* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Kinetic resolution wikipedia , lookup
Marcus theory wikipedia , lookup
Metal carbonyl wikipedia , lookup
Bottromycin wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Elias James Corey wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Ene reaction wikipedia , lookup
Stille reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Discodermolide wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aldol reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
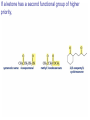
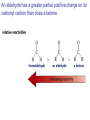
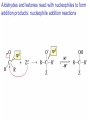
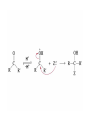
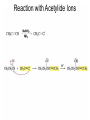
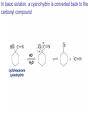
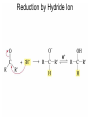
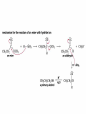
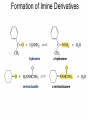
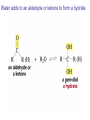
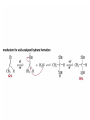
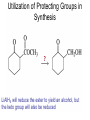
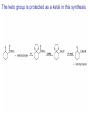
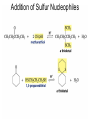
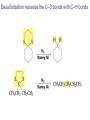
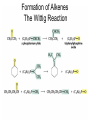
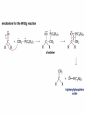
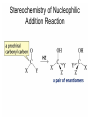
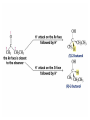
Organic Chemistry 4th Edition Paula Yurkanis Bruice Chapter 18 Carbonyl Compounds II Radicals Irene Lee Case Western Reserve University Cleveland, OH ©2004, Prentice Hall Nomenclature of Aldehydes If the aldehyde group is attached to a ring, If a compound has two functional groups, the one with the lowest priority is indicated by its prefix Nomenclature of Ketones If a ketone has a second functional group of higher priority, An aldehyde has a greater partial positive charge on its carbonyl carbon than does a ketone • Steric factors contribute to the reactivity of an aldehyde • The carbonyl carbon of an aldehyde is more accessible to the nucleophile • Ketones have greater steric crowding in their transition states, so they have less stable transition states Aldehydes and ketones react with nucleophiles to form addition products: nucleophile addition reactions If the nucleophile that adds to the aldehyde or ketone is an O or an N, a nucleophilic addition–elimination reaction will occur Formation of a New Carbon–Carbon Bond Using Grignard Reagents Grignard reagents react with aldehydes, ketones, and carboxylic acid derivatives Reaction with Acetylide Ions In basic solution, a cyanohydrin is converted back to the carbonyl compound Synthesis Using Cyanohydrin Reduction by Hydride Ion Utilization of DIBAL to Control the Reduction Reaction The reduction of a carboxylic acid with LiAlH4 forms a single primary alcohol Acyl chloride is also reduced by LiAlH4 to yield an alcohol An amide is reduced by LiAlH4 to an amine Aldehydes and ketones react with a primary amine to form an imine This is a nucleophilic addition–elimination reaction The pH of the reaction must be controlled Dependence of the rate of the reaction of acetone with hydroxylamine on the pH of the reaction: a pH rate profile Aldehydes and ketones react with secondary amines to form enamines Formation of Imine Derivatives Deoxygenation of the Carbonyl Group Water adds to an aldehyde or ketone to form a hydrate Why is there such a difference in the Keq values? The equilibrium constant for the reaction depends on the relative stabilities of the reactants and products Addition of an Alcohol to an Aldehyde or a Ketone Utilization of Protecting Groups in Synthesis LiAlH4 will reduce the ester to yield an alcohol, but the keto group will also be reduced The keto group is protected as a ketal in this synthesis The more reactive aldehyde is protected with the diol before reaction with the Grignard reagent • The OH group in an alcohol can be protected as a trimethylsilyl ether • The OH group in a carboxylic acid can be protected as an ester • An amino group can be protected with an acetyl group Addition of Sulfur Nucleophiles Desulfurization replaces the C–S bonds with C–H bonds Formation of Alkenes The Wittig Reaction Preparation of the Phosphonium Ylide If two sets of reagents are available for the synthesis of an alkene, it is better to use the one that requires the less sterically hindered alkyl halides • The Wittig reaction is completely regioselective • This reaction is the best way to make a terminal alkene • Stable ylides form primarily E isomers, and unstabilized ylides form primarily Z isomers • Stable ylides have a group (C=O) that can share the carbanion’s negative charge Stereochemistry of Nucleophilic Addition Reaction • Nucleophiles that form unstable addition products form conjugated addition products, because the conjugate addition is not reversible • Nucleophiles that form stable addition products can form direct addition products or conjugate addition products • If the rate of direct addition is slowed down by steric hindrance, a Grignard reagent will form the conjugate addition product Nucleophilic Addition to a,b-Unsaturated Carboxylic Acid Derivatives Enzyme-Catalyzed Additions to a,bUnsaturated Carbonyl Compounds