* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 10: Alkyl Halides
Kinetic resolution wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Marcus theory wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Elias James Corey wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Discodermolide wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
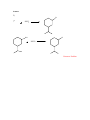
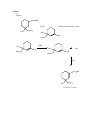
Alkyl Halides Alkyl Halides (RX)—compounds with a halogen bonded to a saturated carbon (sp3 hybridized carbon). Nomenclature Alkyl halides are named in the same way as alkanes. Treat the halogen as a substituent on the alkane parent chain. Example Cl 2-chloropentane Reactions to Know Synthesis of Alkyl Halides You learned in chapter 5 how to make an alkyl halide by a radical reaction between a halogen and an alkane in uv light. 1. From Alkenes by Allylic Bromination: Br H C C C NBS C C C Allylic Bromination with NBS is analogous to the radical reaction with an alkane, a halogen and uv light (Ch. 5). The NBS can be thought of as producing a Br radical. The Br radical removes a hydrogen, leaving an allylic radical and forming HBr. This allylic radical reacts with Br2 (which is formed from NBS reacting with the HBr formed from the removal of a hydrogen from the alkene) to give the product. H C C C Br C C HBr C Br2 Br C C C Br Why does the reaction only occur at the allylic position? Allylic radicals are more stable than alkyl or vinylic radicals: Stability of Radicals H R C R H H H R C R C C C C C C C Vinylic < methyl < primary R R H H < secondary < tertiary < allylic Allylic radicals are so stable because they are resonance stabilized. Because the allylic radical is more stable, it has a lower energy of activation and more collisions per unit of time result in a successful reaction at the allylic position. 2. From Alkenes (Chapters 6 & 7): C C + HBr H Br C C 3. From Alcohols (a): OH X HX C Reactivity of alcohols: 3º > 2º > 1º C You’ll learn the mechanism for this reaction in Ch. 11. 4. From Alcohols (b): OH Cl SOCl2 C 1º and 2º alcohols (PBr3 PCl3 and PCl5 halogenate similarly). C Pyridine Practice Problems 1) H2C NBS CHCH2CH2CH3 ? Answer 2) Cl ? SOCl2 Answer Reactions of Alkyl Halides 1) Organometallic Reactions–Formation of Gilman Reagents a. General Reaction: RX + 2Li Rδ+Liδ− + LiBr Example: H3C Br 2Li δ− CH3 Li δ+ LiBr b. General Reaction: ether 2 Rδ-Liδ+ + CuI - R2Cu Li + + LiI Gilman Reagent Example: 2CH3δ−Liδ+ 2) ether CuI (CH3)2Cu−Li+ LiI Corey-House (Formation of Gilman Reagent) General Reaction: R2Cu- Li+ + R’X ether R’R + LiX + RCu Example: (CH3)2Cu−Li+ Cl CH2CH3 H3C CH3Cu CH2CH3 This reaction is useful because it forms new carbon-carbon bonds, lengthening the carbon chain. The alkyl halide loses a halogen and gains the alkyl group from the Gilman Reagent. 3) Formation of Grignard Reagent General Reaction: ether RX + Mg RMgX Example: Br ether MgBr Mg Like Gilman Reagents, Grignard Reagents are useful because they can be used to lengthen a carbon chain. You’ll study Grignard reagents and the reactions they are involved in during the second semester of organic chemistry. LiCl Practice Problems 3) Prepare: CH2MgBr From: H3C CH2CH3 and any other needed reagents H3C H3CH2C CH3 Answer 4) Prepare: CH3 From: ClCH2CH3 HC and any other needed reagents CH2CH3 CH3 Answer Answer 1) H2C CHCH2CH2CH3 NBS ? Br H2C CHCH2CH2CH3 NBS H2C CHCHCH2CH3 Return to Problem Answer 2) Cl ? SOCl2 Cl OH SOCl2 Return to Problem Answer 3) Prepare: CH2MgBr From: H3C CH2CH3 and any other needed reagents H3C CH3 H3CH2C NBS H3C Mg H3C H3CH2C CH3 CH2Br H3CH2C ether CH2MgBr H3C CH2CH3 Return to Problem Answer 4) Prepare: CH3 From: ClCH2CH3 HC and any other needed reagents CH2CH3 CH3 CH3 ClCH2CH3 (CH3CH)2CuLi CH3 HC CH2CH3 CH3 Return to Problem