCTTGGACAGCTG) resides 55 bp downstream of the chromosome 1 breakpoint and 643 bp downstream of the chromosome 5 breakpoint. 7 SUPPLEMENTARY REFERENCES 1. Dixon JR, Selvaraj S, Yue F et al: Topological domains in mammalian genomes identified by analysis of chromatin interactions. Nature 2012; 485: 376-380. 2. Rosenbloom KR, Armstrong J, Barber GP et al: The UCSC Genome Browser database: 2015 update. Nucleic Acids Res 2015; 43: D670-681. 3. Huang N, Lee I, Marcotte EM, Hurles ME: Characterising and predicting haploinsufficiency in the human genome. PLoS Genet 2010; 6: e1001154. 4. Scheffer DI, Shen J, Corey DP, Chen ZY: Gene Expression by Mouse Inner Ear Hair Cells during Development. J Neurosci 2015; 35: 6366-6380. 5. Shen J, Scheffer DI, Kwan KY, Corey DP: SHIELD: an integrative gene expression database for inner ear research. Database (Oxford) 2015; 2015: bav071. 6. Online Mendelian Inheritance in Man, OMIM®. Johns Hopkins University (Baltimore, MD): McKusick-Nathans Institute of Genetic Medicine, 2015. 7. Kato T, Kurahashi H, Emanuel BS: Chromosomal translocations and palindromic ATrich repeats. Curr Opin Genet Dev 2012; 22: 221-228. 8 8. Verdin H, D'Haene B, Beysen D et al: Microhomology-mediated mechanisms underlie non-recurrent disease-causing microdeletions of the FOXL2 gene or its regulatory domain. PLoS Genet 2013; 9: e1003358. 9