* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Kinetic resolution wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Elias James Corey wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Marcus theory wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Aromaticity wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
George S. Hammond wikipedia , lookup
Discodermolide wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Ene reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Aromatization wikipedia , lookup
Asymmetric induction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Hydroformylation wikipedia , lookup
Wolff rearrangement wikipedia , lookup
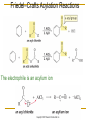
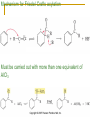
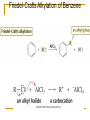
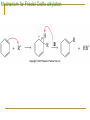
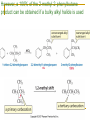
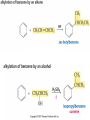
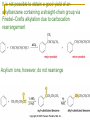
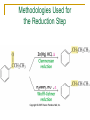
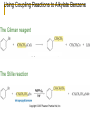
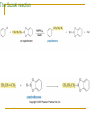
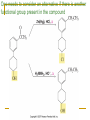
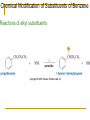
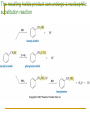
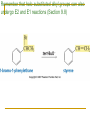
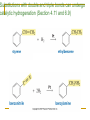
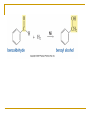
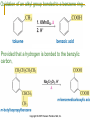
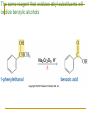
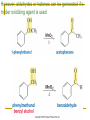
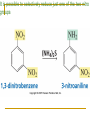
Friedel–Crafts Acylation Reactions The electrophile is an acylium ion Mechanism for Friedel-Crafts acylation Must be carried out with more than one equivalent of AlCl3 Friedel-Crafts Alkylation of Benzene Mechanism for Friedel-Crafts alkylation The carbocation will rearrange to a more stable species However, a 100% of the 2-methyl-2-phenylbutane product can be obtained if a bulky alkyl halide is used It is not possible to obtain a good yield of an alkylbenzene containing a straight-chain group via Friedel–Crafts alkylation due to carbocation rearrangement Acylium ions, however, do not rearrange Methodologies Used for the Reduction Step Using Coupling Reactions to Alkylate Benzene The Gilman reagent The Stille reaction The Suzuki reaction One needs to consider an alternative if there is another functional group present in the compound Chemical Modification of Substituents of Benzene Reactions of alkyl substituents The resulting halide product can undergo a nucleophilic substitution reaction Remember that halo-substituted alkyl groups can also undergo E2 and E1 reactions (Section 9.8) Substitutions with double and triple bonds can undergo catalytic hydrogenation (Section 4.11 and 6.9) Oxidation of an alkyl group bonded to a benzene ring… Provided that a hydrogen is bonded to the benzylic carbon, The same reagent that oxidizes alkyl substituents will oxidize benzylic alcohols However, aldehydes or ketones can be generated if a milder oxidizing agent is used Reducing a Nitro Substituent It is possible to selectively reduce just one of the two nitro groups