* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PowerPoint 演示文稿
Kinetic isotope effect wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Elias James Corey wikipedia , lookup
Discodermolide wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Marcus theory wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Ene reaction wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
George S. Hammond wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
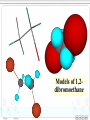
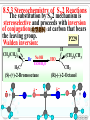
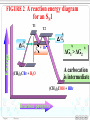
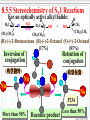
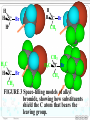
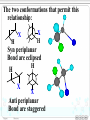
Chapter 8 Alkyl Halides 8.1 IUPAC Nomenclature of Alkyl Halides 8.2 Classes of Alkyl Halides 8.3 Preparation of Alkyl Halides 8.3.1 Addition of HX or X2 to Alkenes, Alkynes 8.3.2 Preparation of Alkyl Halides from Alcohols and HX 8.3.3 Exchange between Halides 8.3.4 Halogenation of Alkanes A. Chlorination of Methane Substitution reaction B. Mechanism of Methane Chlorination Homolytic breaking Heterolytic breaking Radical reactions Chain reactions Stability of alkyl radicals 8.3.5 Allylic Bromination of Alkenes 8.4 Reactions of Alkyl Halides 8.4.1The sites of reactions of alkyl halides 8.5 Nucleophilic Substitution 8.5.1 Nucleophilic Substitution 8.5.2 A Mechanism for the SN2 Reaction 8.5.3 Stereochemistry of SN2 Reactions 8.5.4 A Mechanism for the SN1 Reaction 8.5.5 Stereochemistry of SN1 Reactions 8.5.6 Factors Affecting the Rate of SN1 Reactions and SN2 reactions 1. The structures of substrates 2. The Nucleophile 3. The leaving group 4. The solvent 8.6 Elimination Reactions 8.6.1 Dehydrohalogenation of Alkyl halides 8.6.2 Dehydration of Alcohols 8.6.3 Mechanisms of Elimination Reactions A. The E2 reaction B. The E1 Reaction 8.6.4 Stereochemistry of Elimination Reactions 8.6.5 Nucleophilic Substitution Versus Elimination 1.The structure of the substrate 2. The basicity of the reagent 3. The temperature of the reaction Halogen-substiuted organic R H X : compounds. F H Cl Cl Cl Cl C H F Cl Trichloroethylene (solvent) Br C H H Dichlodifloro- Bromomethane a fumigant methane (薰剂) a refrigerant F F Cl C C H F Br Halothane(氟烷) (a anesthetic)(麻醉剂) 8.1 IUPAC Nomenclature of Alkyl Halides a. Halogen: as a functional group P218 For simple alkyl groups: Common name Alkyl + halide H I Cl Ex. CH3F Methyl floride Pentyl chloride (甲基氟) (戊基氯) Cyclohexyl iodide (环己基碘) b. Halogen as a substituent. Subsitutive For branched alkyl groups: names • Number: begin at the end nearer the first substituent, regardless of X- or R-. • Properly numbered from either end, list them in alphabetical order. CH3 Cl CH3 CH3CHCH2CHCHCH2CH3 1 2 3 4 Cl 5 6 7 CH3CHCH2CH2CHCH3 6 5 4 3 2 Br 1 4,5-Dichloro-2-methylheptane (2-甲基-4,5-二氯庚烷 ) 2-Bromo-5-methylhexane (2-甲基-5-溴己烷) 8.2 Classes of Alkyl Halides According to the type of the carbon that bears the functional group. RCH2X Primary alkyl halides (伯卤代烃) CH3 RCHR' Secondary Alkyl halides CH3CCH2Br (仲卤代烃) X CH3CHCH2CH3 CH3 Br 2-Bromobutane 1-Bromo-2,2-dimethyl propane CH3 H3C Cl R3CX Tertiary alkyl halides (叔卤代烃) cis-1,4-Dimethyl chlorocyclohexane Models of 1,2dibromoethane 8.3 Preparation of Alkyl Halides 8.3.1 Addition of HX or X2 to Alkenes, Alkynes O O Ex. H2C CHCH2Cl + HBr PhCOOCPh BrCH2CH2CH2Cl 8.3.2 Preparation of Alkyl Halides from Alcohols and HX P222, 7.3 Ex. OH + HBr heat Br + H2O (73%) Reagents: HBr, HCl, PX3, PX5, SOCl. (Thionyl chloride) (亚硫酰氯) The order of reactitivity: HX: HI > HBr > HCl >> HF Alcohols: 3°> 2° > 1° > Methanol 8.3.3 Exchange between Halides CH3 CH3 Ex. H3C C Cl + NaI ZnCl2, CS2 r.t H3C CH2CH3 8.3.4 Halogenation of Alkanes R H + X2 h or heat CH4 h or heat CH3Cl h CH2Cl2 Chloro- Dichloromethane Methane CH2CH3 (96%) Carbon tetrachloride A. Chlorination of Methane: Cl2 + NaCl I R X + HX Common reagents: Cl2 or Br2. Reactivity: F2 > Cl2 > Br2 >I2 Cl2 C Ch.P 178 Cl2 h (四氯化碳) CHCl3 Cl2 h CCl4 Chloroform Tetrachloro methane (氯仿) Substitution reaction: The reaction in which a atom or a group in mole. is replaced by another one. B. Mechanism of Methane Chlorination Two ways for the breaking of a covalent bond: Homolytic cleveage(均裂): Reactive intermediates: radicals or free radicals Methane chlorination A species that bears is a homolytic cleveage. a unpaired electron. A B A + B Heterolytic cleavage (异裂): A B A + B Polar breaking: cation and anion Radical reaction: The reaction is promoted by light or by heat. Chain initiation (链的引发阶段) initiator Step 1 Dissociation of a chlorine mole.. Cl Cl h 2 Cl P219,7.2 Chain propagation (链的增长阶段) Step 2 Radicals react with a mole. Cl H + CH3 The radical Cl + H CH3 reacts with Cl CH3 + Cl Cl Cl + CH3 the mole. of product Cl + H CH2Cl Cl Cl + CH2Cl Cl H + CH2Cl Cl CH2Cl + Cl Chain termination (链的终止阶段) Step 3 The reactions between the radicals. Cl + CH3 H3C + CH3 Cl + Cl Cl CH3 H3C CH3 Cl Cl Chain reactions: The chain initiation is rate-determing step, to product the radical. The reaction whose mechanisms invole a series of steps with each step producing a reactive intermediate that cause the next step to occur. Halogenation hof higher alkanes: CH3CH2CH2CH3 + Cl2 H 35 ° CH3CH2CH2CH2Cl + CH3CHCH2CH3 H 28% R R C R C R C H R R Cl 72% Primary < Secondary < Tertiary Increasing the stability of alkyl radicals The reaction gives a mixture of isomers: CH3CHCH3 CH3 CH3 CH3 Cl2 h / 25 ° CH3CHCH2Cl + CH3CCH3 Primary hydrogen Tertiary hydrogen (63%) (37%) Cl 9 1 63% 37% The rate of the reactivity for the hydrogen: Tertiary hygrogen = 37% ≈5.0 Primary hydrogen 63% / 9 Reactivity for different type of hydrogens H R H in mole.: R C H R C H R C H H R R Bromination Primary < Secondary < Tertiary 1.0 3.5 5.0 is higher selective: Increasing the reactivity CH3 CH3 CH3CHCH2CH2CH3 + Br2 h 60 ° CH3CCH2CH2CH3 + HBr Br (76%) 8.3.5 Allylic Bromination of Alkenes O NBS N Br H H Br O h¦Í ,CCl4 O + Mechanism of the reaction: O N O O H + Br + Br + HBr Allylic radical O HBr + O O N Br h¦Í H H N H N Br O Br2 + H Br Br2 + Br Stability of radicals O vinylic < methyl N H < 1°< 2°< 3° O < allyic 7.4 Reactions of Alkyl Halides The sites of reactions of alkyl halides : A polar covalent bond readily broken Nucleophilic substitution δ+ C H Elimination C H :B δ- X Nu: 8.5 Nucelophilic Substitution Paul Walden made a remarkable discovery: O O O O PCl5 HOCCH2CHCOH Et O HOCCH2CHCOH 2 OH (–)-Malic acid Cl (+)-Chlorosuccinic acid (苹果酸) [α]D = -2.3° (卤代琥珀酸) AgO, H 2O AgO, H 2O O P225,7.5 O PCl5 HOCCH2CHCOH Ether Cl (–)-Chlorosuccinic acid O O HOCCH2CHCOH OH (+)-Malic acid Inversion in configuration [α]D = +2.3° Born 14 July 1863; died 24 January 1957. Paul Walden was a Latvian chemist who, while teaching at Riga, discovered the Walden inversion, a reversal of stereochemical configuration that occurs in many reactions of covalent compounds (1896). Due to this discovery, Walden's name is Paul Walden mentioned almost in all textbooks on organic chemistry published throughout the world. 1863-1957 Walden revealed autoracemization and put the foundations to electrochemistry of nonaqueous solutions. Walden is also known for Walden's rule, which relates the conductivity and viscosity of nonaqueous solutions. 8.5.1Nucelophilic Substitution(亲核取代反应) General type of the reaction: Nu + C C L P226 Nu + L Ex. The reaction of ROH with HX HX + R OH heat 1. R X + H2O 2. H + O H R X The reaction of alkyl halides with sodium hydroxide: HO + H3C Br X and HO CH3OH + Br are nuclophiles(亲核试剂) Nucleophiles: A species with an unshared electron pair. A Lewis base. A reagent attacks a sp3-hybridized carbon with partially positive charge,displacing a substituent. A nucleophilic substitution reaction (亲核取代反应) The leaving group: a substituent departs from central carbon with a pair of bonding electrons. Substrates: like alkyl halides (卤代烷), a sp3-hybridized carbon with a leaving group. C X C C X X H2C CHCH2 X C C X Functional groups transformation by nucleophilic substitution reactions of alkyl halides Ex. CH3ONa + CH3CH2Br CH3CHCH3 + NaI Br P227, Table 7.1 acetone CH3OCH2CH3 + NaBr CH3CHCH3 + NaBr I 8.5.2 A Mechanism for the SN2 Reaction (Substitution nucleophilic bimolecular) SN1 Mechanism of nucleophlic substitution SN2 Hydrolysis of methyl bromide: Hydroxide ion in aqueous solution(水溶液). HO + H3C Br H2O CH3OH +Br Reaction rate = k[CH3Br][-OH] Alkali(碱) In a single step process without intermediate. Second-order reaction kinetics P228,7.7 (二级反应) HH H HO + H H C Br - HO C H H - Br HO C H + Br H An transition state • The nucleophile approaches the central C from back side – the side opposite the bond to the leaving group Nu L C + Nu C L + + Energy △G + OH + H H C Br H H HO C L Ch.P188, (五) HO C Br - C Nu H H Reaction progress + Br • The formation of a bond of C-Nu and the breaking of C-L occur at the same time. • The breaking of a bond is assisted by the formation of a bond . 8.5.3 Stereochemistry of SN2 Reactions The substitution by SN2 mechanism is stereoselective and proceeds with inversion of conjugation(构型翻转) at carbon that bears the leaving group. P229 Walden inversion: H H CH3(CH2)5 H3C C Br NaOH EtOH-H2O (S)-(+)-2-Bromooctane + δ- HO C (CH2)5CH3 CH3 (R)-(-)-2-Octanol δ- + Like an umbrella in the gale An inversion of configuration Cl H H3C H + OH H3C H H OH + Cl 8.5.4 A Mechanism for the SN1 Reaction (Substitution Nucleophilic Unimolecular) In the hydrolysis of tert-butyl bromide: P232,7.8 (CH3)3C Br + H2O CH3)3C OH + HBr Reaction rate = k[CH3)3C-Br] A first-order reaction Edward Davies Hughes (1906-1963) Sir Christopher (Kelk) Ingold 1893-1970 ** Ingold was one of the founders of the electronic theory of organic chemistry and made many contributions to reaction mechanisms and molecular spectroscopy. Orientation and relative rates of aromatic nitration were used, in his early work, to test the theory. Studies of aliphatic substitutions and eliminations, often with his long-time collaborator E. D. Hughes, led to I ncorporation into the standard language of chemistry of such words as nucleophile, electrophile, inductive and mesomeric (resonance) effects, and such symbols as SN1, SN2, E1, E2, BAC2 and others. His monumental book "Structure and Mechanism in Organic Chemistry" (1953) was for years an authoritative text in the field. His forays into molecular spectroscopy first demonstrated the hexagonal symmetry of benzene's ground state and gave a quantitative description of its first excited state; he was the first to show that the first excited state of acetylene is bent. Ingold sought to unite physical, organic and inorganic chemistry. His papers were models of exposition, clarity and precision. The vigorous and sometimes vitriolic manner with which he dealt with opponents, in print, contrasted markedly with his personal kindness and courtesy. Ingold received many awards including the Davy (1946) and Royal (1952) Medals of the Royal Society and the first James Flack Norris Award in Physical Organic Chemistry of the ACS (1965). Mechanism of the reaction: Step1 The alkyl halide dissociates to a carbocation and a halide. CH3 CH3 H3C slow C Br (rate-determing step) H3C C + Br CH3 CH3 Step 2 The carbocation reacts rapidly with water as nucleophile to produce an alkyloxonium ion. CH3 CH3 H3C + C O H fast H3C H C O CH3 CH3 H H Step 3 The transfer of a proto to a mole. of water to produce a neutral alcohol. A fast acidCH3 CH3 H base reaction H fast H C C OH HC C O 3 CH3 H + O 3 H CH3 FIGURE 2 A reaction energy diagram for an SN1 T1 T2 + + △ G2 + + △G1 + + C + Br ΔG1 > ΔG2 + + A carbocation is intermediate (CH3)3CBr + H2O (CH3)3COH + HBr Reaction progress 8.5.5 Stereochemistry of SN1 Reactions For an optically active alkyl halide: H H H3C H 2O C Br EtOH CH3(CH2)5 H3C C OH + CH3(CH2)5 H HO C CH3 (CH2)5CH3 (R)-(-)-2-Bromooctane (R)-(-)-2-Octanol (S)-(+)-2-Octanol (17%) (83%) Inversion of conjugation Retention of conjugation (构型翻转) (构型保留) Nu Nu Nu + More than 50% Racemic product Nu P234 Less than 50% 8.5.6 Factors Affecting the Rate of SN1 Reactions and SN2 reactions 1. Structure of substrates SN2 reactions: The order of reactivity: Tertiary < Secondary < Primary < Methyl Relative 500 40,000 2,000,000 reactivity <1 Increasing SN2 reactivity CH3 H3C R The steric hindrance R' C X of alkyl group. C CH2 X CH3 Nu Neopentyl halides is very unreactive. R'' P230 Nucleophile carries out a back-side displacement H H C Br H H3C H C Br H H C Br CH3 CH3 H3C C Br CH3 CH3 FIGURE 3 Space-filling models of alkyl bromide, showing how substituents shield the C atom that bears the leaving group. SN1 reactions CH3 RCH2 R2CH RCH CHCH2 CH2 R3C Methyl < Primary < Secondary = Allyl = Benzyl < Tertary Less stable Carbocation stability Reactivity for SN1 reactions 2. Nucleophiles More stable Ch. P195 The rates of SN2 reactions depends on both the concentration and identity of the attacking Nucleophile: The nucleophile is usually a Lewis base Increasing the concentration of a nucleophile increases the rate of an SN2 reaction. The stronger the nucleophilicity of a reagent, the more rapid the reaction. • Nucleophilicity roughly parallels basicity when comparing nucleophiles that have the same attacking atom. RO > HO >> RCOO > ROH > H2O • Negatively charged nucleophiles are usually more reactive than neutral ones. HO > H2O RO > ROH • nuclophilicity is also related to polarizability (可极化度). Distortion RS > RO , I > Br > Cl > F 3. The leaving group The leaving group affects both SN2 reactions and SN1 reactions. The best leaving groups should be the weakest bases. The weak bases stabilize a negative charge most effectively I TsO Br HO , NH2 , OR Cl F Relative 1 200 10,000 30,000 60,000 reactivity << 1 The greater the extent of charge stabilization by the leaving group, the lower the energy of the transition state for SN2 reaction and the more rapid the reaction. The best leaving groups: O S CH3 P326,8.14 O CH3 S O O O O p-Toluenesulfonate Methylsulfonate ion(对甲基苯磺酸根负离子) ion( 甲磺酸根负离子) TsO MsO O O Tosylate: ROH + CH3 S Cl RO S O CH3 + HCl O Transformation of leaving R OTs + Nu R Nu + OTs groups. Basicity: OH >> H2O R OH H H Br R O H R Br + H2O 4. The solvent P320, 8.12 SN1 reactions: Dielectric constant (ε)(介电常数) is a parameter to measure the polarity of solvent. The polar solvent with a higher dielectric constant. Polar solvents favor the dissociation of alkyl halides to form carbocations. Solvent mole. orient around H H H H the carbocation. CH CH O H H O 3 O + C O H H O O H H H H H3C C Cl + ROH CH3 3 H3C C OR + HCl CH3 EtOH 40% H2O/ 80% H2O/ H2O 60%EtOH 20%EtOH Relative reactivity: 1 100 14,000 100,000 more Less Solvent reactivity reactivity reactivity SN2 reaction: Protic solvents: containing –OH, –NH groups. worst solvents for SN2 reactions. They all have active hydrogen atoms that allow them to form hydrogen bonds with nuclophiles, so that decrease the nucleophilicity of reagents. O R In contrast to protic solvent, H O O R polar aprotic solvents increase R H Nu H the rates of SN2 reactions. H O Ex. R CH3 O O H C N CH3 CH3 O CH3 S N CH3 P N CH3 N CH3 H3C CH3 CH3 N,N-Dimethyl formamide Dimethyl sulfoxide (DMF)(N,N-二甲基甲酰胺) (DMSO)(二甲亚砜) HMPA or HMPT CH3CH2CH2CH2 Br + N3- CH3CH2CH2CH2 N3 + Br- Solvent CH3OH H2O DMSO DMF CH3CN HMPA Relative 1 7 1,300 2,800 5,000 200,000 reactivity Less reactive Solvent reactivity More reactive 8.6 Elimination Reactions 1. Nucleophilic substitutions Ionic reactions 2. Elimination reactions The Eliminaiton Reaction: C C Y L Elimination -YL C C P124, 4.9 The atoms or the groups are removed from adjacent C atoms in a molecule is βElimination or 1,2-Elimination. 8.6.1 Dehydrohalogenation of Alkyl halides The loss of a hydrogen and a halogen from adjacent C atoms of an alkyl halide to yield an alkene. Ex. CH3CHCH3 EtONa ° EtOH, 55° CH3CH CH2 + NaBr + EtOH (79%) Br The reaction is carried out in the present of a stronger base. EtONa-EtOH KOH-EtOH KOC(CH3)3- ROH: 2EtOH + 2Na is used to primary alkyl halides 2EtONa + H2 H C β C α + B C C + HB + X X The base attacks the β–H, X: leaving group The regioselectivity of the elimination: H CH3 H H C C C CH3 EtOK °° H2C EtOH, 70 CH3 CH3 C + C CH2CH2 CH3 (29%) H Br H CHCH3 (71%) The elimination follows Zaitsev’s rule: βelimination predominates in the direction that leads to the more highly substituted alkene. 8.6.2 Dehydration of Alcohols P126 The loss of a hydrogen and a hydroxyl group from adjacent C atoms of an alcohol by acid-catalyst: CH3 H3C C OH H2SO4 CH3 Heat CH3 CH3 C CH2 + H2O (82%) The reaction also follows Zaisev’s rule: CH3 OH 2-Methylcyclohexanol CH3 H2SO4 Heat CH3 + P236,7.9 1-Methyl3-Methylcyclohexene cyclohexene (84%) (16%) Questions: For dehydration of alcohols, why the acid must be used? Which group is leaving group? 8.6.3 Mechanisms of Elimination Reactions A. The E2 reaction (Elimination bimolecular): C 2H5O - + + C2H5O H H C C C X C X - C C + C2H5OH + Br Rate = k R X C2H5O At the same time, 1. C-H bond breaking 2. C=C πbond formation 3. C-X bond breaking take place. Central C atom: sp3-hybrid sp2-hybrid Reactivity of the E2 reaction: RF < RCl < RBr < RI The lower strength of C-X, the higher reactivity. B. The E1 Reaction(Elimination unimolecular) (CH3)3C OH + (CH3)3C OC2H5 (CH3)3C Cl 80%C2H5OH 20%H2O (83%) Substitution H2C C(CH3)2 (17%) Elimination For the elimination: Rate = k[ (CH3)3C Cl] P239,7.10 Step 1 Alkyl halide dissociates by heterolytic cleavage of C-X bond. CH3 H3C C Cl CH3 Slow CH3 H3C C + Cl CH3 Step 2 EtOH acts at a base to remove a proton from the carbocation to give the alkene. H CH3CH2O H CH3 H C C Fast CH3CH2O H H + H2C H CH3 Step1 is rate-determining one. C CH3 CH3 Reactivity of the E1 reaction: RCH2X < R2CHX < R3CX Incresing rate of the E1 reaction The similar mechanism for the dehydration of alcohols. 8.6.4 Stereochemistry of Elimination Reactions The E2 Reaction: In the transition state of the E2 reaction, - B H C H C C X - C X With a periplanar (全平面的) geometry. The two conformations that permit this relationship: X H X H Syn periplanar Bond are eclipsed H H X X Anti periplanar Bond are staggered B H + H B H C C Br Br Br β- H and X are on the opposite side. Anti elimination(反式消除) for the E2 reaction. Br H H cis-1,4- KOC(CH3)3 HOC(CH3)3 (1) k 1 > k2 KOC(CH3)3 Br H HOC(CH3)3 (2) H trans-1,4- conformations determined from electron diffraction studies by the Norwegian physical chemist Odd Hassel. Thus conformational analysis, which ever after changed the way organic chemists think about structure, synthesis and chemical reactivity, came into being. Barton and Hassel shared the 1969 Nobel Prize in Chemistry for their seminal work. In a long and distinguished career, Barton went on to make major contributions to organic photochemistry (the Barton Reaction), and to the invention of new reactions and their application to natural products synthesis. A native of England, Barton's initial positions were at Imperial College (London), Harvard, Birkbeck College (London) and Glasgow University. He became Professor at Imperial College, London (1955-1978), then Director of the Institut de Chemie des Derek Harold Richard Barton Substances Naturelles in Gif-sur-Yvette (France, 1977-85), and in 1986 he became 1918-1997 Dow Distinguished Professor of Chemical In 1950 Barton published a 4-page Invention at Texas A. and M. University. paper in Experientia entitled "The He received many honors besides the Nobel, Conformation of the Steroid including the 1995 Priestley Medal (USA) and the 1995 Lavoisier Medal (France); Nucleus", in which he analyzed an unusual honor for a chemist was his how molecular shape affects chemical behaviour. The paper was appointment (1989) by the Governor of developed using cyclohexane ring Kentucky as a Kentucky Colonel. 8.6.5 Nucleophilic Substitution Versus Elimination P239, 7.11 C (a) H C Nu C X E2 C + Nu H + X (b) H C SN2 Nu C Elimination + X Substitution The three factors that affect the relative rates of E2 and SN2 reaction: 1.The structure of the substrate CH3CH2O Na + CH3CH2Br C2H5OH CH3CH2OCH2CH3 + CH2 CH2 ° 55 C -NaBr For primary alkyl halide, the substitution is highly favored. SN2 (90%) E2 (10%) CH3CHCH3 + CH3CH CH2 CH3CH2O Na + CH3CHCH3 C2H5OH ° 55 C Br -NaBr OCH2CH3 S N2 For secondary alkyl halide, elimination reaction is highly (21%) favored. As crowding at the E2 (79%) central C atom decreases, E2of nucleothe rate philic attack by the Lewis base increases. CH3CH2O SN2 How about tertiary halide in the presence of a stronger Lewis base? Ex. CH3 CH3CH2O Na + H3C C Br CH3 C H OH CH3 2 255°C -NaBr CH3 C H3C C CH3 + CH3 OCH2CH3 2. The basicity of the reagent (9%) CH2 (91%) CH3O + CH3(CH2)15CH2CH2 Br CH3OH CH3(CH2)15CH CH2 65 °C + E2 SN 2 CH3(CH2)15CH2CH2 OCH3 (1%) (99%) CH3 H3C C O + CH3(CH2)15CH2CH2 Br (CH3)3COH 40 °C E2 SN2 (85%)(15%) CH3 CH3(CH2)15CH CH2 + CH3(CH2)15CH2CH2 OC(CH3)3 • The elimination reaction is favored in the presence of a stronger sterically hindered base. • Increasing the nucleophilicity of the reagent favors the SN2 reaction. Less basicity than hydroxide: CH3CH(CH2)5CH3 KCN CH3CH(CH2)5CH3 DMSO Cl 2-Chlorooctane CN 2-Cyanooctane(70%) (2-氰基辛烷) 3. The temperature of the reaction Increasing the temperature of the reaction favors E2 reaction. Problems to Chapter 8 P246 7.22(c), (d) 7.23(b) 7.29 7.30(b), (c), (f) 7.32 7.33(a) 7.34 7.35(b) 7.39 7.42 7.45 7.46 7.47 7.48 7.49 7.51 7.52 7.53 7.55 7.56 7.58 Additional Problems: 1* Among the Walden cycles carried out by Kenyon and Phillips is the following series of reactions reported in 1923. Explain the results, and indicate where Walden inversion is occurring. OH OTs CH3CHCH2 TsCl [α]D = + 33.0° CH3CHCH2 CH3CH2OH [α]D = + 31.1° OCH2CH3 CH3CHCH2 [α]D = - 19.9° K O K+ CH3CHCH2 CH3CH2Br OCH2CH3 CH3CHCH2 [α]D = + 23.5°