* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 10. Alkyl Halides - faculty at Chemeketa
Survey
Document related concepts
Elias James Corey wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Asymmetric induction wikipedia , lookup
Ene reaction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
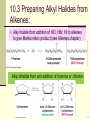
Polythiophene wikipedia , lookup
Discodermolide wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
George S. Hammond wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Transcript
10. Organohalides Why this Chapter? Reactions involving organohalides are less frequently encountered than other organic compounds, but reactions such as nucleophilic substitutions/eliminations that they undergo will be encountered Alkyl halide chemistry is model for mechanistically similar but more complex 2 What Is an Alkyl Halide An organic compound containing at least one carbon- halogen bond (C-X) X (F, Cl, Br, I) replaces H Can contain many C-X bonds Properties and some uses Fire-resistant solvents Refrigerants Pharmaceuticals and precursors 3 10.1 Naming Alkyl Halides Find longest chain, name it as parent chain (Contains double or triple bond if present) Number from end nearest any substituent (alkyl or halogen) 4 Number all halogens and list different in alphabetical order. 5 Naming if Two Halides or Alkyl Are Equally Distant from Ends of Chain Begin at the end nearer the substituent whose name comes first in the alphabet 6 Common Names Treat halogen as parent with alkyl side group 7 Learning Check: Give names for the following: 8 Solution: Give names for the following: Iodobutane (butyl iodide) 1-chloro-3-methylbutane (isopentyl chloride) 1,5-dibromo-2,2-dimethylpentane 1-chloro-3-ethyl-4-iodopentane 1,3-dichloro-3-methylbutane 2-bromo-5-chlorohexane 9 10.2 Structure of Alkyl Halides C-X bond is longer as you go down periodic table C-X bond is weaker as you go down periodic table C-X bond is polarized with slight positive on carbon and slight negative on halogen 10 10.3 Preparing Alkyl Halides from Alkenes: Alkyl halide from addition of HCl, HBr, HI to alkenes to give Markovnikov product (see Alkenes chapter) Alkyl dihalide from anti addition of bromine or chlorine 11 10.3 Preparing Alkyl Halides from Alkanes: Radical Halogenation Alkane + Cl2 or Br2, heat or light replaces C-H with C- X but gives mixtures Hard to control Via free radical mechanism It is usually not a good idea to plan a synthesis that uses this method 12 Radical Halogenation of Alkanes If there is more than one type of hydrogen in an alkane, reactions favor replacing the hydrogen at the most highly substituted carbons (not absolute) 9 1o H’s 1 3o H So would expect 10:90 ratio if # of H’s was sole factor 13 Relative Reactivity: Chlorination Based on quantitative analysis of reaction products, relative reactivity is estimated Order parallels stability of radicals 14 Relative Reactivity: Chlorination 1 9 3o H 1o H’s So would expect 10:90 ratio if # of H’s was sole factor 1 9 3o H 1o H’s 1x5=5 9x1=9 So would predict 5:9 ratio Or 5/14 : 9/14 = 35.7: 64.3 15 Learning Check: Predict ratio of mono-chlorinated butanes 16 Solution: Predict ratio of mono-chlorinated butanes 6 4 1o H’s 2o H’s So would expect 60:40 ratio if # of H’s was sole factor 6 4 1o H’s 2o H’s 6x 1 =6 4 x 3.5 = 14 So would predict 6:14 ratio Or 6/20 : 14/20 = 30: 70 17 Learning Check: How many constitutional isomers will form in the radical mono-chlorination of 2,3-dimethylpentane? 1. 2. 3. 4. 5. 3 4 5 6 7 18 Solution: How many constitutional isomers will form in the radical mono-chlorination of 2,3-dimethylpentane? 1. 2. 3. 4. 5. 3 4 5 6 7 19 Learning Check: The selectivity of chlorine radical is 1.0 : 3.5 : 5.0 for 1°, 2° and 3° hydrogens, respectively. If only monochlorides were to form in the radical chlorination of 1,3-dimethylcyclobutane, what is the expected yield of the tertiary chloride? 1. 2. 3. 4. 5. 1/6 1/5 1/4 1/3 1/2 20 Solution: The selectivity of chlorine radical is 1.0 : 3.5 : 5.0 for 1°, 2° and 3° hydrogens, respectively. If only monochlorides were to form in the radical chlorination of 1,3-dimethylcyclobutane, what is the expected yield of the tertiary chloride? 1. 2. 3. 4. 5. 1/6 1/5 1/4 1/3 1/2 21 Relative Reactivity: Bromination Reaction distinction is more selective with bromine than chlorine 22 Relative Reactivity: Bromination Formation of C radical more costly with Br. So Br more picky thus more selective. 23 Learning Check: Which of these radicals is expected to be the least selective? 1. 2. 3. 4. 5. HO• F• Cl• Br• I• 24 Solution: Which of these radicals is expected to be the least selective? 1. 2. 3. 4. 5. HO• F• Cl• Br• I• 25 10.4,5 Preparing Alkyl Halides from Alkenes: Allylic Bromination Allylic position more reactive Allylic radical stabilized by resonance 26 Allylic Stabilization Allyl radical is delocalized More stable than typical alkyl radical by 40 kJ/mol (9 kcal/mol) Allylic radical is more stable than tertiary alkyl radical 27 Allylic Bromination: NBS N-bromosuccinimide (NBS) selectively brominates allylic positions Requires light for activation A source of dilute bromine atoms 28 Allylic Bromination: NBS 29 Allylic Bromination: NBS Product mix favors more substituted C=C and less hindered Br 30 Examples: NBS Br Br CH3 CH3 CH2 Br Br CH3 Br CH2 31 Use of Allylic Bromination Allylic bromination with NBS creates an allylic bromide Reaction of an allylic bromide with base produces a conjugated diene, useful in synthesis of complex molecules 32 Learning Check: Predict the products of allylic bromination w/ NBS NBS NBS 33 Solution: Predict the products of allylic bromination w/ NBS NBS NBS CH3 Br CH3 CH3 CH3 CH3 + CH3 C CH CH CH2 CH3 CH3 C CH CH CH Br H CH3 CH3 C CH CH CH2 Br CH3 CH3 CH3 Br C CH CH CH CH3 H Br Br 34 10.6 Preparing Alkyl Halides from Alcohols Reaction of tertiary C-OH with HX is fast and effective Add HCl or HBr gas into ether solution of tertiary alcohol Primary and secondary alcohols react very slowly and often rearrange, so alternative methods are used 35 Example: 36 Alkyl Halides from Alcohols Alternative methods for slow primary and secondary alcohols to avoid rearrangements. 37 Learning Check: Prepare the following from alcohols 38 Solution: Prepare the following from alcohols OH OH CH3 CH3 CH3 CH3 CH CH2 CH CH3 CH3 C CH3 OH CH3 CH2 HCl PBr3 CH2 OH CH CH2 C CH3 CH3 CH3 CH2 CH2 CH2 CH PBr3 CH3 HCl 39 10.7 Reactions of Alkyl Halides: Grignard Reagents Reaction of RX with Mg in ether or THF Product is RMgX – an organometallic compound (alkyl-metal bond) R is alkyl 1°, 2°, 3°, aryl, alkenyl X = Cl, Br, I 40 Grignard Reagents Forms a basic/nucleophilic Carbon Example: 41 10.8 Organometallic Coupling Reactions Alkyllithium (RLi) forms from RBr and Li metal RLi reacts with copper iodide to give lithium dialkylcopper (Gilman reagents) Lithium dialkylcopper reagents react with alkyl halides to give alkanes 42 Examples: 43 Utility of Organometallic Coupling in Synthesis Coupling of two organometallic molecules produces larger molecules of defined structure Aryl and vinyl organometallics also effective Coupling of lithium dialkylcopper molecules proceeds through trialkylcopper intermediate 44 Palladium-catalyzed Tributyltin chloride Works well with aryl or vinyl halides 45 10.9 Oxidation and Reduction in Organic Chemistry In organic chemistry, we say that oxidation occurs when a carbon or hydrogen that is connected to a carbon atom in a structure is replaced by oxygen, nitrogen, or halogen Oxidation is a reaction that results in loss of electron density at carbon (as more electronegative atoms replace hydrogen or carbon) Oxidation = LEO Gaining an Oxygen Or Losing a Hydrogen Oxidation: break C-H (or C-C) and form C-O, C-N, C-X 46 Reduction Reactions Organic reduction is the opposite of oxidation Results in gain of electron density at carbon (replacement of electronegative atoms by hydrogen or carbon) Reduction = GER Losing an Oxygen Or Gaining a Hydrogen Reduction: form C-H (or C-C) and break C-O, C-N, C-X 47 Oxidation Levels Functional groups are associated with specific levels CH4 -4 -3 -2 0 +2 +4 48 Examples: 49 Examples: 50 Learning Check: Which of these is not a redox reaction? 1. 2. x HCl Cl x Br2 Br Br 3. x H2 Pd 4. x H2O/DMSO 5. x OH NBS 1. OsO4 Br HO OH 2. NaHSO3/H2O 51 Solution: Which of these is not a redox reaction? 1. 2. x HCl Cl x Br2 Br Br 3. x H2 Pd 4. x H2O/DMSO 5. x OH NBS 1. OsO4 Br HO OH 2. NaHSO3/H2O 52 Biological Halides 53 Learning Check: Radical chlorination of (2R)-fluorobutane yields 2-chloro-3fluorobutane as one of the products. Which is the best prediction for the stereochemistry of this product? 1. 2. 3. 4. 5. two enantiomers in equal amounts two diastereomers in different amounts two diastereomers in equal amounts four stereoisomers, in different amount each four stereoisomers, with enantiomeric pairs in equal amounts, but diastereomeric pairs in non-equal amounts 54 Solution: Radical chlorination of (2R)-fluorobutane yields 2-chloro-3fluorobutane as one of the products. Which is the best prediction for the stereochemistry of this product? 1. 2. 3. 4. 5. two enantiomers in equal amounts two diastereomers in different amounts two diastereomers in equal amounts four stereoisomers, in different amount each four stereoisomers, with enantiomeric pairs in equal amounts, but diastereomeric pairs in non-equal amounts 55 Learning Check: Which mechanistic step determines the regiochemistry of the radical-chain chlorination of 2-methylpropane? 1. 2. 3. 4. 5. protonation step hydrogen abstraction step chlorine abstraction step chloride addition step initiation step 56 Solution: Which mechanistic step determines the regiochemistry of the radical-chain chlorination of 2-methylpropane? 1. 2. 3. 4. 5. protonation step hydrogen abstraction step chlorine abstraction step chloride addition step initiation step 57 Learning Check: Dipole moments of CH3–X are 1.85, 1.87, 1.81, and 1.62 D for F, Cl, Br, and I, respectively. What is the best explanation of the small differences in dipole moments? 1. 2. 3. 4. 5. The electronegativity of halogens in alkyl halides is nearly constant. The inductive effects are counterbalanced by hyperconjugation. The electronegativity effects are counterbalanced by bond strengths. Lone-electron pairs are more delocalized on smaller halogens. The charge-separation and bond-lengths trends have opposite effects. 58 Solution: Dipole moments of CH3–X are 1.85, 1.87, 1.81, and 1.62 D for F, Cl, Br, and I, respectively. What is the best explanation of the small differences in dipole moments? 1. 2. 3. 4. 5. The electronegativity of halogens in alkyl halides is nearly constant. The inductive effects are counterbalanced by hyperconjugation. The electronegativity effects are counterbalanced by bond strengths. Lone-electron pairs are more delocalized on smaller halogens. The charge-separation and bond-lengths trends have opposite effects. 59 Learning Check: Which of the following is not the product of the radical chain-reaction of 2-pentene with NBS? 1. 2. 3. 4. 60 Solution: Which of the following is not a product of the radical chain-reaction of 2-pentene with NBS? 1. 2. 3. 4. 61 Learning Check: What is the best reagent to carry out the following transformation? Br 1. 2. 3. 4. 5. PBr3 HBr NBS/hv/CCl4 Br2/hv NBS/H2O/DMSO 62 Solution: What is the best reagent to carry out the following transformation? Br 1. 2. 3. 4. 5. PBr3 HBr NBS/hv/CCl4 Br2/hv NBS/H2O/DMSO 63 Learning Check: What is the best reagent to carry out the following transformation? OH 1. 2. 3. 4. 5. Br PBr3 HBr/ether NBS/hv/CCl4 Br2/hv NBS/H2O/DMSO 64 Solution: What is the best reagent to carry out the following transformation? OH 1. 2. 3. 4. 5. Br PBr3 HBr/ether NBS/hv/CCl4 Br2/hv NBS/H2O/DMSO 65 Learning Check: Which sequence of reagents is best to accomplish the following transformation? Br 1. 2. 3. 4. 5. D KOH followed by BD3/THF KOH followed by NaBH4 Mg/ether followed by D2O Li/pentane followed by D2/CuI NaNH2 followed by D2/Pd 66 Solution: Which sequence of reagents is best to accomplish the following transformation? Br 1. 2. 3. 4. 5. D KOH followed by BD3/THF KOH followed by NaBH4 Mg/ether followed by D2O Li/pentane followed by D2/CuI NaNH2 followed by D2/Pd 67 Learning Check: The selectivity of chlorine radical is 1.0 : 3.5 : 5.0 for 1°, 2° and 3° hydrogens, respectively. Assuming that only monochlorides are produced in the radical chain chlorination of 2,3-dimethybutane, what would be the expected ratio of the two isomeric alkyl chlorides formed in the reaction? 1. 2. 3. 4. 5. 1/6 1/4 1/2 3/5 5/6 68 Solution: The selectivity of chlorine radical is 1.0 : 3.5 : 5.0 for 1°, 2° and 3° hydrogens, respectively. Assuming that only monochlorides are produced in the radical chain chlorination of 2,3-dimethybutane, what would be the expected ratio of the two isomeric alkyl chlorides formed in the reaction? 1. 2. 3. 4. 5. 1/6 1/4 1/2 3/5 5/6 69 Learning Check: The reaction shown produces 5 different bromides. Which of the compounds listed is one of the expected products? NBS CCl4 1. 2. 4. 3. 5. 70 Solution: The reaction shown produces 5 different bromides. Which of the compounds listed is one of the expected products? NBS CCl4 1. 2. 4. 3. 5. 71 Learning Check: Which of the following molecules would not yield an organolithium reagent when treated with Li in pentane? 1. 2. 3. 4. 5. iododecane 4-bromo-1-pentanol phenyl bromide 4-bromobutyl methyl ether cyclohexylbromide 72 Solution: Which of the following molecules would not yield an organolithium reagent when treated with Li in pentane? 1. 2. 3. 4. 5. iododecane 4-bromo-1-pentanol phenyl bromide 4-bromobutyl methyl ether cyclohexylbromide 73 Learning Check: What is the IUPAC name of the following molecule? Cl 1. 2. 3. 4. 5. 2,4-dimethylheptyl-6-chloride 2,4,6-trimethyl-6-chlorohexane 2-chloro-4,6-dimethylheptane 1-chloro-1,3,5-trimethylhexane 2-chloro-4-methyl-isoheptane 74 Solution: What is the IUPAC name of the following molecule? Cl 1. 2. 3. 4. 5. 2,4-dimethylheptyl-6-chloride 2,4,6-trimethyl-6-chlorohexane 2-chloro-4,6-dimethylheptane 1-chloro-1,3,5-trimethylhexane 2-chloro-4-methyl-isoheptane 75 Learning Check: What is the product of the following sequence of reactions? I I 1. 2Li CuI pentane ether ether 3. 2. 4. 5. 76 Solution: What is the product of the following sequence of reactions? I I 1. 2Li CuI pentane ether ether 3. 2. 4. 5. 77