* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 5-8_PathEvByCertainTransmitter_SomorjaiD
Survey
Document related concepts
Cell membrane wikipedia , lookup
G protein–coupled receptor wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell growth wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Endomembrane system wikipedia , lookup
Cytokinesis wikipedia , lookup
Purinergic signalling wikipedia , lookup
Chemical synapse wikipedia , lookup
Glutamate receptor wikipedia , lookup
List of types of proteins wikipedia , lookup
NMDA receptor wikipedia , lookup
Transcript
Pathological events maybe generated by certain transmitter(s).
Somorjai Dávid GZGU7X 2016.10.13.
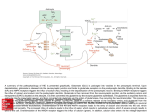
Neurotransmitters are endogenous bioactive substances synthesized by neurons. They are released from the cells, act via speci_c receptors coupled to
the membrane of postsynaptic target structures and modify the electric and
metabolic conditions of the a_ected cells.
types of neurotransmitters:
1. (classic) amine type:
{ production of: synthesizing enzymes
{ axonal transport of: synthesizing enzymes, storage vesicles
{ stored in: vesicles (40-50 nm)
{ supply of constituents: from cell body, local synthesis, re-uptake
2. peptid type:
{ production of: peptide precursors + converting enzymes
{ axonal transport of: storage vesicles
{ stored in: vesicles (80-200 nm - dense core vesicles)
{ supply of constituents: from cell body
Receptors are complex proteins that show high-affnity binding of transmitter
ligands
types of receptors:
1. ionotropic receptors:
{ speci_c ion channels in the membrane that allow the passage of ions
upon activation
{ fast postsynaptic response
2. metabotropic receptors/GPCRs (G Protein-Coupled Receptors):
{ coupled to G proteins via the intracellular domain
G protein activation evokes secondary responses in the cell that alter
their metabolism
{ slower postsynaptic response (and longer duration)
glutamate changes the flow of ions across the membrane of the postsynaptic cell. Usually
glutamate’s influence on the postsynaptic cell is excitatory; i.e. glutamate tends to cause the
postsynaptic neuron to fire an action potential and to propagate a signal to the next
neuron in the circuit.
the N-methyl-d-aspartate (NMDA) receptor, which has been implicated in memory,
development and epilepsy. Although active when applied, the amino acid aspartate is not
thought to be released as a neurotransmitter. By activating these receptors, the large amount
of glutamate released into the brain during a stroke or trauma can cause tissue damage, a
process called excitotoxicity. There are also at least sixmetabotropic receptors for glutamate.
The term excitotoxicity was first used by Olney (1986) to describe the ability of L-glu, as well
as structurally related amino acids, to kill nerve cells, a process that has been proposed to take
place not only in acute but also chronic diseases of the central nervous system
Excitotoxicity results from the excessive activation of iGluRs and leads to a characteristic loss
of post-synaptic structures including dendrites and cell bodies. Moreover, the susceptibility of
neurons to excitotoxicity can change tremendously with age. Acute excitotoxic nerve cell
death is thought to occur in response to a variety of severe insults including cerebral ischemia,
traumatic brain injury (TBI), hypoglycemia, and status epilepticus
It’s more difficult to study chronic excitotoxicity in culture partly because it is not entirely clear
how to define “chronic” in the context of cell culture. Does chronic mean a low dose given for
24 h rather than a higher dose given for 5–10 min or is it more complicated than that?
In summary, increases in extracellular L-glu in vivo can affect nerve cell survival and brain function. The
consequences seem to be highly dependent on the degree of L-glu increase but even a 10% increase
appears to affect nerve cell structure and survival particularly in the context of aging suggesting that
chronic excitotoxicity may be particularly relevant to age-related neurodegenerative diseases.
Excitotoxicity Theory
Glutamate exerts its effects by binding to specific receptors on nerve cells. There are various
types of glutamate receptors. Here we will concern ourselves with two types of glutamate
receptors, which are called the NMDA receptor and the non-NMDA receptor. These
receptors all contain glutamate-binding sites. Once glutamate binds to the receptor,
glutamate “excites” the cells by causing positive ions to flow into the cell, increasing the
cell’s electrical charge. The increased charge triggers changes in the neuron that ultimately
result in the release of many neurotransmitters at the end of the cell.
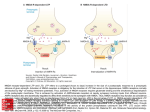
NMDA receptors have the special ability to let in large amounts of calcium ions (Ca2+), its opening is
blocked by a single magnesium ion (Mg2+). The NMDA receptor needs both the binding of glutamate
and an increase in cell charge before it opens. Because the non-NMDA receptors are not blocked, the
binding of glutamate alone opens these receptors and allows positively charged ions to flow into the
cell. Ion pumps present in the cells remove some of the positive ions, preventing the charge inside
the cell from rising too quickly. These ion pumps will only work if there is sufficient amount of energy
in the cell.
In HD nerve cells, in contrast, the lower amount of energy available reduces the ability of the ion
pumps to prevent a rapid increase in cell voltage
Excitotoxicity results from the excessive activation of iGluRs and leads to a characteristic loss
of post-synaptic structures including dendrites and cell bodies.
Acute excitotoxic nerve cell death is thought to occur in response to a variety of severe insults
including cerebral ischemia, traumatic brain injury (TBI), hypoglycemia, and status epilepticus