* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Key Strengths and Weaknesses of 3D Models Advantages
Survey
Document related concepts
Biochemical switches in the cell cycle wikipedia , lookup
Cell membrane wikipedia , lookup
Endomembrane system wikipedia , lookup
Cell encapsulation wikipedia , lookup
Signal transduction wikipedia , lookup
Programmed cell death wikipedia , lookup
Cell growth wikipedia , lookup
Cytokinesis wikipedia , lookup
Tissue engineering wikipedia , lookup
Cellular differentiation wikipedia , lookup
Hyaluronic acid wikipedia , lookup
Cell culture wikipedia , lookup
List of types of proteins wikipedia , lookup
Transcript
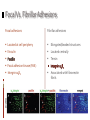
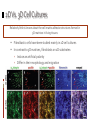
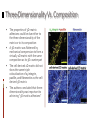
Kenneth M. Yamada & Edna Cukierman Introduction 2D cell cultures Animal models 2D cell cultures • Studies in standard cell culture have produced many important conceptual advances • Nevertheless, cells grown on flat 2D tissue culture substrates can differ considerably from physiological 3D environments Animal models • Animal models provide definitive tests of the importance of specific molecules and processes • There can, however, be puzzling discrepancies between conclusions from gene ablation studies In Vitro 3D Models In vitro 3D tissue models provide a third approach that bridges the gap between traditional cell culture and animal models 5 types of In Vitro 3D Models: Organotypic explant cultures Stationary or rotating microcarrier cultures Micromass cultures Free cells in rotating vessel Gel-based cultures In Vitro 3D Models The morphologies of fibroblasts, including cytoskeletal organization and types of cell adhesions, are also more similar to their in vivo behavior Planar fibronectin Cell-derived 3D matrix In Vitro 3D Models 3D models are closer to physiological conditions than routine 2D cell culture, but they also have their weaknesses, such as their lack of vasculature and ability to progress Key Strengths and Weaknesses of 3D Models Advantages • Cell morphology and signaling are often more physiological than routine 2D cell culture • Permit rapid experimental manipulations and testing of hypotheses • Permit much better real-time and/or fixed imaging by microscopy than in animals Disadvantages • Vary in their ability to mimic in vivo tissue conditions • Currently lack vasculature and normal transport of small molecules • Generally mimic static or short-term conditions, whereas in vivo systems often progress In Vitro 3D Models Comparisons of 2D and 3D models reveal that the latter are better, but not exact, models of in vivo tissues 3D-Dependent Cell Behavior and Signaling Function 2D versus 3D Regulatory Mechanisms Cell Shape Loss of epithelial cell polarity and altered epithelial and fibroblast shape in 2D Growth factor receptors and pathways; cell-adhesion signals Gene Expression Cells in 2D versus 3D often have different patterns of gene expression ECM, hormones, and adhesion molecules Growth 3D matrix-dependent regulation of cell growth Adhesion and growth factor-related pathways, apoptotic genes Morphogenesis 3D matrix-induced vessel sprouting and gland branching ECM, adhesion, growth factorrelated pathways, apoptotic genes Motility Altered single and collective cell motility patterns in 3D matrices ECM and its regulators; adhesions and growth factor-related pathways Differentiation 3D matrix-induced cell differentiation ECM and growth factors; motor molecules Model Choice Affects Outcome In any 3D model system, the specific cellular and matrix microenvironment provided to cells can substantially influence experimental outcome Model Choice Affects Outcome Physical properties such as matrix stiffness and cell polarity in 3D models can also play surprisingly important roles Matrix stiffness Stiffness (compliance) of the extracellular matrix can be sensed by cells through bidirectional interaction between the cell surface integrin receptors and the contractile cytoskeleton Cell and tissue polarity Polarity in vivo depends both on the cell type and the cellular microenvironment, and it is important for tissue organization and direction secretion of products Matrix Stiffness The stiffness of a matrix can affect the distribution of cell surface integrin receptors, types of cell adhesions, cytoskeletal structures, cell proliferation, and other factors Cell and Tissue Polarity Cells explanted into routine tissue cultures often flatten and lose differentiation markers; they generally regain their correct polarity when placed back in 3D culture conditions Edna Cukierman, et al. Introduction Our current knowledge about the roles of cell-matrix adhesions in cell adhesion, migration, signaling, and cytoskeletal function is derived primarily from studies on planar 2D tissue culture substrates There are two types of cell-matrix adhesions: Focal adhesions Fibrillar adhesions Focal Vs. Fibrillar Adhesions Focal adhesions Fibrillar adhesions Located at cell periphery Elongated/beaded structures Vinculin Located centrally Tensin Focal adhesion kinase (FAK) Integrin αVβ3 Associated with fibronectin fibrils 2D Vs. 3D Cell Cultures Relatively little is known about the cell-matrix adhesive structures formed in 3D matrices in living tissues Fibroblastic cells have been studied mainly in 2D cell cultures In contrast to 3D matrices, fibroblasts on 2D substrates: Induce an artificial polarity Differ in their morphology and migration “3D-Matrix Adhesions” FOCAL ADHESIONS Located at cell periphery Vinculin FIBRILLAR ADHESIONS Elongated/beaded structures Located centrally Tensin Focal adhesion kinase (FAK) Integrin αVβ3 3D-MATRIX ADHESIONS Associated with fibronectin fibrils Associated with fibronectin fibrils Cell Response to 3D-Matrix Adhesions #1: CELL ATTACHMENT The cell-derived 3D matrix was more effective (by a factor of over 6) in mediating cell adhesion than was 2D substrates Cell Response to 3D-Matrix Adhesions #2: CELL MORPHOLOGY Fibroblasts in 3D matrices achieved their final, elongated morphology by 5 hours. Fibroblasts on cell-derived 2D matrices eventually achieved the same morphology, but it took over 18 hours. Cell Response to 3D-Matrix Adhesions #3: MIGRATION Cells showed enhanced migration rates (by a factor of 1.5) in 3D matrices relative to individual protein-coated surfaces. Cell Response to 3D-Matrix Adhesions #4: PROLIFERATION Proliferation rates of fibroblasts in the cell-derived 3D matrices were more than double fibronectin-coated 2D substrates. Three-Dimensionality Vs. Composition The properties of 3D-matrix adhesions could be due either to the three-dimensionality of the matrix or to its composition A 3D-matrix was flattened by mechanical compression to form a virtually 2D matrix with the same composition as its 3D counterpart The cell-derived 2D matrix did not show the same triple colocalization of α5 integrin, paxillin, and fibronectin as the cellderived 3D matrix The authors concluded that threedimensionality was important to achieving “3D-matrix adhesions” Conclusions Cukierman et al. speculated that focal and fibrillar adhesions studied in vitro represent exaggerated precursors of in vivo 3D-matrix adhesions Fibroblasts initially require culture for days at high cell density to generate 3D matrices and evolve 3D-matrix adhesions, yet when added back to cell-free 3D matrices, they begin regenerating matrix adhesions within 5 minutes Requirements for 3D-matrix adhesions include three-dimensionality, integrin α5β1, fibronectin, other matrix components, and pliability (data not discussed in this presentation)