* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download ch3b FA11 - Cal State LA
Mitochondrion wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Pharmacometabolomics wikipedia , lookup
Enzyme inhibitor wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Biosynthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Biochemical cascade wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Metabolomics wikipedia , lookup
Electron transport chain wikipedia , lookup
Metabolic network modelling wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Basal metabolic rate wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Biochemistry wikipedia , lookup
Microbial metabolism wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Citric acid cycle wikipedia , lookup
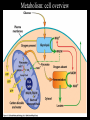
Metabolism • Collection of biochemical rxns within a cell • Metabolic pathways – Sequence of rxns – Each step catalyzed by a different enzyme • Enzymes of a pathway often physically interact to form large complexes – Limits amount of diffusion needed at each step of the pathway – The product of the preceding step is the reactant in the following step – Metabolic intermediates are the products formed along the way towards the ‘final’ product Pyruvate dehydrogenase 3 enzyme activities oxaloacetate Metabolism • Catabolic: breakdown from complex to simple – Yield raw materials for synthesis of other molecules – Convergent: diverse starting materials broken down to conserved set of intermediates (pyruvate, Acetyl-CoA) – Yield chemical energy transiently stored in NADH and ATP • Anabolic: synthesis from simple to complex – Consume raw materials and chemical energy stored in NADPH and ATP – Divergent Metabolism • Catabolic: breakdown from complex to simple – Yield raw materials for synthesis of other molecules – Convergent: diverse starting materials broken down to conserved set of intermediates (pyruvate, Acetyl-CoA) – Yield chemical energy transiently stored in NADH and ATP • Anabolic: synthesis from simple to complex – Consume raw materials and chemical energy stored in NADPH and ATP – Divergent Oxidation and reduction • Redox reactions: the gain (reduction) or loss (oxidation) of electrons – Reducing agents = lose e- = get oxidized – Oxidizing agents = gain e- = get reduced Fe0 + Cu2+ <---> Fe2+ + Cu0 Reducing agent + oxidizing agent <---> oxidized + reduced – Metals show complete transfer of e• Reducing agents reduce the charge on oxidizing agents Oxidation and reduction • Redox reactions: the gain (reduction) or loss (oxidation) of electrons – Changes in organic molecules shift the degree of e- sharing • Carbon in C-H bond is reduced • Carbon in C=O bond is oxidized – EN diffs result in e- spending less time around C when bonded to O CH4 + 2O2 --> CO2 + 2H2O Capture and Use of E • Alkanes are highly reduced organic compounds (E rich) – Not well tolerated by most cells • Fatty acids and sugars are well tolerated C6H12O6 + 6O2 --> 6CO2 + 6H2O ADP + Pi --> ATP • Theoretical Yield ~ 93 ATP • Actual (aerobic) ~ 36 ATP ΔG°’= -686 kcal/mol ΔG°’= +7.3 kcal/mol 39% efficient – Marathon runner • Actual (anaerobic) = 2 ATP – Sprinter 2% efficient Glycolysis • Glucose + 2NAD + 2ADP + 2Pi --> 2pyruvate + 2ATP + 2NADH K’eq ΔG°’ ΔG for actual cell conditions Two modes of E extraction • 1. Extraction of H+ and 2e- (:H-) – NAD+ + H: --> NADH – Extraction of :H- is done by dehydrogenase enzymes Nicotinamide Adenine Dinucleotide (NAD) • add :H- to the nicotinamide ring • Most NADH destined for electron-transport chain • Add phosphate to ribose 2’-OH creates NADP/NADPH rAMP • Another example of an ES complex with a covalent intermediate Two modes of E extraction • 2. Substrate level phosphorylation of ADP --> ATP – transfer of phosphate from higher energy compounds to lower energy ones • ATP is not the highest energy compound Another substratelevel phosphorylation in step 10 Glycolysis: summary • Steps 1, 3 – 2 ATP consumed • Step 4 – 6C sugar split into two 3C sugars • Step 6 – Redox reaction: NAD+ + :H- --> NADH • Step 7, 10 – Substrate level phosphorylation • Glucose + 2NAD+ + 2ADP + 2Pi --> 2Pyruvate + 2ATP + 2NADH • No O2 used, anaerobic Fermentation can regenerate NAD+ • Under anaerobic conditions – Skeletal muscle: Pyruvate + NADH ---> Lactate + NAD+ – Yeast: Pyruvate ---> Acetaldehyde + CO2 Acetaldehyde + NADH ---> Ethanol + NAD+ - O2 Fermentation can regenerate NAD+ • Under anaerobic conditions – Skeletal muscle: Pyruvate + NADH ---> Lactate + NAD+ – Yeast: Pyruvate ---> Acetaldehyde + CO2 Acetaldehyde + NADH ---> Ethanol + NAD+ • Under aerobic conditions – Pyruvate enters TCA cycle – NAD+ regenerated by electron transport chain (oxidative phosphorylation) + O2 Reducing power • Synthesis of fats from sugar requires reduction of metabolites H-C-OH + :H- + H+ ---> H-C-H + H2O • NADPH is used as reducing agent for Anabolic pathways NADH + NADP+ <---> NAD+ + NADPH transhydrogenase Metabolic regulation • Covalent modification of enzymes – Phosphorylation uncharged charged • Serine H2C-OH --> H2C-O-PO32protein kinases P enz enz protein phosphatases • Threonine • Tyrosine • e.g., phosphorylation activates glycogen phosphorylase Metabolic regulation • Allosteric modulation (Allostery) – Binding of a molecule to the enzyme activates or inhibits it – Binding occurs at an ‘allosteric site’ on the enzyme – Feedback inhibition: • Final product of a pathway inhibits the first enzyme in the pathway • Keeps level of product from getting higher than needed • A + B --> C ; C + D --> E • E is an allosteric inhibitor that binds to allosteric site blocking 1st rxn Metabolic regulation • Most cells have enzymes for both glycolysis and gluconeogenesis • Allostery controls which is dominant and provides sensitivity to energy needs • Step 2, phosphofructokinase – ATP = allosteric inhibitor – AMP = allosteric activator • Step 2, fructose bisphosphatase – AMP = allosteric inhibitor ATP --> ADP + Pi ADP + ADP --> ATP + AMP Metabolism: cell overview The Metabolome • The collection of all metabolites within a given cell or organism • Metabolomics: the systematic study of the unique chemical fingerprints of various cellular processes – The liver metabolome versus the muscle metabolome – The cancer metabolome(s) dogs can smell breast, lung, skin cancer with 88-97% accuracy! – Other disease metabolomes