* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Stryer An overview of the citric acid cycle
Genetic code wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Mitochondrial replacement therapy wikipedia , lookup
Peptide synthesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Butyric acid wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Phosphorylation wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Specialized pro-resolving mediators wikipedia , lookup
Mitochondrion wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Microbial metabolism wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Electron transport chain wikipedia , lookup
Biochemistry wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
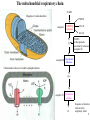
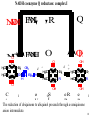
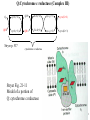
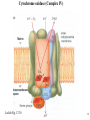
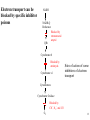
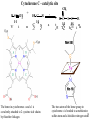
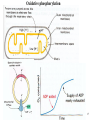
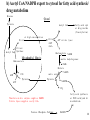
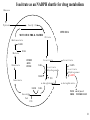
Lecture 6B – outline Mitochondrial function (e.g. hepatocytes) 1) citric acid cycle as an energy source a) pyruvate or a-ketoglutarate dehydrogenase b) lipoic acid therapy 2) the respiratory chain as an energy source 3) oxidative phosphorylation and uncouplers 4) membrane transporters and shuttles a) cytosolic NADH oxidation b) acetyl CoA (NADPH export) c) transport systems in the mitochondria d) gluconeogenesis and glucose transport 1 Compartmentalization of the major pathways of metabolism 2 1. CITRIC ACID CYCLE AS AN ENERGY SOURCE An overview of the citric acid cycle Stryer 3 Acetyl CoA + 3 NAD+ + FAD + GDP + Pi + 2 H2O 2 CO2 + 3 NADH + FADH2 + GTP + 2H+ + CoA toxic! 120uM plasma citrate complexes Fe 4 The citric acid cycle is a source of biosynthetic precursors Glucose Pyruvate ATP, CO2 Phosphoenolpyruvate Acetyl CoA ADP, Pi Amino acids Oxaloacetate Succinyl CoA Porphyrins Citrate Stryer Fig. 20-17. Biosynthetic roles of the citric acid cycle. Intermediates drawn off for biosyntheses are replenished by the formation of oxaloacetate from pyruvate. aketoglutarate Amino acids 5 Control of the citric acid cycle Stryer Fig. 20-22. Control of the citric acid cycle and the oxidative decarboxylation of pyruvate: * indicates steps that require an electron acceptor (NAD+ or FAD) that is regenerated by the respiratory chain. 6 2. THE MITOCHONRIAL RESPIRATORY CHAIN AS AN ENERGY SOURCE 7 The mitochondrial respiratory chain NADH Diagram of a mitochondrion FMNH2 complex I NADH-Q reductase 2Fe-2S 4Fe-4S Q FADH2 in flavoproteins succinate:Q reductase (complex II) complex III Cytochrome reductase Chemiosmotic theory of oxidative phosphorylation cyt c complex IV Cytochrome oxidase O2 Sequence of electron carriers in the respiratory chain 8 Origin of mitochondria: the endosymbiont hypothesis The endosymbiont hypothesis suggests that mitochondria have evolved from anaerobic bacteria which were phagocytosed by eukaryote cells at the time oxygen appeared on earth, Similarities between mitochondria and bacteria include the presence of: • cardiolipin •transporters • ribosomes • circular RNA and DNA Therefore mitochondria protein synthesis should be inhibited by: • TETRACYCLINE • CHLORAMPHENICOL. E.g. The extensive use of these drugs can inhibit 1. Bone marrow mitochondrial protein synthesis leading to a decline in the production of white or red cells. 2. Intestinal epithelial cells causing them to cease dividing. 9 NADH coenzyme Q reductase: complex I M N N A D H F + N A D O O F M N H 2 H C OC 3 H C OC 3 C C H 3 C H 3 N C (C H C C C H )10 H 2 2 C H O Q H 2x O C e- Q e R + H C OC 3 H C OC 3 C O H eC C H 3 C R C O H + + A H OC 3C H OC 3C C C C H 3 C R C O H C o eR e 1 0S I n ( U B The reduction of ubiquinone to ubiquinol proceeds through a semiquinone anion intermediate. 1 10 Q:Cytochrome c reductase (Complex III) Q cyt b (+2) QH Fe-S(+2) cyt c1(+3) cyt c(Fe+2) QH cyt b (+3) QH2 Fe-S(+3) cyt c1(+2) cyt c(Fe+3) Stryer p. 537 cytochrome c reductase Stryer Fig. 21-11 Model of a portion of Q: cytochrome c reductase 11 Cytochrome oxidase (Complex IV) Lodish Fig. 17-30 12 Electron transport can be blocked by specific inhibitor poisons NADH NADH-Q Reductase QH2 Blocked by rotenone and amytal Cytochrome b Blocked by antimycin Cytochrome c1 Sites of action of some inhibitors of electron transport Cytochrome c Cytochrome Oxidase Blocked by CN- , N3 -, and CO O2 13 Cytochrome C - catalytic site RC C H 2 H V i o f + n C H 3 H SC H C 2 y o t R y l f h The heme in cytochromes c and c1 is covalently attached to 2 cysteine side chains by thioether linkages ' s e RC S C H t e iH 2 g Tr o h t h e h e R ' n e u i opp m The iron atom of the heme group in cytochrome c is bonded to a methionine 14 sulfur atom and a histidine nitrogen atom Cytochrome C - soluble NOT membrane bound 1. 26/104 amino acids residues have been invariant for > 1.5 x 109 years. 2. Met 80 and His 18 - coordinate Fe. 3. 11 residues from number 70 - 80 lining a hydrophobic crevice have remained virtually unchanged throughout all cytochrome c regardless of species or even kingdom. 4. A number of invariant arginine and lysine clusters can be found on the surface of the molecule. Cytochrome c has a dual function in the cell. Electron transport for ATP production AND the major cause of most programmed cell death (apoptosis) is initiated by the release of cytochrome c into the cytosol! 15 3. OXIDATIVE PHOSPHORYLATION AND UNCOUPLERS 16 Oxidative phosphorylation 17 4. Mitochondrial MEMBRANE TRANSPORTERS A) Cytosolic NADH oxidation B) Acetyl CoA (NADPH export) C) Transport systems in the mitochondria D) Gluconeogenesis and glucose transport 18 a) Cytosolic NADH oxidation: membrane transporters glycerol phosphate shuttle (Bucher shuttle) 19 b) Acetyl CoA/NADPH export to cytosol for fatty acid synthesis/ drug metabolism Gl uc o s e Cyt osol Py r uv a t e Ac e t y l CoA at hi gh c on c e n t r a t i o n Ci t r a t e Ci t r a t e Ac e t y l CoA Sy nt h as e Ci t r a t e +ATP +CoA f a t t y a c i d s yn t he or dr ug m e t a b ol i ( N- a c e t y l a t i on ) ATP c i t r at e l y as e Ox a l o a c e t a t e NADH Mi t ochondr i al Mat r i x ma l at e d e hy dr o ge n as e Ox a l o a c e t a t e NAD+ ADP M al a t e NADP+ ma l i c e n z y me CO2 Py r uv a t e Py r uv a t e ATP NADPH CO2 f a t t y a c i d s yn t he s i s or P4 50 c a t a l y z e d dr ug m e t a bol i s m Th e r e f or e m a l i c e nz ym e s u ppl i e s NADPH Ci t r a t e Lya s e s up pl i e s a c e t yl CoA. Pe nt o s e Pho s ph a t e Pa t hwa y NADPH 20 Isocitrate as an NADPH shuttle for drug metabolism Gl uc os e Py r u va t e Ac e t yl CoA CYTO SO L M I TO CH O NDRI AL M ATRI X Ci t r a t e Ox a l oa c e t a t e NADH M al at e Fu m a r a t e NAD+ CI TRI C ACI D CYCLE I s oc i t r a t e NAD+ NADH Su c c i n a t e i s oc i t r a t e de hy dr og e n as e NADP+ i s oc i t r a t e de hy dr og e n as e NADPH CO 2 a- k e t og l u t a r a t e NADH I s oc i t r a t e a- k e t og l u t a r a t e NAD+ P4 50 c at al yz e d DRUG M ETABO LI S M Su c c i n yl CoA CO2 21 d) Gluconeogenesis and glucose export by the liver ! 3 irreversible steps Major antidiabetic drug METFORMIN Inhibits gluconeogenesis Decr Hepatic Glucose Synth. 22 Glucagon 51aa & Insulin 29aa • Pancreas synthesises both peptide hormones • Insulin required for cells (e.g.liver,muscle,fat) to take up glucose and synthesise glycogen. • Glucagon hepatocyte receptors signals glycogenolysis (glycogen breakdown to glucose then increases gluconeogenesis pyruvate -- glucose) • Drugs. Dipeptidyl peptidase-4 inhibitor (Januvia, new anti type 2 diabetes) increases incretin , a GI hormonal peptide inhibitor of glucagon which lowers plasma glucose. • Metformin, antidiabetic drug inhibits gluconeogenesis but also can inhibit mitoch.complex I causing lactic acidosis. 23