* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter Seven - U of L Class Index
Kinetic isotope effect wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Elias James Corey wikipedia , lookup
Discodermolide wikipedia , lookup
Hydroformylation wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Marcus theory wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Stille reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Ene reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
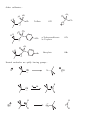
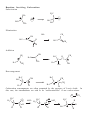
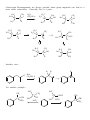
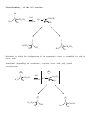
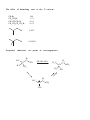
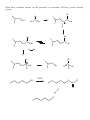
Nucleophilic Substitution Reactions These reactions are polar and usually involve negatively charged nucleophiles. X: Br X Br The organic substrates for such reactions are usualy alcohols of alkyl halides. Electrophilic centers exist by virtue of the electronegativity difference between carbon and leaving group bond. The Leaving Group Generally, good leaving groups are WEAK BASES. X Br X: Br All of the halides are good leaving groups except fluoride. Hydroxide is very basic, and a woeful leaving group. X OH X: OH One solution… O O Cl S N S OH CH 3 X: O A su lfo n at e est er . "Mesyla te - OMs" O O S CH 3 CH3 O Mesyl Ch lo r id e O O O X O S O CH 3 Other sulfonates… O O H H S S CF3 OTf Tr iflat e O O H S CH 3 O O H CF3 O O H O O H p -To lu en esu lfo n at e o r To sy lat e OTs O S Br O OBs Br osy lat e Neutral molecules are spiffy leaving groups… X: X OH OH H H Br OH O H H Br O H Br H O H The Nucleophile… Good nucleophiles are usually basic, or polarizable. Rules to Remember… 1) A negatively charged species is usually more nucleophilic than its conjugate base. 2) For a given atom and charge, nucleophilicity parallels basicity. O H 3 CH 2 C H O O O C O H3 C 3) Within a group of the periodic table, bigger is better in terms of nucleophilic character of the attacking atom. 4) Steric effects can effect nucleophilicity… H 3 CH 2 C O H3 C H3 C C O N H3 C Classify your substrate! Structural features determine whether certain reactions will take place which mechanism operates. H CH 3 CH3 CH2 OH CH 3 CH 3 CH 2 Cl H3 C H 3 CH 2 C CH2 Cl CH 3 Br CH2 OH CH 3 H2 C H2 C C OH H3 C CH 3 Br H3 C C C H H2 C OH C H H3 C H CH 2 CH 2 Cl C Br Br OH H2 C C H Alkyl halides mechanisms: (and many alcohols) undergo substitution by t w o Sn 1 and Sn 2 . The Sn 1 mechanism involves the formation of a carbocation intermediate in the ratedetermining step. 3°, benzylic and allylic substrates undergo Sn 1 reaction because they form relatively stable carbocations. 1° substrates undergo Sn2 reaction because they are sterically uncluttered. 2° substrates undergo both reaction types, but sluggishly. The Sn 1 Reaction H3 C H3 C H2 O C H3 C H3 C Br C OH + HBr H3 C H3 C Rea ct ion s in wh ich t h e solv en t p a r tici p at es as a n u celo p h ile ar e called solv oly sis r ea ct io n s. The Mechanism… H3 C C CH3 Br H3 C H3 C C H3 C H H3 C Br CH3 H C H3 C H3 C H Br O H H3 C C H3 C H3 C The reaction profile… O OH + HBr Since the SN1 reaction involves carbocations that are formed in the RDS, then only those substrates that form relatively stable carbocations will undergo this reaction. What makes for a stabilized H2 C CH 2 Cl H 2C H 2C carbocation? Br CH 2 C H 2C C H H3 C H H2 C O H3 C CH 2 O Cl Alkyl carbocations bonds. are stabilized by hyperconjugation to adjacent 3° 1° H H C H C H H H C H C H C H H H C H H H Vinyl (and aryl) carbocations cannot be stabilized by resonance. H Cl C H H C C H C H = H H H So, the order of stability of carbocations is: Benzyl = allyl > 3° > 2° > 1° > CH3 + > vinyl, aryl C C H C-H The Hammond Postulate – a useful and oft used tool to rationalize relative rates of reactions. the “The structure of a transition state tends to resemble that species to which it is closer in energy.” For endothermic processes… For exothermic processes… the TS is closer in energy to the p r o d u c t s and therefore said to be "product-like" i n structure. As a result, we can i n f e r something about the energy of t h e transition state from the stability of t h e product. H 3C CH 3 C H3 C H 3C Br C H 3C Nu : H 3C H3 C Br CH 3 the TS is closer in energy to the reactants a n d therefore said to be "reactant-like" in s t r u c t u r e . As a result, we can infer something about t h e energy of the transition state from the stability of the r e a c t a n t . C H3 C H3 C Nu CH 3 C H H Br C H3C C Br H Nu H H H Nu : Therefore, when comparing reactions that involve carbocation intermediates, the one involving the more stable carbocation intermediate will be… Reactions Involving Carbocations Substitution H3 C CH3 C H3 C C Nu Nu H3 C H3 C CH3 Elimination OH2 CH 3 H3 C CH3 H C C H C H3C CH 2 H Addition CH 3 CH 3 H2 C H3 C C C C CH3 H3 C H2 C CH3 C H3 C CH 3 CH 3 CH 3 Rearrangement H3 C H3 C C H3 C H3 C CH3 C C H3 C H CH3 C CH 3 H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. Cl H3 C H3 C C H Cl C H H AlCl 3 H3 C H3 C C H Cl C AlCl 3 H3 C H H C H3 C AlCl 3 H H C H Carbocation Rearrangements are always possible when group migration can lead to a more stable carbocation. Generally, this is a pain… H3 C H3 C C C H3 C H3 C H3 C C CH3 I H 2 O, CH3 OH, ∆ H3 C H3 C C H C H3 C H3 C H3 C H3 C C CH3 I H H3 C C H C C H3 C CH3 H CH3 H3 C CH 3 C C HO CH 3 C H3 C H H3 C H3 C C H3 C H3 C C CH3 OH CH3 H CH3 OH H3 C H3 C C H HO C CH3 H CH3 Another case… H Cl H H H 2 O, CH3 OH, ∆ Yet another example… H3 C CH 3 C H3 C C CH 3 CH 3 H3 C CH3 CH CH2 Cl CH 3 Cl Al Cl 3 H3 C AlCl3 C CH 3 CH 3 Stereochemistry of the SN 1 reaction Br C H3 C NaI CH2 CH 3 Ph Acet o n e H 3 CH 2 C Ph H3 C C CH2 CH3 Ph I I C C CH 3 H3 C CH2 CH 3 Ph Reactions in which the configuration of an asymmetric center is scrambled are said to occur with Sometimes (depending on conditions), reactions occur with only partial racemization… Br - Br C H3 C NaI CH2 CH 3 Ph Ac et o n e H 3 CH 2 C Ph H3 C C CH2 CH3 Ph I I C C CH 3 H3 C CH2 CH 3 Ph The SN 2 Reaction Best for 1° subtrates. And methyl, of course. H3 C I Br CH 3 I Br The mechanism involves a single elementary process that is bimolecular. Stereochemistry of the SN 2 Reaction The collision in this reaction must occur such that the nucleophile attacks the back side of the C-LG bond. H I H H C I Br H C Br H H In chiral substrates, this means that the reaction occurs exclusively H3 C S H3 C H 3 CH2 C C Br H inversion of the absolute stereochemistry. reaction. H3 C S with CH3 CH2 CH3 C Br H This is an example of a stereospecific Stereospecific: stereoisomeric A reaction in which the mechanism dictates that different reactants give different stereoisomeric products. Why? H H H C I Br structure C Br H H Substrate I H and SN 2 Relat iv e r at e o f r eac tion CH3 Br 100 CH3 CH 2 Br 1 .3 1 CH3 CH 2 CH 2 Br 0 .8 1 Su b st r at e H3 C CHBr 0 .0 1 5 H3 C CH3 H3 C C Br 0 .0 0 4 CH3 So, the order of reactivity for SN 2 is: CH 3 > 1° > 2° >>3° > vinyl, aryl The effect of branching 100 1 .3 1 0 .8 1 0 .5 2 CH3 Br CH3 CH 2 Br CH3 CH 2 CH 2 Br CH3 CH 2 CH 2 CH 2 Br Neopentyl next to the 1° carbon… Br 0 .0 52 Br 0 .0 00 0 1 substrates H2 C H3 C C H3 C are prone to rearrangement… OTs CH 3 OH/ H2 O H2 C H3 C C CH 3 H3 C H2 C H3 C C CH 3 CH 3 OH CH 3 Solvent dependence of SN 1 and SN 2 reactions… SN 1 reactions are facilitated by using polar, protic solvents – solvents that are capable of stabilizing ionic intermediates. Examples: SN 2 reactions are facilitated by using polar, aprotic solvents – solvents that will not mask the nucleophile with a large solvation sphere. Examples: Making alkyl iodides – the Finkelstein Reaction Aceton e R Br R NaI I Pitfalls in the synthesis of amines… CH3 CH 2 Br :NH 3 CH3 CH 2 NH 2 :NH 3 CH3 CH 2 NH 3 Br CH3 CH 2 NH 2 CH3 CH 2 NH 3 Br NH 4 Br Br CH3 CH 2 NH 2 CH 2 CH 3 CH3 CH 2 Br OK for preparing 3° amines from 2° amines… Cl NH HO + N Cl 70 % and quaternary ammonium compounds. CH 3 N CH 3 CH 3 I N CH 3 H3 C CH 3 Preparing alkyl halides from alcohols – SN 1 or SN 2. 3° alcohols react via SN1… H 3C H3 C H C H3 C Br H3 C H 3C OH H C H3 C H3 C O CH 3 CH 3 C H O H H3 C H3 C C H3 C Br H 1° alcohols by SN 2 H 2 SO 4 , NaBr CH3 CH 2 CH 2 CH 2 OH CH3 CH 2 CH 2 CH 2 Br sometimes facilitated by Lewis acids Zn Cl 2 , HCl CH 3 CH2 CH2 CH2 OH CH 3 CH2 CH2 CH2 Cl H H3 CH2 CH 2 CH 2 C O Zn Cl 2 Some other reagents… OH OH PBr 3 SOCl2 Br 60% Cl 78% Both these reactions operate on the principal of converting OH into a better leaving group… Br Br P P OH Br O Br Br H Br OR P P O O OR H Br Br OR OR P P O Br OR H O OR H SOCl 2 O OH Cl S O Cl Ethers in substitution Making them… reactions: The Williamson ether synthesis O O H O Br NaH, DMF This is just an SN 2 reaction, so the substrate dependence for SN 2 applies. and breaking them… HI O I H I I O O H H TMSI, Trimethylsilyl Iodide I Me 3 Si H3 C O I H3 C SiMe 3 O CH 3 SiMe 3 O Epoxides are good substrates for substitution reactions With good nucleophiles and neutral conditions, it is an SN 2 reaction, and occurs w i t h inversion at the reaction center… O O Nu Nu : H H N3 NaN 3 , Et OH, H 2 O H OH OH H O H N N N3 H N N N H N H O O H H OH H In SN 2 ring-opening of epoxides, the nucleophile tends to attack the least hindered end of the epoxide. O HO Ph SH, Ph S H3 C 89 % H3 C H H SPh Under acidic conditions, the nucleophile adds to the most substituted end. This is a little complicated. This resembles, but is not, an SN 1 process. O H3 C H3 C CH3 OH, H3 O+ H3 C H3 C OH H 3 CO The intermediate – carbocation, or not H O O O CH2 H3 C H3 C H H = CH2 H3 C H3 C CH2 H3 C H3 C H H H O O O = H3 C H3 C CH2 CH2 H3 C H3 C CH2 H3 C H3 C Attack occurs at that carbon that is best able to stabilize positive charge development. H O -H+ CH 2 H3 C H3 C H3 C H3 C H3 CO O H CH 3 OH A Comparison of SN 1 and SN 2 SN 1 Mechanism Kinetics Substrate dependence SN 2 2 or more steps involving a 1 step bimolecular process carbocation intermediate first order in substrate second order over all, first only order in substrate and nucleophile Those substrates capable Those substrates that are of forming stabilized sterically unhindered at carbocations the substitution site. Stereochemistry Occurs with racemization or partial racemization at the reaction center Occurs exclusively with inversion at the reaction center Importance o f nucleophile Not involved in RDS, but better if less basic form of nucleophile is employed Leaves in RDS therefore it is important Polar protic solvents are best. Involved in RDS therefore reactivity is important Importance group Solvent of leaving Leaves in RDS therefore it is important Polar aprotic solvents are best.