* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download NUCLEOPHILIC SUBSTITUTION & ELIMINATION ON Csp 3
Kinetic resolution wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Elias James Corey wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Discodermolide wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Marcus theory wikipedia , lookup
Hydroformylation wikipedia , lookup
Stille reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Ene reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
George S. Hammond wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Petasis reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
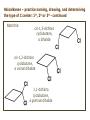
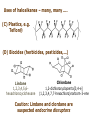
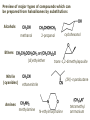
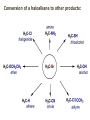
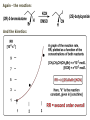
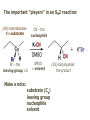
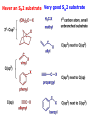
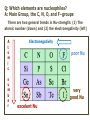
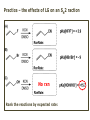
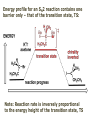
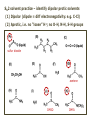
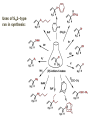
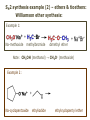
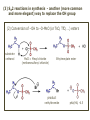
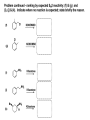
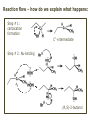
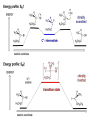
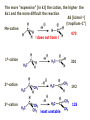
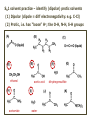
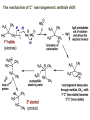
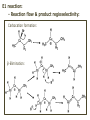
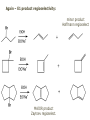
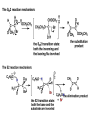
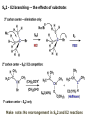
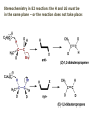
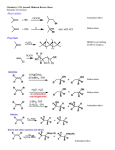
ORGANIC CHEMISTRY 1 Chapter 6, Part 1 (1) Haloalkanes, preparation & properties (2) Nucleophilic Substitution Reactions -or: - How to make alcohols, ethers, esters, amines, nitriles, … (3) Elimination Reactions – or: -How to make alkenes from haloalkanes and alcohols Based on Organic Chemistry, by L.G. Wade, 7th ed; Compiled by: Dr. Peter Ilich, St. John’s University Queens, New York, Spring 2012 Halogenation of alkanes – How does it happen: Halogenation of alkanes – continued – up to CCl4: In branched alkanes regioselectivity becomes important: 2º C: major product 3º C: the only product Haloalkanes – review of common alkyl groups: 4 butanealkanes: 2 propanealkanes: 1-chloropropane (propylchloride) 2-bromopropane (isopropylchlride) 1-bromobutane (butylchloride) 2-chlorobutane (sec-butylchloride) 1-bromo-2-methyl propane (isobutylbromide) 2-choro-2-methyl propane (tert-butylchloride) Haloalkane – practice naming, drawing, and determining the type of C center: 1º, 2º or 3º formula: name: iodomethane (methyl iodide) 2-chloropentane (isopentyl chloride) 1,2-dichlorocylobutane Haloalkanes – practice naming, drawing, and determining the type of C center: 1º, 2º or 3º - continued Note this: cis-1,3-dichloro cyclobutane, a dihalide cis-1,2-dichloro cyclobutane, a vicinal dihalide 1,1-dichloro cyclobutane, a geminal dihalide [6.2-5] Properties and uses of haloalkanes: (A) Polar C- X bond: (B) Immiscible with but heavier than water: water alkane water haloalkane Uses of haloalkanes – many, many …. (C) Plastics, e.g. Teflon® (D) Biocides (herbicides, pesticides,…) Lindane 1,2,3,4,5,6hexachlorocyclohexane Chlordane 1,2-dichlorocyclopenta[3,4-e] (1,2,3,4,7,7-hexachloro)norborn-3-ene Caution: Lindane and clordane are suspected endocrine disruptors Not all haloalkanes and C-halogen compounds are manmade; there are thousands of natural C-halogenides, e.g. (3S)-bromomethyl-(6R)-bromo2,(3S),7-trichloro-1-nonene, a human anticancer agent secreted by marine the alga Portieria hornemanii, [Gribble GW, Acc. Chem. Res. 1998, 31, 141-152]. Thyroxine, T4, a hormone secreted by the human thyroid gland is a natural tetraiodo organic compound Q: Now that you have a supply of haloalkanes - made by radical halogenation – what other compounds can you make out of them? A: Many other kinds of compounds – for example: - alcohols thioalcohols (mercaptanes) ethers, linear and cyclic thioethers (sulfides), linear and cyclic peroxides esters amines (1º-, 2º-, 3º- and 4º- amines) cyanides (nitriles) azides alkanes alkynes Preview of major types of compounds which can be prepared from haloalkanes by substitution: Alcohols methanol 2-propanol cyclohexanol Ethers (di)ethylether Nitrile (cyanides) Amines trans-1,2-dimethylepoxide (2R)-cyanobutane ethanenitrile methylamine N-ethylmorpholine tetramethyl ammonium Conversion of a haloalkane to other products: Example of a reaction of conversion of haloalkane - conversion of bromobutane to butylcyanide: The reaction: (2R)-bromobutane (2S)-butylcyanide Reaction type: Substitution, Nucleophilic, 2nd order – SN2 Again - the reaction: And the kinetics: The important “players” in an SN2 reaction: (2R)-bromobutane: the substrate – the nucleophile Br – the leaving group, LG DMSO = solvent Make a note: -CN substrate (Cα) leaving group nucleophile solvent (2S)-butylcyanide: the product Effects of substrate on an SN2 reaction: Rxn rate: Structure: fastest 1º Cα, short fast 1º Cα, longer slower 1º Cα, but Cβ branched slow NO rxn 2º Cα 3º Ca Never an SN2 substrate Very good SN2 substrate The effects of substrate on SN2 rate – practice: Rank the following triads of SN2 rxn by expected rate; fastest=1st The effects of substrate on SN2 rate – practice: Rank the following SN2 rxn by expected rate; fastest=1st Identify and tag Cα in each substrate and classify it as 1º, 2º; then look at Cβ The next player the nucleophile: very fast Observe these reactions: What is the fast different? What is slow the same? very slow SN2 - the nucleophile effect - again: Observe: What is common to all these compounds? Make a note: A nucleophile is an electron-rich atom or group of atoms - a Lewis base Very good Nu Good Nu Fair Nu Poor Nu Q: Which elements are nucleophiles? A: Main Group, the C, N, O, and F–groups There are two general trends in Nu-strength: (1) The atomic number (down) and (2) the electronegativity (left) A t o m i c n u m b e r Electronegativity poor Nu very good Nu excelent Nu Nu-strength: electronegativity & atomic number Nucleophile as a Brønsted base It was said that a nucleophile is a Lewis base (el-rich, capable of donating an electron pair) As a rule, nucleophiles are Brønsted bases (Brønsted base – capable of accepting H+) but the relation is not simple: Within the same group elements become weaker Brønsted bases but better nucleophiles: SN2 – player # 3 - the leaving group, LG: Observe these two SN2 reactions: What do you see? The stronger the conjugate acid the better the leaving group; this holds without exception Practice – the effects of LG on an SN2 raction No rxn Rank the reactions by expected rate: Leaving Group and the pKa of conjugate acid Table: best LG good LG fair LG never a LG SN2 – putting it all (almost) together: - The substrate: 1o C (or, not so good, 2º C) - The nucleophile: good - The leaving group: low pKa of conjugate acid - The solvent: polar, aprotic (next slides) The reaction flow & the transition state: + KBr SN2 – a concerted reaction How does this reaction occur? – practice: (1) Identify Cα, (2) identify Nu and (3) add el-pairs and partial charges as needed, (4) write down the mechanistic arrows, (5) draw the TS and (6) label it, and (7) complete the reaction: #2 #1 Energy profile for an SN2 reaction contains one barrier only – that of the transition state, TS: Note: Reaction rate is inversely proportional to the energy height of the transition state, TS The player # 4 in SN2 reactions – the solvent Observe these exp data: We explain this effect by different solvation of anions and cations in dipolar aprotic solvents, as in this scheme of solvation of KCN in dimethylsulfoxide, DMSO: unsolvated (bare) and highly active nucleophile SN2 solvent practice – identify dipolar protic solvents (1) Dipolar (dipole = diff electronegativity: e.g. C-Cl) (2) Aprotic, i.e. no “loose” H+; no O-H, N-H, S-H groups sulfur dioxide acetone DMSO DMFA Review of SN2 reactivity – three possible cases: Summary of the Ch. 6, part 1 – What have we learned today? - Haloalkanes can be converted to alcohols, thioalcohols, ethers, thioethers, amines, nitriles, azides, peroxides, . . . and a number of other type compounds - The predominant reaction types in these conversions are second order nulcleophilic substitutions, SN2 - SN2 reaction can be shown to depend on substrate, leaving group, nucleophile and solvent - Optimal reaction parameters and conditions are established and the SN2 mechanism is derived ORGANIC CHEMISTRY 1 Chapter 6, Part 2 (1) Uses of SN2 reactivity in synthesis (2) Other SN reactions: SN1 - SN1 variables: substrate, nucleophile, leaving group, solvent - The carbocation intermediate paths (3) Elimination, 1st order & competition with SN1 (4) Eliminatino, 2nd order Based on Organic Chemistry, by L.G. Wade, 7th ed; Compiled by: Dr. Peter Ilich, St. John’s University Queens, New York, Spring 2012 Summary of SN2 type reactions – three possible cases: Part 2: SN2 reactions – what are they good for? - Example (1) - Preparation of alcohols: iodomethane methanol and thioalcohols: bromoethane ethanethiol (mercaptoethanol) Uses of SN2–type rxn in synthesis: SN2 synthesis example (2) – ethers & tioethers: Williamson ether synthesis: Example 1: Na-methoxide methylbromide dimethyl ether Note: CH3OH (methanol) → CH3O- (methoxide) Example 2: Na-cyclopentoxide ethyliodide ethylcyclopentyl ether Uses of SN2 in drug design an example: Ciguatera – a GI and a neuropathological condition caused by a natural toxin, ciguatoxin, found in certain tropical fish Treating ciguatera required using toxin to study its mode of action; as toxin is difficult to isolate it had to be prepared de novo – using SN2 ether cyclization But note: SN2 in synthesis – practice Williamson synthesis: Another example – nucleophilic methylation in biology: - Nucleic acids (C, A) N-methylation, for transcription regulation - Neurotransmitters, NH2-CH2CH2-OPO3 N-methylation (S)-Adenosylmethionine , AdoMet, SAM, = the Nature's methylation agent - Fatty (oleic) acid C-methylatinon The rxn: substrate = methyl-sulfonium, leaving group = sulfide SN2 synthesis – another matter of concern the pKa of the LG and the reaction direction: pKa(CH3COOH/CH3COO-) = +4.7 pKa(HCl/Cl-) = -7 Note that HCl is ~ 1010 or ten billion times stronger acid and Cl- is a much better leaving group; the reaction (a) will happen but the reaction (b) will not. Q: How do we “make” SN2 work in the case (b)? SN2 in synthesis (3) - replacement of OH group: (1) Acidification of –OH to –OH2+ (hydronium group) Note: pKa(HOH/HO-) +15.7 and pKa(H3O+/H2O) -1.7 Acidification of –OH to –OH2+ - the mechanism #1 protonation of OH #2 nucleophilic attack by Br- SN2 in synthesis – the removal of OH group; practice Try to complete the following 2 reactions; - observe the differences: Suggest a way out (or around): (3) SN2 reactions in synthesis - another (more common and more elegant) way to replace the OH group (2) Conversion of –OH to –O-MsO (or TsO, TfO, …) esters substrate =ethanol MsCl = Mesyl chloride (methanesulfonyl chloride) Ethylmesylate ester product =ethylbromide pKa(HA) -6.5 [6.13] A new page – and a new chemistry: The same substrate but a different reaction Substitution, nucleophilic - but a different one: The reaction: (2R)-bromobutane (2S)-butanol (2R)-butanol (Optically inactive racemic mixture) [6.13] The reaction rate - experimental data: The reaction: (CH3)3C-Br + HOH → (CH3)3C-OH + K+Br- The concentration vs. time - exp data: The rate of the reaction changes with the conc. of the substrate, (CH3)3CBr, but is independent on the concentration of water, the nucleophile: RR ∝ [HOH]º RR = k [(CH3)3CBr]1 [HOH]0 = 1st order substitution = SN1 Reaction flow – how do we explain what happens: Step # 1: carbocation formation C+-intermediate Step # 2: Nu-binding (R,S)-2-butanol Note 2 things: (i) The reaction occurs in steps; it is consecutive (ii) In the 1st step a carbocation intermediate forms Clearly, Ea1 > Ea2, and the 1st step, formation of carbocation intermediate, is the rate-determining step The more “expensive” [in kJ] the cation, the higher the Ea1 and the more difficult the reaction ΔE [kJmol-1] (tropilium-C+) Me-cation 473 ! does not form ! 1º-cation 301 2º-cation 192 3º-cation 125 least unstable SN1 – the substrate effects; practice: SN1 & SN2 – the leaving group properties & ranking: best LG good LG fair LG never a LG Leaving group competition – practice: Assign pKa values: pKa=+3.9, pKa=0.0, pKa=-13 Fastest: pKa = Medium fast: pKa = Slow: pKa = Review of the SN1 reaction determinants: - The substrate – Csp3 crowded, a good C+ - The nucleophile - It does not matter - The Leaving Group – same as in SN2 (pKa!) - The solvent in SN1 reactions – Protic solvents SN1 solvent practice – identify (dipolar) protic solvents (1) Dipolar (dipole = diff electronegativity: e.g. C-Cl) (2) Protic, i.e. has “loose” H+; the O-H, N-H, S-H groups ethanol acetamide acetic acid water dihydrogensulfide Experimental kinetic data for solvolysis of tert-butyl chloride: Note that in EtOH/HOH mixtures the HO- is the nucleophile Explanation of the SN1 reaction mechanism (“strong ion-pair”) through interactions with a protic solvent SN1 alert – Carbocation Rearrangement: Frank C. Whitmore (UPenn, 1887-1947): ... carbocation rearrangements result when ... "an atom in an electron-hungry condition seeks its missing electron pair from the next atom in the molecule". A reaction: solvolysis of neopentyl iodide. The mechanism of C+ rearrangement: methide shift Carbocation intermediate → rearrangement practice the substrate the product Summary of differences: SN2 vs. SN1 SN2 SN1 Substrate: 1 Cº, uncrowded 3º, C crowded Nucleophile: good: I-, Br-, CN-, R3N, N3- irrelevant good, low pKa of conjug acid good, low pKa of conjug acid Solvent: polar aprotic; DMSO, acetone OH, SH, NH type solvent Reaction flow: concerted, transition state stepwise, C+ intermediate Leaving group: SN2 vs. SN1 “game” – practice field: More on SN1 – consider this: SN1 E1 Make a note: Every SN1 is accompanied by an E1 reaction. SN1 vs. E1: E1 reaction: - Reaction flow & product regioselectivity: Carbocation formation: β-Elimination: Again – E1 product regioselectivity: minor product Hoffmann regioselect MAJOR product Zaytsev regioselect. What a carbocation can do? (4 things) (1) Go forward & form a racemic mixture of products (2) Go backward & form a racemic mixture of the reactant (3) Undergo β-elimination and from an alkene (4) Rearrange and do (1), (2), (3) E1 + carbocation rearrangement – practice: Other types of elimination reactions: E2 When a nucleophile Nu: replaces the leaving group on Csp3 in a concerted (smooth, continuous) way this is an SN2 reaction. When the same nucleophile is a strong Broensted base, it can lead to a concerted elimination, or the so-called E2 reaction: SN2 - E2 branching -- the effects of substrate: 1º carbon center – SN2 only Make note: No rearrangement in SN2 and E2 reactions Stereochemistry in E2 reaction: the H and LG must be in the same plane – or the reaction does not take place: More on E2 stereochmistry: Summary Ch. 6 – What have we learned today?