* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download CHAPTER 9 CELLULAR RESPIRATION Life is Work Types of
Fatty acid synthesis wikipedia , lookup
Signal transduction wikipedia , lookup
Biochemical cascade wikipedia , lookup
Butyric acid wikipedia , lookup
Metalloprotein wikipedia , lookup
Mitochondrion wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Phosphorylation wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Photosynthesis wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Electron transport chain wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Biochemistry wikipedia , lookup
Citric acid cycle wikipedia , lookup
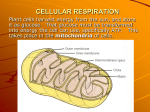
10/25/2010 Life is Work CHAPTER 9 CELLULAR RESPIRATION y Living cells require transfusions of energy from outside sources to perform their many tasks: y Chemical work y Transport work y Mechanical work y Energy stored in the organic molecules of food ultimately comes from the sun Energy flows into an ecosystem as sunlight and leaves as heat y In contrast, chemical elements essential to life are recycled y Photosynthesis generates oxygen and organic molecules y Respiration breaks this fuel down, generating ATP y In this chapter, we will focus on the key pathways of aerobic respiration: y Glycolysis y Citric acid cycle y Oxidative phosphorylation Catabolic pathways = oxidizing fuels y Catabolic pathways = metabolic pathways that release stored energy by breaking down complex molecules Types of cellular respiration y Aerobic Respiration (‘cellular respiration’): y Oxygen is consumed as a reactant along with the organic fuel y Compounds that can participate in exergonic reactions can act as fuels y With the help of enzymes, a cell can systematically degrade macromolecules that are rich in potential energy y Energy taken out of chemical storage can be used to do work y Cells of most eukaryotes and prokaryotes y Anaerobic Respiration: y Example: fermentation y Degradation of organic fuel without the use of oxygen y Cells of some prokaryotes and some eukaryotes 1 10/25/2010 The Stages of Cellular Respiration Overall process: Organic compound Oxygen Carbon dioxide y Glycolysis: Water Energy y y y y Technically not part of cellular respiration Catabolic pathway that occurs in cytosol Breaks down glucose to two smaller molecules Dehydrogenases transfer e- from NAD+ to NADH y We will learn the steps of cellular respiration by tracking the degradation of the sugar glucose: C6H12O6 + 6 O2 y Citric Acid Cycle y Takes p place in the mitochondria ((matrix)) eukaryots y y Takes place in cytosol in prokaryots y Dehydrogenases transfer e- from NAD+ to NADH 6 CO2 + 6 H2O + Energy (ATP, heat) y Oxidative phosphorylation: electron transport and chemiosmosis y Glucose is the fuel that cells most often use y Accepts e- from NADH (H+ + e-) y Pass through many molecules and the end it is combined with oxygen to y Exergonic process where ∆G = -686 Kcal/mol y The energy released makes ATP form water REDOX REACTIONS y Loss of electrons = oxidation y Gain of electrons = reduction (reduce + charge) y Reducing agent = electron donor y Oxidizing agent = electron acceptor NAD+ and NADH y Dehydrogenase removes a pair of hydrogen atoms (2eand 2p+) from the substrate and oxidizes it y NAD+ is an electron carrier (a coenzyme) y It acts as an oxidizing agent during respiration y The enzyme delivers 2 e- and 1p+ to the coenzyme y Oxidation = loss of electrons (NAD+) y Oxidizing agent = takes away electrons y The other p+ (H+) is released to the surrounding H+ 2H solution H 2 10/25/2010 GLYCOLYSIS y Oxidizes glucose to pyruvate y Glycolysis Overall: y Glucose (6 C sugar) is split to two 3 C sugars y Pyruvate y Two phases: y Energy investment y Energy payoff y Oxygen is not necessary y CO2 is not released y NET ATP YIELD = 2 ATP + 2 NADH 3 10/25/2010 Citric Acid Cycle y Glycolysis releases less than a 1/4 of the chemical energy stored in glucose y Most M t off the th energy remains i in i the th two t molecules l l off pyruvate y If oxygen is available, pyruvate enters a mitochondrion (eukaryotes) y Active transport y Acetyl CoA is very unstable (potential energy? Ender/exergonic?) y Citric Acid Cycle = Krebs Cycle (1930s) y Oxidizes organic fuel derived from pyruvate y The cycle generates: y 1 ATP y 3 NADH y 1 FADH2 y 1 NADH (junction) 4 10/25/2010 Oxidative phosphorylation and chemiosmosis y So far, glycolysis and the citric acid cycle have produced only 4 ATP molecules per glucose molecule (substrate-level phosphorylation) y 2 ATP from glycolysis + 2 NADH y 2 ATP from citric acid cycle + 4 NADH + FADH2 y NADH and FADH2 molecules account for most of the energy extracted from the glucose molecule Electron Transport y The electron transport chain is a collection of molecules embedded in the inner membrane of the mitochondrion (eukaryotic cells) y Most components are proteins, which exist in complexes (I – IV) y Other non protein components are essential for the functioning of enzymes, they are called prosthetic groups y Electrons from NADH are transferred to first molecule: y flavoprotein (flavin mononucleotide) y Iron-sulfer protein y Coenzyme y Q y Cytochromes (heme prosthetic groups) y Oxygen (very electronegative) Why not release all the energy in just one step? + energy 2 H2 O2 2 H2 O Chemiosmosis: energy-coupling mechanism y Many copies of ATP synthase populate the inner membrane of the mitochondrion y It uses energy of an existing ion gradient to power ATP y synthesis y In the mitochondrial inner membrane there is a pump that creates a difference in H+ concentration between the matrix and the intermembrane space y Also considered a difference in pH 5 10/25/2010 Chemiosmosis y Process in which energy stored in the form of a hydrogen ion gradient across a membrane is used to drive cellular work, such as the synthesis of ATP y ATP synthase is a multisubunit complex with four main parts: y Stator y Rotor y Internal Rod y Catalytic knob y To read: Fig. 9.15 Inquiry How do we maintain the H+ gradient? y That is the function of the electron transport chain y The exergonic flow of electrons from NADH (or FADH2) down to oxygen is used to pump H+ across the membrane y ATP synthase is the only route through the membrane (for H+) y Certain members of the electron transport chain accept and release protons (H+) y The H+ gradient that results is referred to as protonmotive force Peter Mitchell y Chemiosmosis is an energy-coupling mechanism that uses energy stored in the form of an H+ gradient across a membrane to drive cellular work y Examples: was awarded the Nobel Prize in 1978 for originally proposing the chemiosmotic model y Chloroplasts: use it to generate ATP during phothosynthesis y Prokaryotes: use it to make ATP and also to rotate their flagella and pump nutrients and waste across the membrane 6 10/25/2010 … ATP production? y During respiration, most energy flows: y Glucose > NADH > ET Chain > Proton MF > ATP Electron shuttles span membrane CYTOSOL or 2 FADH2 2 NADH Glycolysis y 1 NADH = 1 pair electrons = pumps 10 H+ MITOCHONDRION 2 NADH Glucose 6 NADH 2 NADH 2 Pyruvate 2 FADH2 Citric acid cycle 2 Acetyl CoA Oxidative phosphorylation: electron transport and chemiosmosis y 3 – 4 H+ = 1 ATP y 1 NADH = 2.5 – 3.3 ATP = ~ 3 y 1 FADH2 = 1.5 – 2 ATP = ~ 2 + 2 ATP + 2 ATP Maximum per glucose: + about 32 or 34 ATP About 36 or 38 ATP y READ: p. 176-177 Reasons that affect ATP yield Fermentation and Anaerobic Respiration Anaerobic Respiration y The estimated ATP yield from aerobic respiration is y Takes place in certain prokaryotic organisms contingent on an adequate supply of oxygen to the cell (environments without O2) y These organisms have electron transport chains, but y However, However cells can oxidize organic fuel and generate ATP without the use of oxygen: y Anaerobic respiration y Fermentation Fermentation y It harvests chemical energy without using oxygen or any electron transport chain y Glycolysis oxidizes glucose to two molecules of the end product is a different molecule y Ex: SO42y H2S is a by-product (instead of water) Fermentation y There must be a sufficient supply of NAD+ y Electrons can be transferred to pyruvate instead pyruvate (oxidizing agent NAD+) y Glycolysis is exergonic and makes ATP by substratelevel phosphorylation y Fermentation expands glycolysis so that it continuously y Types of fermentation (end product): y Alcohol Fermentation y Lactic Acid Fermentation generates ATP y How?? 7 10/25/2010 Alcohol fermentation y Piruvate is converted to ethanol in two steps: y Release of CO2 = acetaldehyde y Acetaldehyde is reduced d db by NADH = ethanol y Ex: yeast: brewing, winemaking and baking Lactic Acid Fermentation y Pyruvate is reduced directly by NADH to form lactate y No release of CO2 y Used in the dairy insudstry to make yogurt and cheese y Human muscles (when O2 is scarce) Fermentation and Aerobic Respiration Compared Obligate / Facultative y Obligate Anaerobes: y Some organisms carry out only fermentation or anaerobic y Both processes use glycolysis to oxidize glucose and other respiration y They cannot survive in the presence of oxygen y The processes have different final electron acceptors: an y Facultative Anaerobes: y Yeasts and many bacteria can make enough ATP to survive using either fermentation or respiration organic fuels to pyruvate organic molecule (such as pyruvate or acetaldehyde) in fermentation and O2 in cellular respiration y Cellular respiration produces 38 ATP per glucose molecule; fermentation produces 2 ATP per glucose molecule y Our muscle cells behave as facultative anaerobes Fig. 9-19 Glucose y Obligate anaerobes carry out fermentation or anaerobic CYTOSOL respiration and cannot survive in the presence of O2 y Yeast and many bacteria are facultative anaerobes, Glycolysis Pyruvate No O2 present: Fermentation meaning that they can survive using either fermentation or cellular respiration O2 present: Aerobic cellular respiration y In a facultative anaerobe, pyruvate is a fork in the metabolic road that leads to two alternative catabolic routes MITOCHONDRION Ethanol or lactate Acetyl CoA Citric acid cycle 8 10/25/2010 The Evolutionary Significance of Glycolysis y Glycolysis occurs in nearly all organisms y Oldest fossil bacteria date to ~3.5 bya y But atmospheric O2 only started to accumulate ~ 2.7 2 7 bya y Glycolysis probably evolved in ancient prokaryotes before there was oxygen in the atmosphere Other Catabolic pathways y Free glucose molecules are not common in the diets of humans and other animals y Most our calories com from fats, proteins, sucrose and starch y All these molecules can be used by cellular respiration to make ATP y Carbohydrates: y Starch can be hydrolyzed y Glycogen can also be hydrolyzed y Proteins: y First they are digested (aa) y Aa can be used to build proteins or can be converted to intermediates of glycolysis and the Krebs cycle (d (deamination i ti + enzymes)) y Fats: y Digested to glycerol and fatty acids y Glycerol = glyceraldehyde-3-phosphate (glycolysis) y Fatty acids via Beta oxidation = 2 C fragments = enter Krebs cycle as Acetyl CoA (NADH and FADH2 also generated) Anabolic Pathways y Cells need substance as well as energy y Food must also provide the atoms necessary to make new molecules Example y Dihydroxyacetone phosphate (intermediate compound generated during glycolysis) can be converted to one of the precursors of fat y Intermediates of glycolysis and the citric acid cycle can be diverted into anabolic pathways as precursors for other molecules y So, when we eat more food than we need, we store fat even if our diet is fat-free. y Humans can make about half of the 20 essential aa, the rest come from diet y Metabolism is remarkably versatile and adaptable 9 10/25/2010 Feedback Mechanisms y Feedback inhibition is the most common control mechanism for anabolic pathways Phosphofructokinase y Important switch in metabolic control y The cell can speed up or slow down the entire catabolic process by controlling the first step of the glycolytic pathway y An end product of the anabolic pathway inhibits the enzyme that catalyzes an early step in the pathway y Catabolism is also regulated through positive and negative feedback y Phosphofructokinase is an allosteric enzyme with receptor sites for specific inhibitors and activators: y Inhibited by ATP y Stimulated by AMP y Also sensitive to citrate (1st product Citric Acid Cycle) 10