* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Table of Contents - Milan Area Schools
Fatty acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Metabolic network modelling wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Phosphorylation wikipedia , lookup
Blood sugar level wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Electron transport chain wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Citric acid cycle wikipedia , lookup
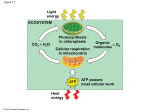
Cellular Pathways that Harvest Chemical Energy 7 Cellular Pathways that Harvest Chemical Energy • Energy and Electrons from Glucose • Glycolysis: From Glucose to Pyruvate • Pyruvate Oxidation • The Citric Acid Cycle • The Respiratory Chain: Electrons, Protons, and ATP Production • Fermentation: ATP from Glucose, without O2 • Contrasting Energy Yields • Relationships between Metabolic Pathways • Regulating Energy Pathways 7 Energy and Electrons from Glucose • The sugar glucose (C6H12O6) is the most common form of energy molecule. • Cells obtain energy from glucose by the chemical process of oxidation in a series of metabolic pathways. 7 Energy and Electrons from Glucose • Principles governing metabolic pathways: Metabolic pathways are formed by complex chemical transformations which occur in separate reactions. Each reaction in the pathway is catalyzed by a specific enzyme. Metabolic pathways are similar in all organisms. In eukaryotes, many metabolic pathways are compartmentalized in organelles. The operation of each metabolic pathway can be regulated by the activities of key enzymes. 7 Energy and Electrons from Glucose • When burned in a flame, glucose releases heat, carbon dioxide, and water. C6H12O6 + 6 O2 6 CO2 + 6 H2O + energy • The same equation applies for the biological, metabolic use of glucose. 7 Energy and Electrons from Glucose • About half of the energy from glucose is collected in ATP. • G for the complete conversion of glucose is –686 kcal/mol. • The reaction is therefore highly exergonic, and it drives the endergonic formation of ATP. 7 Energy and Electrons from Glucose • Three metabolic processes are used in the breakdown of glucose for energy: Glycolysis Cellular respiration Fermentation Figure 7.1 Energy for Life 7 Energy and Electrons from Glucose • Glycolysis produces some usable energy and two molecules of a three-carbon sugar called pyruvate. • Glycolysis begins glucose metabolism in all cells. • Glycolysis does not require O2; it is an anaerobic metabolic process. 7 Energy and Electrons from Glucose • Cellular respiration uses O2 and occurs in aerobic (oxygen-containing) environments. • Pyruvate is converted to CO2 and H2O. • The energy stored in covalent bonds of pyruvate is used to make ATP molecules. 7 Energy and Electrons from Glucose • Fermentation does not involve O2. It is an anaerobic process. • Pyruvate is converted into lactic acid or ethanol. • Breakdown of glucose is incomplete; less energy is released than by cellular respiration. 7 Energy and Electrons from Glucose • Redox reactions transfer the energy of electrons. • A gain of one or more electrons or hydrogen atoms is called reduction. • The loss of one or more electrons or hydrogen atoms is called oxidation. • Whenever one material is reduced, another is oxidized. Figure 7.2 Oxidation and Reduction Are Coupled 7 Energy and Electrons from Glucose • An oxidizing agent accepts an electron or a hydrogen atom. • A reducing agent donates an electron or a hydrogen atom. • During the metabolism of glucose, glucose is the reducing agent (and is oxidized), while oxygen is the oxidizing agent (and is reduced). 7 Energy and Electrons from Glucose • The coenzyme NAD is an essential electron carrier in cellular redox reactions. • NAD exists in an oxidized form, NAD+, and a reduced form, NADH + H+. • The reduction reaction requires an input of energy: NAD+ + 2H NADH + H+ • The oxidation reaction is exergonic: NADH + H+ + ½ O2 NAD+ + H2O Figure 7.3 NAD Is an Energy Carrier Figure 7.4 Oxidized and Reduced Forms of NAD 7 Energy and Electrons from Glucose • The energy-harvesting processes in cells use different combinations of metabolic pathways. • With O2 present, four major pathways operate: Glycolysis Pyruvate oxidation The citric acid cycle The respiratory chain (electron transport chain) • When no O2 is available, glycolysis is followed by fermentation. Table 7.1 Cellular Locations for Energy Pathways in Eukaryotes and Prokaryotes 7 Glycolysis: From Glucose to Pyruvate • Glycolysis can be divided into two stages: Energy-investing reactions that use ATP Energy-harvesting reactions that produce ATP 7 Glycolysis: From Glucose to Pyruvate • The energy-investing reactions of glycolysis: In separate reactions, two ATP molecules are used to make modifications to glucose. Phosphates from each ATP are added to the carbon 6 and carbon 1 of the glucose molecule to form fructose 1,6-bisphosphate. The enzyme aldolase splits the molecule into two 3-C molecules that become glyceraldehyde 3phosphate (G3P). Figure 7.6 Glycolysis Converts Glucose to Pyruvate (Part1) Figure 7.6 Glycolysis Converts Glucose to Pyruvate (Part2) 7 Glycolysis: From Glucose to Pyruvate • The energy-harvesting reactions of glycolysis: The first reaction (an oxidation) releases free energy that is used to make two molecules of NADH + H+, one for each of the two G3P molecules. Two other reactions each yield one ATP per G3P molecule. This part of the pathway is called substrate-level phosphorylation. The final product is two 3-carbon molecules of pyruvate. Figure 7.6 Glycolysis Converts Glucose to Pyruvate (Part3) Figure 7.6 Glycolysis Converts Glucose to Pyruvate (Part 4) Figure 7.7 Changes in Free Energy During Glycolysis 7 Pyruvate Oxidation • Pyruvate is oxidized to acetate which is converted to acetyl CoA. • Pyruvate oxidation is a multistep reaction catalyzed by an enzyme complex attached to the inner mitochondrial membrane. • The acetyl group is added to coenzyme A to form acetyl CoA. One NADH + H+ is generated during this reaction. Figure 7.8 Pyruvate Oxidation and the Citric Acid Cycle (Part 1) 7 The Citric Acid Cycle • The citric acid cycle begins when the two carbons from the acetate are added to oxaloacetate, a 4-C molecule, to generate citrate, a 6-C molecule. • A series of reactions oxidize two carbons from the citrate. With molecular rearrangements, oxaloacetate is formed, which can be used for the next cycle. • For each turn of the cycle, three molecules of NADH + H+, one molecule of ATP, one molecule of FADH2, and two molecules of CO2 are generated. Figure 7.8 Pyruvate Oxidation and the Citric Acid Cycle (Part 2) 7 The Respiratory Chain: Electrons, Protons, and ATP Production • The respiratory chain uses the reducing agents generated by pyruvate oxidation and the citric acid cycle. • The flow of electrons in a series of redox reactions causes the active transport of protons across the inner mitochondrial membrane, creating a proton concentration gradient. • The protons then diffuse through proton channels down the concentration and electrical gradient back into the matrix of the mitochondria, creating ATP in the process. • ATP synthesis by electron transport is called oxidative phosphorylation. 7 The Respiratory Chain: Electrons, Protons, and ATP Production • The respiratory chain consists of four large protein complexes bound to the inner mitochondrial membrane, plus cytochrome c and ubiquinone (Q). Figure 7.10 The Oxidation of NADH + H+ 7 The Respiratory Chain: Electrons, Protons, and ATP Production • NADH + H+ passes its hydrogen atoms to the NADH-Q reductase protein complex. • The NADH-Q reductase passes the hydrogens on to ubiquinone (Q), forming QH2. • The QH2 passes electrons to cytochrome c reductase complex which in turn passes them to cytochrome c. • Next to receive them is cytochrome c oxidase complex. Then they are passed to O2. • Reduced oxygen unites with two hydrogen ions to form water. Figure 7.11 The Complete Respiratory Chain 7 The Respiratory Chain: Electrons, Protons, and ATP Production • As electrons pass through the respiratory chain, protons are pumped by active transport into the intermembrane space against their concentration gradient. • This transport results in a difference in electric charge across the membrane. • The potential energy generated is called the proton-motive force. 7 The Respiratory Chain: Electrons, Protons, and ATP Production • Chemiosmosis is the coupling of the protonmotive force and ATP synthesis. • NADH + H+ or FADH2 yield energy upon oxidation. • The energy is used to pump protons into the intermembrane space, contributing to the protonmotive force. • The potential energy from the proton-motive force is harnessed by ATP synthase to synthesize ATP from ADP. Figure 7.12 A Chemiosmotic Mechanism Produces ATP (Part 1) Figure 7.12 A Chemiosmotic Mechanism Produces ATP (Part 2) 7 The Respiratory Chain: Electrons, Protons, and ATP Production • Synthesis of ATP from ADP is reversible. • The synthesized ATP is transported out of the mitochondrial matrix as quickly as it is made. • The proton gradient is maintained by the pumping of the electron transport chain. 7 The Respiratory Chain: Electrons, Protons, and ATP Production • Two key experiments demonstrated that: A proton gradient across a membrane can drive ATP synthesis. The enzyme ATP synthase is the catalyst for this reaction. Figure 7.13 Two Experiments Demonstrate the Chemiosmotic Mechanism 7 Fermentation: ATP from Glucose, without O2 • When there is an insufficient supply of O2, a cell cannot reoxidize cytochrome c. • Then QH2 cannot be oxidized back to Q, and soon all the Q is reduced. • This continues until the entire respiratory chain is reduced. • NAD+ and FAD are not generated from their reduced form. • Pyruvate oxidation stops, due to a lack of NAD+. • Likewise, the citric acid cycle stops, and if the cell has no other way to obtain energy, it dies. 7 Fermentation: ATP from Glucose, without O2 • Some cells under anaerobic conditions continue glycolysis and produce a limited amount of ATP if fermentation regenerates the NAD+ to keep glycolysis going. • Fermentation uses NADH + H+ to reduce pyruvate, and consequently NAD+ is regenerated. 7 Fermentation: ATP from Glucose, without O2 • Some organisms are confined to anaerobic environments and use only fermentation. These organisms lack the molecular machinery for oxidative phosphorylation. They also lack enzymes to detoxify the toxic by-products of O2, such as H2O2. 7 Fermentation: ATP from Glucose, without O2 • In lactic acid fermentation, an enzyme, lactate dehydrogenase, uses the reducing power of NADH + H+ to convert pyruvate into lactate. • NAD+ is replenished in the process. • Lactic acid fermentation occurs in some microorganisms and in muscle cells when they are starved for oxygen. Figure 7.14 Lactic Acid Fermentation 7 Fermentation: ATP from Glucose, without O2 • Alcoholic fermentation involves the use of two enzymes to metabolize pyruvate. • First CO2 is removed from pyruvate, producing acetaldehyde. • Then acetaldehyde is reduced by NADH + H+, producing NAD+ and ethanol. Figure 7.15 Alcoholic Fermentation 7 Contrasting Energy Yields • A total of 36 ATP molecules can be generated from each glucose molecule in glycolysis and cellular respiration. • Each NADH + H+ generates 3 ATP molecules, and each FADH2 generates 2 ATP by the chemiosmotic mechanism. • Fermentation has a net yield of 2 ATP molecules from each glucose molecule. • The end products of fermentation contain much more unused energy than the end products of aerobic respiration. Figure 7.16 Cellular Respiration Yields More Energy Than Glycolysis Does (Part 1) Figure 7.16 Cellular Respiration Yields More Energy Than Glycolysis Does (Part 2) 7 Relationships between Metabolic Pathways • Glucose utilization pathways can yield more than just energy. They are interchanges for diverse biochemical traffic. • Intermediate chemicals are generated that are substrates for the synthesis of lipids, amino acids, nucleic acids, and other biological molecules. Figure 7.17 Relationships Among the Major Metabolic Pathways of the Cell 7 Relationships between Metabolic Pathways • Catabolic interconversions: Polysaccharides are hydrolyzed into glucose, which passes on to glycolysis. Lipids are converted to fatty acids, which become acetate (then acetyl CoA), and glycerol, which is converted to an intermediate in glycolysis. Proteins are hydrolyzed into amino acids, which feed into glycolysis or the citric acid cycle. 7 Relationships between Metabolic Pathways • Anabolic interconversions: Gluconeogenesis is the process by which intermediates of glycolysis and the citric acid cycle are used to form glucose. Acetyl CoA can form fatty acids. Intermediates can form amino acids. The citric acid cycle intermediate aketoglutarate is the starting point for the synthesis of purines. Oxaloacetate is a starting point for pyrimidines. 7 Relationships between Metabolic Pathways • The levels of the products and substrates of energy metabolism are remarkably constant. • Cells regulate the enzymes of catabolism and anabolism to maintain a balance, or metabolic homeostasis. 7 Relationships between Metabolic Pathways • What happens if inadequate food molecules are available? Glycogen stores in muscle and liver are used first. Fats are used next. But the brain can only use glucose, so it must be synthesized by gluconeogenesis which uses mostly amino acids. Therefore, proteins must be broken down. After fats are depleted, proteins alone provide energy. 7 Regulating Energy Pathways • Metabolic pathways work together to provide cell homeostasis. • Positive and negative feedback control whether a molecule of glucose is used in anabolic or catabolic pathways. Figure 7.19 Regulation by Negative and Positive Feedback 7 Regulating Energy Pathways • The amount and balance of products a cell has is regulated by allosteric control of enzyme activities. • Control points use both positive and negative feedback mechanisms. • The main control point in glycolysis is the enzyme phosphofructokinase. • This enzyme is inhibited by ATP and activated by ADP and AMP. 7 Regulating Energy Pathways • The main control point of the citric acid cycle is the enzyme isocitrate dehydrogenase which converts isocitrate to a-ketoglutarate. • NADH + H+ and ATP are inhibitors of this enzyme. NAD+ and ADP are activators of it. • Accumulation of isocitrate and citrate occurs, but is limited by the inhibitory effects of high ATP and NADH. • Citrate acts as an additional inhibitor to slow the fructose 6-phosphate reaction of glycolysis and also switches acetyl CoA to the synthesis of fatty acids. Figure 7.20 Feedback Regulation of Glycolysis and the Citric Acid Cycle (Part 1) Figure 7.20 Feedback Regulation of Glycolysis and the Citric Acid Cycle (Part 2)