* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download patriciazuk.com
Survey
Document related concepts
Cell membrane wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell nucleus wikipedia , lookup
Extracellular matrix wikipedia , lookup
Signal transduction wikipedia , lookup
Endomembrane system wikipedia , lookup
Cell culture wikipedia , lookup
Programmed cell death wikipedia , lookup
Kinetochore wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cellular differentiation wikipedia , lookup
Spindle checkpoint wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Transcript
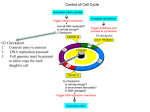
LECTURE PRESENTATIONS For CAMPBELL BIOLOGY, NINTH EDITION Jane B. Reece, Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Robert B. Jackson Chapter 12 & 18 Control of DNA Lectures by Erin Barley Kathleen Fitzpatrick © 2011 Pearson Education, Inc. Eukaryotic Cell Cycle Control • the frequency of cell division varies with the type of cell – differences result from regulation at the molecular level – cancer cells manage to escape the usual controls on the cell cycle • cell cycle is driven by specific chemical signals present in the cytoplasm EXPERIMENT Experiment 1 S G1 S S Experiment 2 M G1 RESULTS When a cell in the S phase was fused with a cell in G1, the G1 nucleus immediately entered the S phase—DNA was synthesized. M M When a cell in the M phase was fused with a cell in G1, the G1 nucleus immediately began mitosis—a spindle formed and chromatin condensed, even though the chromosome had not been duplicated. Phases of the Cell Cycle • consists of two phases – Mitotic (M) phase = mitosis and cytokinesis) – Interphase = cell growth and copying of chromosomes in preparation for cell division • Interphase - about 90% of the cell cycle – – – – can be divided into sub-phases G1 phase -“first gap” S phase “synthesis” G2 phase - “second gap” Phases of the Cell Cycle – G1 phase - time in phase depends on species • • • • normal cell functions growth in size mRNA and protein synthesis in preparation for S phase critical phase in which cell commits to division or leaves the cell cycle to enter into a dormancy phase (G0) – S phase - 6 to 8 hours • synthesis of histone proteins & DNA replication – G2 phase – 2 to 5 hours • • • rapid cell growth – may function to simply control cell size protein synthesis in preparation for M phase duplication of the centrioles/centrosomes http://www.wisconline.com/objects/in dex.asp?objID=AP136 04 Some terms to know -parent cell = cell about to undergo division -daughter cell = cell that results from either mitosis or meiosis -somatic cell = any cell within the body other than an egg or sperm -somatic cell has two complete sets of chromosomes -one set is called the haploid number of chromosomes (n) -therefore the cell is said to be diploid (2n) e.g. humans n = 23 (2n = 46) -germ cell or gamete = sex cell -gamete has only one set of chromosomes and is haploid Cellular Organization of the Genetic Material • all the DNA in a cell constitutes the cell’s genome • REMINDER: eukaryotic chromosomes consist of chromatin, a complex of DNA and protein that condenses during cell division • in most cells - DNA molecules in a cell are condensed and packaged into chromosomes • prokaryotics have a single chromosome called a genophore • eukaryotic cells posses number of chromosomes 20 m every eukaryotic species has a characteristic number of chromosomes in each cell nucleus e.g. humans – n=23 e.g. drosophila – n=2 e.g. dog – n=39 • when not dividing – much of the eukaryotic DNA is in its loosest formation = chromatin – allows access to the machinery for DNA replication and transcription • in preparation for cell division - DNA is replicated and condenses into chromosomes • chromosome = organized structure of DNA and protein – chroma = color – soma = body • the building material of a chromosome is chromatin • each duplicated chromosome is made of two sister chromatids = joined copies of the original chromosome – these chromatids will separate during cell division and be partitioned into each daughter cell • chromatids are joined by a structure called a centromere Sister chromatids Centromere 0.5 m Centromere • condensed region within the chromosome • responsible for the accurate segregation of sister chromatids during mitosis & meiosis • shared by sister chromatids during mitosis • site of the centromere where spindle microtubules attach – area of DNA and protein = kinetochore Sister chromatids Centromere 0.5 m Kinetochore • • • • • a structure of DNA and proteins located in the centromere for the attachment of the chromosome to the spindle during mitosis and meiosis one MT attaches to one kinetochore on one chromatid a 2nd MT attaches to the kinetochore on the other chromatid attachment of MTs to the plates of the kinetochore results in movement toward the poles • a “tug of war” results – chromosomes move back and forth and eventually settle in the metaphase plate Chromatid Outer Plate Microtubules Kinetochore Microtubules Inner Plate Control of DNA Structure: Condensation of Chromatin • nucleosome = DNA helix wrapped around a histone protein core – responsible for organizing the DNA as chromatin • • • • other proteins are involved organizing chromatin as chromosomes non-histone proteins in the nucleus form a chromosome scaffold 300nm fiber specific sequences in the chromosome called scaffold-associated regions or SARs interact with the scaffold to create the 300 nm fiber next level of “packing” is a 700 nm fiber – not much known about this http://www.ndsu.edu/pubweb/~mcclean/plsc431/eukarychrom/eukaryo3.htm chromosome scaffold Chromosome and Chromosomes: Confusion!!! • prior to cell division – the duplicated chromatin condenses into its most dense form = chromosome – two sister chromatids joined by a centromere – typically called a duplicated chromosome • during cell division - the two sister chromatids separate • once separated - the chromatids are still called chromosomes DNA condensation animation - http://www.biostudio.com/demo_fr eeman_dna_coiling.htm • Mitosis is conventionally divided into five phases – – – – – Prophase Prometaphase Metaphase Anaphase Telophase • Cytokinesis overlaps the latter stages of mitosis 10 m G2 of Interphase Centrosomes (with centriole pairs) Nucleolus Chromatin (duplicated) Nuclear envelope Plasma membrane Prophase Early mitotic spindle Aster Centromere Chromosome, consisting of two sister chromatids Prometaphase Fragments of nuclear envelope Kinetochore Metaphase Nonkinetochore microtubules Kinetochore microtubule Anaphase Metaphase plate Spindle Centrosome at one spindle pole Telophase and Cytokinesis Cleavage furrow Daughter chromosomes Nuclear envelope forming Nucleolus forming Mitosis 1. http://www.loci.wisc.edu/outreach/bioclips/CDBio.html Prophase: prior to prophase, the replicated DNA is beginning to condense into sister chromatids joined at the centromere (duplicated) chromosome 1. the centrioles (replicated at G2) move apart from each other 2. the spindle forms between the centrioles (made of microtubules) 3. the kinetochore forms and the condensing duplicated chromosomes attach to the spindle 4. the nucleoli disappear Spindle – structure that includes the two centrioles, two asters and the spindle microtubules than span the cell Aster – a radial array of short MTs extending from the centrioles 2. Prometaphase: used to be known as “late prophase” 1. the nuclear envelope fragments – allows growth of spindle into region where chromosomes are located 2. the DNA of the duplicated chromosomes becomes even more condensed 3. some chromosomes attach to spindle via kinetochore = kinetochore microtubules 4. non-kinetochore microtubules begin to form and grow towards opposite pole Prometaphase Fragments of nuclear envelope Kinetochore Nonkinetochore microtubules Kinetochore microtubule 3. Metaphase: centrioles are at opposite ends of the cell and the spindle is complete 1. the chromosomes move and line up along a central zone= metaphase plate -the tug of war at pro-metaphase eventually positions the chromosomes midway alone the length of the cell 2. non-kinetochore MTs interact with the opposite pole & the aster MTs make contact with the plasma membrane – the spindle is now complete 10 m 3 Metaphase 4. Anaphase: shortest of the mitotic phases 1. the chromatid pairs separate into daughter chromosomes 2. one chromatid/chromosome moves toward one centriole of the cell, the other the opposite -pulled apart by the action of the spindle – the kinetochore MTs begin to shorten -PLUS non-kinetochore MTs grow – this elongates the cell ** At the end of this phase – each end of the cell has equivalent numbers of chromosomes – same number as the parent cell **the sister chromatids separate because of enzymatic activity -an enzyme called separase cleaves a protein known as cohesin (protein in the centromere that holds the sister chromatids together) -separates the sister chromatids 4. Telophase: reverse of Prophase 1. nuclear envelope reforms – two daughter nuclei result -part of the new nuclear membrane is recycled from the old fragments, other parts are made new by the cell 2. the nucleoli reappear 3. the spindle disappears as the MTs depolymerize 4. daughter chromosomes uncoil ** Cytokinesis starts during late anaphase and is well underway during telophase (a) Cleavage of an animal cell (SEM) Cytokinesis: division of cytoplasm -separates the parent into two daughter cells -differs in animal cells and plant cells Animal cell Cytokinesis: results from cleavage pinches into two daughters -actin filaments assemble to form a contractile ring along the equator of the cell -actin interacts with myosin proteins – causes the ring to contract -actin-myosin interaction first forms a “cleavage furrow” - slight indentation around the circumference of the cell -continued interaction divides the cell by a “purse string” mechanism Cleavage furrow Contractile ring of microfilaments 100 m Daughter cells Plant cell Cytokinesis: No cleavage furrow possible -vesicles bud from the Golgi apparatus and migrate to the middle of the cell -vesicles coalesce to produce a cell plate -other vesicles fuse to the plate bringing in new building materials -cell plate grows and eventually splits the cell into two daughter cells Cell plate 10 m (b) Cell plate formation in a plant cell (TEM) Vesicles forming cell plate 5 Telophase Wall of parent cell Cell plate 1 m New cell wall Daughter cells Binary Fission in Bacteria • bacteria and archaea reproduce by binary fission – the chromosome replicates and the two daughter chromosomes actively move apart – the plasma membrane pinches inward, dividing the cell into two – BUT there is no ordered segregation of the duplicated chromosome – duplicate it and divide it into two cells Origin of replication E. coli cell 1 Chromosome replication begins. 2 Replication continues. 3 Replication finishes. 4 Two daughter cells result. Cell wall Plasma membrane Bacterial chromosome Two copies of origin Origin Origin The Evolution of Mitosis • mitosis probably evolved from binary fission • certain protists exhibit types of cell division that seem intermediate between binary fission and mitosis (a) Bacteria Bacterial chromosome Chromosomes (b) Dinoflagellates Microtubules Intact nuclear envelope Kinetochore microtubule (c)Diatoms and some yeasts Intact nuclear envelope Kinetochore microtubule (d) Most eukaryotes Fragments of nuclear envelope Cell Cycle Checkpoints • interphase not only allows the cell to perform its normal functions but also allows the cell to check whether it is ready to enter mitosis • cell cycle is controlled by a control system that coordinates and triggers key events in the cell cycle • progression through the cell cycle requires a combination of internal and external signals – these signals control whether the cell is ready to continue on into the S and M phases • so the control system of the cell cycle monitors these signals and determines whether to proceed through the cell cycle • there are specific points along the cell cycle where “decisions” are made by this control system = CHECKPOINTS G checkpoint • Checkpoint = control point where “stop” and “go-ahead” signals regulate the cell cycle 1 Control system S G1 M G2 M checkpoint G2 checkpoint Cell Cycle Checkpoints • major checkpoints – G1, G2 and M – G1 checkpoint – G1/S progression through a point called the restriction point or START point of the cell cycle – G2 checkpoint – G2/M progression which will lead to the start of mitosis and chromosome alignment – M checkpoint – Metaphase to Anaphase transition where chromatid separation occurs G1 checkpoint Control system G1 M G2 M checkpoint G2 checkpoint S Cell Cycle Checkpoints • for many cells, the G1/S checkpoint seems to be the most important – if a cell receives a go-ahead signal at this G1/S checkpoint will usually complete the S, G2, and M phases and divide – many texts call this checkpoint the Start (yeasts) or Restriction point (mammalian cells) • if the cell does not receive the go-ahead signal - will exit the cycle, switching into a non-dividing state called the G0 phase • most cells in the body are in the G0 phase and remain there • some cells have the ability to leave G0 and re-enter the cell cycle G0 G1 checkpoint G1 (a) Cell receives a go-ahead signal. G1 (b) Cell does not receive a go-ahead signal. IN ANIMAL CELLS A “GO-AHEAD” SIGNAL MUST BE PRODUCED TO OVERRIDE A BUILT IN “STOP” SIGNAL Cell Cycle Control System • the proteins of this system evolved over a billion years ago • so well conserved in eukaryotes – take from human control cells and put into yeast cells – they work!! • much of the early research done – been done in yeast – search for mutations in genes that encode critical parts of the cell cycle control system = cell-division-cycle genes or cdc genes – many mutations cause the cell cycle to arrest a specific points – such as checkpoints • additional work done in frog eggs and in mammalian cell cultures – e.g. immortalized mammalian cell lines The Cell Cycle Control System: Cyclins and CyclinDependent Kinases • two types of regulatory proteins are involved in cell cycle control: cyclins and cyclin-dependent kinases (Cdks) – cyclins named for the cyclical changes in their concentration through the cell cycle – cdks named because their phosphorylation activities requiring their binding to their “partner” cyclin – cdks cannot work as kinases unless they are bound to their partner cyclin The Cell Cycle Control System: Cyclins and CyclinDependent Kinases • cdk activity fluctuates during the cell cycle – cdk proteins are expressed first – activity is controlled by an array of proteins - including the cyclins – cyclical changes in cdk activities leads to cyclical changes in the phosphorylation of their target proteins – cdks phosphorylate key proteins responsible for passing through each checkpoint The Cell Cycle Control System: Cyclins and CyclinDependent Kinases • eukaryotic cells have four classes of cyclins – each act at a specific stage of the cell cycle – eukaryotic cells require three of these classes for their cell cycle • • • • 1. G1/S cyclins 2. S-cyclins 3. M-cyclins 4. G1 cyclins The Cell Cycle Control System: Cyclins and CyclinDependent Kinases • eukaryotic cells have four classes of cyclins – each act at a specific stage of the cell cycle – eukaryotic cells require three of these classes for their cell cycle • 1. G1/S cyclins – active in late G1 – trigger the progression through the G1 restriction point – cyclin E; cdk2 • 2. S-cyclins – activate cdks that help stimulation chromosome duplication – levels stay high until M phase - since they help initiate mitosis – cyclin A; cdk2 • 3. M-cyclins – activate the cdks that stimulate progression through the G2 checkpoint and into Mitosis – cyclin B; cdk1 Cyclins and Cyclin-Dependent Kinases – no you don’t have to know these for your exam!!! FUNCTION G1/entry into cell cycle Entry into cell cycle & S phase Mitosis modified from Cell Biology Alberts et al. • • • work in frog eggs identified a cdk-cyclin complex that triggered the cell’s passage past the G2 checkpoint into the M phase called MPF (maturation-promoting factor) • also called Mitosis-promoting factor – actually - cyclin B and cdk1 cyclin B combines with cdk1 in G2 to produce MPF – – • • • cyclin B levels peak at M phase MPF activity peaks at M phase when enough MPF is made – the cell passes the G2 checkpoint and enters Mitosis during anaphase cyclin B begins to become degraded and cdk1 activity starts to fall M phase stops and G1 begins G G G M G1 S 2 M 1 S 2 MPF activity Cyclin concentration Time (a) Fluctuation of MPF activity and cyclin concentration during the cell cycle M G1 Regulation of CDK activity • • want to control the CDK? control the expression of the cyclin!!! e.g. during anaphase cyclin B becomes degraded and CDK1 activity starts to fall – – • the cdk1 part of the MPF actually degrades its partner cyclin the cdk1 is “recycled” for future cycles M phase stops and G1 begins Cdk Degraded cyclin G2 checkpoint Cdk Cyclin is degraded MPF Cyclin (b) Molecular mechanisms that help regulate the cell cycle Regulation of CDK activity • • • But the CDK is also another level of control the CDK can promote its own activation through positive feedback the CDK can also promote their own inactivation through negative feedback – • • • e.g. degradation of its partner cyclin CDK activation/inactivation is ultimately controlled by additional kinases phosphorylating the CDK some phosphorylation activates it others inhibit it • • SO: activation of specific cyclin-cdk complexes drive progression through the G1 and G2 checkpoints BUT: progression through the M checkpoint requires the degradation of proteins cessation of mitosis – e.g. degradation of cyclin B • • one key regulator of cyclin degradation is the anaphase-promoting complex or APC mitotic cdk/cyclins activate the APC (via phosphorylation) • the APC directs the degradation of anaphase inhibitors in the cell • allows the onset of anaphase • as mitosis proceeds – the APC directs the degradation of the cdk/cyclin complex • the drop in mitotic cdk/cyclin complexes now allows for the decondensing of chromosomes etc….. cyclin/cdk promotes degradation activates APC degradation of inhibitors anaphase Stop and Go Signs: Internal and External Signals at the Checkpoints • there is a link between what is happening inside and outside the cell with the activity of cdk/cyclins • in other words – internal and external signals exert control over cdk/cyclins and the cell cycle • internal signal – e.g. kinetochores not attached to spindle microtubules send a molecular signal that delays anaphase – all chromosomes must be attached to the spindle in order to eventually activate and enzyme called separase – separase breaks down the cohesin proteins within the centromere – the chromatids separate during anaphase Stop and Go Signs: Internal and External Signals at the Checkpoints • some external signals are growth factors – proteins released by certain cells that stimulate other cells to divide = mitogen – more than 50 growth factors identified in eukaryotic cells – some GFs are made by several types of cells – others are quite cell-specific • another external signal - density-dependent inhibition – crowded cells stop dividing • another signal - anchorage dependence – cells must be attached to a substratum in order to divide Scalpels 1 A sample of human connective tissue is cut up into small pieces. External Cell-Cycle Signals Petri dish 2 Enzymes digest the extracellular matrix, resulting in a suspension of free fibroblasts. 3 Cells are transferred to culture vessels. Without PDGF • 4 PDGF is added to half the vessels. 10 m With PDGF platelet-derived growth factor (PDGF) stimulates the division of human fibroblast cells in culture – bind to PDGF receptors on target cells – initiate the progression through the G1 phase into M • GFs like PDGF often initiate a series of internal signaling events that activate a class of kinases called G1 kinases -G1 kinases then exert control over the G1/S cyclins that initiate entry into the cell cycle Loss of Cell Cycle Controls in Cancer Cells • cancer cells exhibit neither density-dependent inhibition nor anchorage dependence • Cancer = known medically as a malignant neoplasm • cells with upregulated cell growth because they do not have a normally controlled cell cycle • uncontrolled division gives rise to cells that can invade surrounding tissues and travel through the lymphatic and circulatory systems to invade other tissues at a distance = malignant cancer • if abnormal cells remain only at the original site - the lump is called a benign tumor Tumor Lymph vessel Blood vessel Glandular tissue Cancer cell 1 A tumor grows from a single cancer cell. Metastatic tumor 2 Cancer cells invade neighboring tissue. 3 Cancer cells spread through lymph and blood vessels to other parts of the body. 4 Cancer cells may survive and establish a new tumor in another part of the body. Causes of Cancer • over 200 cancers that affect humans – Thyroid cancer – 94% five year survival rate – Skin cancer – 81% Mutagen – agent that causes a – Prostate cancer – 74% mutation in the DNA – Breast cancer – 76% Carcinogen – mutagen that causes – Stomach cancer – 17% cancer – Pancreatic – 1% • numerous and diverse causes – 90-95% cases are attributed to environmental causes • Environmental causes = not due to genetic, inherited factors – Tobacco – 25-30% of all cancers – Diet & obesity – 30-35% • negative effects on the immune system and endocrine system • e.g. high salt diet – tied to increased gastric cancer – Infective agents (virus, bacteria) – 15-20% – Radiation – up to 10% Mutagens • mutagen = agent that causes a mutation in the DNA • mutations can arise in a number of ways – 1. errors in replication – 2. errors in genetic recombination (during meiosis) • mutagens can be both physical and chemical agents – physical – X-rays, UV light other forms of high energy radiation – chemical – nucleotide analogs (mimic a NT but incorrectly pair), intercalating agents, others alter chemical properties of the base (alters their pairing capacity) • types of mutations: – small scale – point mutations – large scale – effect large areas of chromosome sequence Mutations in Cancer • genetic mutations can result in the transformation of the genes of cell growth and differentiation – required for the development of cancer • numerous kinds of genetic mutations possible – more than one is usually required – small scale point mutations, deletions and insertions large scale deletion or gain of chromosomal sections, translocation of chromosome sections • transformation of a cancer cell does not always result in increased proliferation – can result in disabled metabolism – loss of cell-cell adherence + increased ability to invade surrounding tissues – metastatic capacity – secretion of angiogenic growth factors – increased vascularization of the tumor Mutations Wild type DNA template strand 3 T A C T T C A A A C C G A T T 5 5 A T G A A G T • small-scale - point mutations: • A. nucleotide pair substitutions – replacement of one NT and its partner for another pair • if there is no change to the eventual codon/amino acid = silent mutation • if it changes the amino acid to a stop codon = nonsense mutation • if it changes the amino acid = missense mutation – still may be no change to the overall structure and function of the protein T T G G C T A A 3 mRNA5 A U G A A G U U U G G C U A A 3 Protein Met Lys Phe Gly Stop Carboxyl end Amino end (a) Nucleotide-pair substitution A instead of G 3 T A C T T C A A A C C A A T T 5 5 A T G A A G T T T G G T T A A 3 U instead of C 5 A U G A A G U U U G G U U A A 3 Met Lys Phe Gly Stop Silent (no effect on amino acid sequence) T instead of C 3 T A C T T C A A A T C G A T T 5 5 A T G A A G T T T A G C T A A 3 A instead of G 5 A U G A A G U U U A G C U A A 3 Met Lys Phe Ser Stop Missense A instead of T 3 T A C A T C A A A C C G A T T 5 5 A T G T A G T T T G G C T A A 3 U instead of A 5 A U G U A G U U U G G C U A A 3 Met Nonsense Stop Mutations (b) Nucleotide-pair insertion or deletion • B. insertions • C. deletions • insertions and deletions can lead to a frame-shift • alters how the codons are read downstream from the mutation Extra A 3 T A C A T T C A A A C C G A T T 5 5 A T G T A A G T T T G G C T A A 3 Extra U 5 A U G U A A G U U U G G C U A A 3 Met Stop Frameshift causing immediate nonsense (1 nucleotide-pair insertion) A missing 3 T A C T T C A A C C G A T T 5T 5 A T G A A G T T G G C T A A 3A U missing 5 A U G A A G U U G G C U A A Met Lys Leu Ala Frameshift causing extensive missense (1 nucleotide-pair deletion) T T C missing 3 T A C A A A C C G A T T 5 5 A T G T T T G G C T A A 3 A A G missing A A 5 A U G U U U G G C U A A 3U Met Phe Gly Stop No frameshift, but one amino acid missing (3 nucleotide-pair deletion) 3 Genes of Cancer • two broad category of genes affected with these mutations: tumor-suppressor genes and proto-oncogenes Genes of Cancer: Oncogenes • • • proto-oncogenes are normal cellular genes responsible for normal cell growth and division – mutation in a proto-oncogene transforms them into an oncogene (cancer causing) conversion of a proto-oncogene to an oncogene can lead to abnormal stimulation of the cell cycle proto-oncogenes can be converted to oncogenes by – translocation of DNA within the genome: if it ends up near an active promoter, transcription may increase – amplification of a proto-oncogene: increases the number of copies of the gene via gene duplication – point mutations in the proto-oncogene or its control elements: cause an increase in gene expression Proto-oncogene DNA Translocation or transposition: gene moved to new locus, under new controls Gene amplification: multiple copies of the gene New promoter Normal growthstimulating protein in excess Point mutation: within a control within element the gene Oncogene Normal growth-stimulating protein in excess Normal growthstimulating protein in excess Oncogene Hyperactive or degradationresistant protein Genes of Cancer: Tumor-Suppressor Genes • tumor-suppressor genes help prevent uncontrolled cell growth – mutations that decrease protein products of tumor-suppressor genes may contribute to cancer onset • tumor-suppressor proteins – repair damaged DNA – control cell adhesion – inhibit the cell cycle using cell-signaling pathways • most studied tumor suppressor gene/protein = p53 – functions as an inhibitor to the cell cycle when DNA damage is sensed by repair mechanisms – ensures that the DNA is repaired before proceeding to mitosis Oncogenes and the Cell Cycle • mutation in a proto-oncogene transforms them into an oncogene • these oncogenes have abnormal cell cycle control checkpoints • e.g. Cyclin D – in normal cells – cyclin D is a link between G1 progression and growth factor production – overproduction of cyclin D/cdk4 in cancer cells drives the cell into and through this phase • acts to shorten the G1 phase – even takes cells out of the G0 phase sooner then they should be – overproduction can result from: • 1. chromosome translocation - next to a very active promoter • 2. gene amplification Cancer Therapies • treatments of tumors – – – – – 1. chemotherapy – targets rapidly proliferating cells 2. radiation 3. immune therapies – antibody therapy e.g. Herceptin, Avastin 4. hormone therapies - Tamoxifen 5. STIs – signal transduction inhibitors • other promising approaches such a nanotechnology – coupling of drugs to nanoparticles capable of entering a cancer cell quite easily – internalization releases drug once inside cell – increase targeting of nanoparticle by coupling particle/drug to an antibody specific to a cancer cell Chemotherapy • chemotherapies are general cytotoxic chemicals that target rapidly dividing cell types – can also kill healthy cells with “fast” cell cycles • e.g. gastric epithelium • generally work by impairing mitosis • numerous categories all with various mechanisms of action – ranging from damaging DNA to inhibiting the “machinery” of mitosis Chemotherapy • numerous categories: – 1. Alkylating agents – original chemotherapies • derived from mustard gas – most common = cisplatin, carboplatin – 2. Anti-metabolites – impede DNA and RNA synthesis • most common = methotrexate – 3. Anti-microtubule agents – plant-derived chemicals that prevent microtubule function • Taxanes (e.g. Taxol) – from the pacific Yew tree – 4. Topoisomerase inhibitors – from the Chinese ornamental tree Camptotheca acuminata – 5. Cytotoxic antibiotics – various mechanisms of action • most common = doxirubicin; derived from a bacteria – 6. Hormone therapy – inhibition of hormone-receptor interaction • most common = Tamoxifen