* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download EAS Friedel-Crafts Alkylation
Kinetic resolution wikipedia , lookup
Elias James Corey wikipedia , lookup
Homoaromaticity wikipedia , lookup
Discodermolide wikipedia , lookup
Marcus theory wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Stille reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Aromaticity wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Vinylcyclopropane rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Aromatization wikipedia , lookup
Hydroformylation wikipedia , lookup
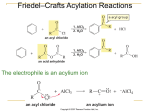
Chem 345 – Organic Reactions Chapter 16 Prepared by José Laboy, MS http: www.chem.wisc.edu/areas /clc (Resource page) Electrophilic Aromatic Substitution #5: Friedel Crafts Alkylation Reaction Electrophile H CH C Cl Mechanism 3 CH2CH3 CH3CH2Cl / AlCl3 (0.1 eq.) Cl Cl Al 2 Cl Cl Cl Cl Al Al Cl H3C CH2 + AlCl4 Cl Cl H 3CH2C H 3CH2C Cl Cl or CH2CH3 CH2CH3 H H H 2C AlCl3 + HCl CH 3 AlCl4 H CH2CH3 CH2CH3 This is a very useful reaction because you can make carbon-‐carbon bonds to aromatic compounds. Electrophiles that are primary carbocations are very reactive. Careful attention should be paid to side reactions that often occur when the carbocation electrophile can rearrange to a more stable one (see example below). Cl + AlCl3 Major Product < 5% H H H Cl Cl Al Cl Cl Cl Al Cl δ Cl Cl rearrangement 1,2-hydride shift C H 3C CH 3 2o carbocation has carbocation character The Friedel-‐Crafts reaction only requires catalytic amounts of the Lewis acid because it is recycled through the reaction. There is a major disadvantage of the alkylation reaction and that is over-‐ alkylation. This is due to the fact that the product of the reaction is activated toward EAS more so than the starting reagent. One way of avoiding over-‐alkylation is to have an excess of the aromatic reagent and adding the electrophile slowly. Another way of performing alkylation is by using an alkene with an acid catalyst or an alcohol with a Lewis acid catalyst (see below). In each case an electrophile is formed. CH 3 H 2C CH HF OH BF3