* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Amino acids
RNA polymerase II holoenzyme wikipedia , lookup
Eukaryotic transcription wikipedia , lookup
Interactome wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Polyadenylation wikipedia , lookup
Transcriptional regulation wikipedia , lookup
RNA silencing wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Western blot wikipedia , lookup
Point mutation wikipedia , lookup
Silencer (genetics) wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Genetic code wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Protein structure prediction wikipedia , lookup
Gene expression wikipedia , lookup
Epitranscriptome wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Proteolysis wikipedia , lookup
Biosynthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
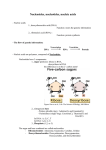
Proteins - Amino acids - Structure - Protein domains - Physicochemical properties - Folding and chaperones - Connective tissue proteins AMINO ACIDS Amino acids: general structure Nonpolar alifatic Aromatic Polar uncharged Positively charged Negatively charged Proteins consist of amino acids and have carboxyland amino-terminal ends STRUCTURE OF PROTEINS Primary structure: sequence of amino acids VLSPADKTNVKAAWGKVGAHAGEYGAEALERMFLS FPTTKTYFPHFDLSHGSAQVKGHGKKVADALTNAV AHVDDMPNALSALSDLHAHKLRVDPVNFKLLSHCLL VTLAAHLPAEFTPAVHASLDKFLASVSTVLTSKYR Amino acid alignment of same proteins from different organisms Protein globule is the functional structure (tertiary structure) Secondary structure: alfa-helix Secondary structure: beta-sheet Tertiary structure: position of all protein atoms in space Quaternary structure: association of two or more molecules Quaternary structure: association of two or more molecules hemoglobin DOMAINS: DISTINCT COMPACT PARTS IN PROTEIN STRUCTURE Domains of pyruvate kinase Domain 1 Domain 2 (catalytic) Domain 3 (regulatory) FOLDING AND CHAPERONES There is no universal way for protein folding (each protein follows its own folding pathway) But there is one general rule: - Hydrophobic amino acids form hydrophobic core inside protein globule Forces that fold protein Folding: example of stages Chaperones Chaperones are the proteins that help other proteins to fold properly. Functions of chaperones: 1. To prevent aggregation of newly synthesized proteins. 2. To assist in folding of unfolded proteins (due to changes of temperature, pH) Prions: “infective” proteins CONNECTIVE TISSUE PROTEINS Keratin Keratin of hairs Keratin of hairs Hydrophobic interactions stabilises supercoiled alfa-helix in keratin Disulfide bonds FIBRILLAR PROTEINS: - COLLAGEN Collagen Collagen triple helix: Gli – Х – Pro Vitamin C is necessary for hydroxylation of proline in collagen Lysine and hydroxy-lysine make cross-bridges in collagen to hold helices together Conjugated proteins - Classification - Functions HEMOPROTEINS: HEMOGLOBIN AND MYOGLOBIN Heme = protoporphyrin + Fe2+ Physiological functions • Myoglobin stores oxygen in muscles. It releases oxygen only when oxygen concentration is very low (hypoxia) • Hemoglobin transfers oxygen from lungs to tissues. It releases oxygen when oxygen concentration is in physiological range. Complex between ligand and protein О2 is bound to heme group Oxygen saturation curves for myoglobin (hyperbolic) and hemoglobin (sigmoidal): different ability to transfer oxygen Lungs: both proteins are saturated with oxygen. Tissues: - myoglobin is 90% saturated (gives back only 10%) - hemoglobin is 50% saturated (gives back 50%) COOPERATIVITY OF OXYGEN BINDING Myoglobin consists of single polypeptide chain, while functional hemoglobin molecule contains four polypeptide chains (which are very similar to myoglobin ones) Myoglobin (single chain) Hemoglobin (one subunit) Hemoglobin (four subunits) Oxygen binding leads to structural (conformational) changes in binding site, which are transmitted to neighboring subunits through contacts between subunits Binding of oxygen molecule by the first subunit enhances oxygen binding to other subunits This feature is called cooperativity 2,3-BISPHOSPHOGLYCERATE AND COOPERATIVITY 2,3-BPG is produced from one of the glycolysis intermediates The presence of 2,3BPG indicates sites of high energy production and, therefore, oxygen demand 2,3-BPG binds to the central cavity of hemoglobin molecule Effects of 2,3-BPG: • 2,3-BPG weakens oxygen binding. Therefore: • High 2,3-BPG in tissue → weak oxygen binding → better oxygen supply This plays role in adaptation to highlands and intensive work HEMOGLOBIN ISOFORMS: - ADULT - FETAL Protein isoforms Synthesis of different isoforms during embryonic development Fetal vs. adult hemoglobin PHYSIOLOGICAL REGULATION OF OXYGEN BINDING: EFFECTS OF PH AND CO2 Effects of pH and CO2 Tissues with high energy demand (high glycolysis): - high CO2 -low pH Nucleic acids - Nitrogen bases - Nucleotides - DNA and RNA DNA and RNA contain nitrogen bases Nucleotides vs. nucleosides How is this nucleoside / nucleotide called? There are some other nitrogen bases • Alkaloids • Products of nitrogen bases breakdown Some bases are not included in nucleic acids structure: alkaloids H caffeine H theophilline theobromine Nitrogen bases which are formed during metabolism from adenine and guanine hypoxanthine xanthine Uric acid Elevation of uric acid causes gout • Uric acid forms insoluble salts • Causes: increased production OR decreased excretion DNA contains minor (modified) bases: regulation of gene expression by methylation RNA contains minor bases: recognition, stability, regulation mRNA tRNA FUNCTIONS ATP as energy carrier high energy bond (macroergic bond, high energy phosphate) = good leaving group, high transfer potential (correct meaning) Nucleotide moieties of coenzymes: coenzyme A Nucleotide moieties of coenzymes: NAD Nucleotide moieties of coenzymes: FAD Regulatory functions of cyclic nucleotides cAMP has positive effect on energy-production pathways (glycolysis). One of the mechanisms of caffeine action is inhibition of cAMP breakdown. This leads to prolongation of cAMP stimulatory action. cGMP mediates muscle relaxation caused by NO. Effect of nitroglycerin on heart muscle relaxation is mediated by cGMP formation. Summary • Nucleotides consist of nitrogen base (A,T,G,C,U) + (deoxy)ribose + phosphate • Nucleotides are the building blocks of nucleic acids (DNA, RNA) • Nucleotides perform signaling functions (cAMP, cGMP) • Nucleotides play role in catalysis (parts of coenzymes). • Nucleotides are energy carriers (ATP, GTP) NUCLEIC ACIDS: DNA Nucleotides (in DNA or RNA) are joined by phosphodiester bonds Conventional order of sequence description is from 5`- to 3`- end: 5` ATG 3` DNA is a double helix DNA STRUCTURE AND DRUGS Intercalation – insertion between planar bases of double helix Intercalating drugs: bacteriostatics proflavin Intercalating drugs for cancer cisplatin Mechanism of intercalation NUCLEIC ACIDS: RNA Differences between RNA and DNA: pentose Lack of OH-group makes DNA more chemically stable: DNA is more suitable for storing genetic information RNA functions in protein synthesis: ribosomal RNA perform catalytic functions RNA is the major structural component of ribosomal subunits 30S rRNA (brown) + proteins (blue): rRNA: 50S RNA functions in protein synthesis: transfer RNA (tRNA) Real conformation in 3D RNA functions in protein synthesis: messenger RNA (mRNA) Other functions of RNA: • Catalysis (ribozymes): ribosome, RNAse P, hammerhead ribozyme. • Regulation of gene expression (RNA interference): small interfering RNAs RNA as catalyst: ribozymes RNase P plays role in: - tRNA processing - regulation of transcription