* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download File
Cell culture wikipedia , lookup
Cell nucleus wikipedia , lookup
Membrane potential wikipedia , lookup
Cellular differentiation wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cell growth wikipedia , lookup
Cell encapsulation wikipedia , lookup
Signal transduction wikipedia , lookup
Cytokinesis wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell membrane wikipedia , lookup
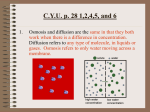
%@*&$* Diffusion and Osmosis Lesson Objectives • • • • • By the end of this lesson you should know that: Cell membranes are semi – permeable Understand the term diffusion Distinguish between a solvent, solute and solution Define osmosis Understand the theory of osmosis Selective permeability of membranes • Distinguish between permeable and impermable!! • Cellophane, visking tubing and dialysis tubing are artificial semi permeable membranes – what about the cell membrane? • Water, oxygen and carbon – dioxide can all pass across these membranes • Other substances have more difficulty What is diffusion? “Diffusion is the movement of the molecules of a liquid or a gas from a region of high concentration to a region of low concentration” • This difference in concentration of gases or liquids between two areas is called the concentration gradient • Diffusion will always take place when such a gradient occurs • This is a passive process – which means it does not require an external source of energy Animation: How Diffusion Works Distinguish between • Solute • Solvent • Solution Examples of diffusion Selectively (semi) permeable membranes • All membranes in biology are similar – around a chloroplast, mitochondrion or the nucleus • They are all semi or selectively permeable? • This simply means that some molecules can pass across the membrane while others cannot so easily • Water, carbon dioxide and oxygen do so easily • Salts, sugars and proteins cannot so easily Osmosis “Osmosis is the movement of water from a region of high water concentration to a region of low water concentration across a semi or selectively permeable membrane” Comparing diffusion and osmosis • Both diffusion and osmosis involve the movement of molecules from regions of high concentration to regions of low concentrations – both processes are passive • Difference is that osmosis requires a semi – permeable membrane while diffusion does not – osmosis is a special case of diffusion Theory of osmosis http://www.stolaf.edu/people/giannini/flashanimat/transport/osmosis.swf BIOL 230 Lecture Guide - Osmosis Animation Lesson Objectives By the end of this lesson you should know that: • The effects of placing animal and plant cells in different solutions • What turgor pressure is with respect to a plant cell • Osmosis and its application of food preservation Osmosis Osmoregulation in Amoeba proteus Osmosis and plant cells Overlay - Animation of plasmolysis Lesson objectives By the end of this lesson you should be able to: • Describe the role of salt or sugar in food preservation • Conduct an activity to demonstrate osmosis • Define and describe active transport Osmosis and food preservation • • • • 1. 2. Bacteria and fungi are similar to plants in that they are enclosed by a cell wall Osmosis is often used to prevent micro – organisms such as bacteria and fungi from growing on food This prevents them from producing harmful toxins and from decaying the food 2 examples of food preservation techniques are Fish and meat in a salty solution Jams and marmalades Active transport • Many cells take in chemicals which are already in high concentration within the cell • Diffusion would cause these chemicals to leave the cell?? • Active transport is the process where chemicals are taken into the cell against the diffusion gradient • Requires energy and is undertaken by proteins in the cell membrane http://www.stolaf.edu/people/giannin i/flashanimat/transport/secondary% 20active%20transport.swf