* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cell Membrane Structure and Transport
Survey
Document related concepts
Lipid bilayer wikipedia , lookup
Cell nucleus wikipedia , lookup
Cytoplasmic streaming wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cell culture wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cell growth wikipedia , lookup
Signal transduction wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cytokinesis wikipedia , lookup
Cell membrane wikipedia , lookup
Transcript
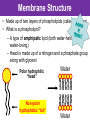
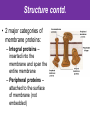
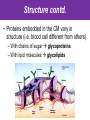
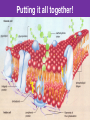
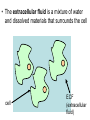
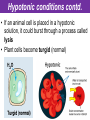
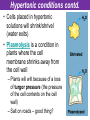
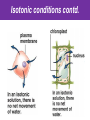
The Living Cell Membrane The Living Cell Membrane • Flexible envelope that holds cytoplasm and organelles in place • Regulates what enters & leaves the cell • Only found in living cells Membrane Structure • Made up of two layers of phospholipids (called a bilayer) • What is a phospholipid? – A type of amphipatic lipid (both water-hating and water-loving) – Head is made up of a nitrogen and a phosphate group along with glycerol Polar hydrophilic “head” Water Non-polar hydrophobic “tail” Water Membrane Structure contd. • Mosaic of various components scattered throughout (i.e. proteins, cholesterol, and carbohydrates) Hydrophilic Hydrophobic Hydrophilic Structure contd. • 2 major categories of membrane proteins: – Integral proteins – inserted into the membrane and span the entire membrane – Peripheral proteins – attached to the surface of membrane (not embedded) Structure contd. • Proteins embedded in the CM vary in structure (i.e. blood cell different from others) – With chains of sugar glycoproteins – With lipid molecules glycolipids Structure contd. • “The Fluid-mosaic membrane model” describes the structure of the CM – Phospholipids can drift/move sideways or rotate in place – Vertical flip-flop of phospholipids is unfavourable (rarely occurs) – Watch this! What gives fluidity? • Saturated vs. Unsaturated fatty acid ‘tails’ Adding Fluidity • Why cholesterol? • The presence of cholesterol molecules decreases permeability and allows the CM to function in a wide range of temperatures – At high temperatures, it maintains its rigidity – At low temperatures, it keeps the CM fluid Structure relates to Function! • What are the functions of the cell membrane? – Provide a barrier – hydrophobic environment prevents most substances from entering the cell – Gatekeeping – opens and closes paths through the cell membrane (i.e. nerve cell K goes in at rest, Na goes in when excited) – Receptor sites – provides a binding site for chemical messengers (like hormones) to induce cell activity (i.e. insulin hormone binds to receptor to open pores to allow sugar in) Structure relates to Function! • What are the functions of the cell membrane? – Transport – transport proteins aid the movement of particles across the membrane • also controls what enters/exits – Structural Support – proteins are attached as microfilaments to maintain cell shape – Catalyze biological reactions – proteins present on the surface of the membrane assist in chemical reactions – Check it out! Putting it all together! • Label your diagram of the cell membrane Movement Across the Cell Membrane How do particles go in and out of the cell if the membrane is selective? • Chemicals in our body must be kept in balance in order for us to survive • Maintenance of constant conditions is called homeostasis • To achieve homeostasis, materials must be exchanged from the inside of the cell to the extracellular environment • There are two types of transport mechanisms across the cell membrane: – Passive transport – Active transport • The extracellular fluid is a mixture of water and dissolved materials that surrounds the cell cell ECF (extracellular fluid) Passive Transport • Movement of materials across the cell membrane without the expenditure of cell energy • 3 types of passive transport: – Diffusion – Osmosis – Facilitated Diffusion • Many small and uncharged molecules can move easily through the membrane (i.e. oxygen and water) • Brownian motion - molecules are constantly moving randomly and colliding with each other – This is the force behind diffusion Diffusion • The movement of particles (solute) from a higher concentration to a lower concentration until they are spread out evenly • Concentration = # particles/unit volume • Movement is “down” the concentration gradient since molecules are in constant and random motion (Brownian motion) • Will occur until equilibrium is met (equal conc.’s on both sides) • Can you think of any examples? – Someone sprays cologne in one end of the hallway but eventually can be smelled at the other end Diffusion contd. • Examples: – Oxygen diffuses from blood (high conc.) to the cell (low conc.) – Carbon dioxide diffuses from the cell (high conc.) into the blood (low conc.) What would happen if I added food colouring to a beaker of water? Why are cells so small? • Copy the following table for a sphere into your notes and fill it out: Diameter (cm) 2 4 6 8 10 Radius (cm) Surface Area (cm2) =4r2 Volume (mL) =4/3r3 Surface Area ÷ Volume What can you conclude? • How does surface area change as the radius increases? • As the size of an object increases, does the surface area or volume increase more rapidly? • What does this have to do with cells? – The amount of nutrients that a cell can take in and the amount of waste that can be expelled depends on the amount of surface area – Thus, as the cell size increases, at a certain point, the cell will not have enough surface area to support its volume Osmosis • Diffusion of water from an area of high concentration to an area of low concentration across a semi-permeable membrane *passive • If a solution has a high solute concentration then it must have a low water concentration (less space for water molecules) • A low solute concentration means high water concentration (more space for water molecules) • Note: the cell membrane is permeable to water Which way will water move? • HYPOTONIC conditions: when the water concentration outside the cell is greater than inside the cell…water moves INTO the cell H2O Outside Inside Hypotonic conditions contd. • If an animal cell is placed in a hypotonic solution, it could burst through a process called lysis • Plant cells become turgid (normal) Which way will water move? • HYPERTONIC conditions: when the water concentration inside the cell is greater than outside the cell…water moves OUT of the cell H2O Outside Inside Hypertonic conditions contd. • Cells placed in hypertonic solutions will shrink/shrivel (water exits) • Plasmolysis is a condition in plants where the cell membrane shrinks away from the cell wall – Plants will wilt because of a loss of turgor pressure (the pressure of the cell contents on the cell wall) – Salt on roads – good thing? Which way will water move? • ISOTONIC conditions: When water concentrations outside and inside the cell are equal, equal amounts of water move in and out of the cell H2O H2O Outside Inside Isotonic conditions contd. Isotonic conditions contd. • Animal cells in isotonic conditions are normal • Plant cells in isotonic conditions are flaccid (lacks stiffness) A B A B *Solutes cannot pass through the membrane • Side A is hypotonic to side A – why? – Side A has more water molecules than B • Water will always diffuse from a hypotonic solution to a hypertonic solution until both sides have equal concentrations hypotonic hypertonic hypertonic isotonic isotonic hypotonic Osmosis in action! • Watch this! Summary – Where is the net movement of water? • Isotonic solution – no net movement of water • Hypotonic solution – net movement of water into the cell • Hypertonic solution – net movement of water out of the cell Homework • Read the case study (Part I and II) and answer the questions