* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download CITRIC ACID CYCLE
Metabolic network modelling wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Metalloprotein wikipedia , lookup
Butyric acid wikipedia , lookup
Mitochondrion wikipedia , lookup
Electron transport chain wikipedia , lookup
Glyceroneogenesis wikipedia , lookup
Photosynthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Microbial metabolism wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Nicotinamide adenine dinucleotide wikipedia , lookup
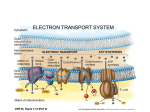
CITRIC ACID CYCLE- discovered by Sir Hans Krebs in 1953. He was awarded Nobel Prize in Medicine 1. 2. 3. 4. The citric acid cycle (also known as the tricarboxylic acid cycle, the TCA cycle, or the Krebs cycle) is a series of chemical reactions of central importance in all living cells that utilize oxygen as part of cellular respiration. In aerobic organisms, the citric acid cycle is part of a metabolic pathway involved in the chemical conversion of carbohydrates, fats and proteins into carbon dioxide and water to generate a form of usable energy. It is the second of three metabolic pathways that are involved in fuel molecule catabolism and ATP production, the other two being glycolysis and oxidative phosphorylation. The citric acid cycle also provides precursors for many compounds such as certain amino acids, and some of its reactions are therefore important even in cells performing fermentation. ACETYL-CoA (Acetyl Coenzyme A, ACoA) Catabolism of carbohydrates, fatty acids, amino acids releases energy from ACoA During glycolysis, glucose is converted to pyruvate which is converted to ACoA by a group of enzymes known as pyruvate dehydrogenase complex NADH is produced during those process In TCA two carbon atoms of the acetylCoenzyme A are ultimately oxidized to CO2 ACoA is a carrier of acyl groups and the catalyzed steps of TCA occurs in the mitochondria. CITRIC ACID CYCLE REACTIONS OF THE CITRIC ACID CYCLE 1. 2. 3. CITRIC SYNTHASE catalyses the condensation of ACoA with oxaloacetate to form citrate. The reaction is reversible, but is a main regulatory point. A low NAD+/NADH ratio and succinylCoA inhibits its activity ACONITASE reversibly catalyses the conversion of citrate to isocitrate ISOCITRATE DEHYDROGENASE oxidatively decarboxylates isocitrate to α-ketoglutarate (2-oxyglutarate). In this process NAD+ s reduced to NADH and CO2 is released. Isocitrate dehydrogenase is inhibited by ATP and NADH and activated by ADP and NAD+ REACTIONS contd. 4. α-KETOGLUTARATE DEHYDROGENASE produces succinyl- CoA from α-ketogluturate and coenzyme A. Another NAD+ is reduced to NADH and CO2 is released. Both NADH and succinyl-CoA inhibit the enzyme complex, 2-oxygluturate dehyrogenase complex 5. SUCCINYL-CoA SYNTHETASE converts succinyl-CoA to succinate. GDP (glyceralaldehyde di-phosphate) is converted to GTP during this step. This is the only step in TCA (citric acid cycle) that involves substrate-level phosphoyrlation to directly produce a high energy phosphate bond. REACTIONS contd. 6. 7. 8. SUUCINATE DEHYDROGENASE SYNTHETASE oxidizes succinate to fumarate. This enzyme is membrane bound in the mitochondria and transfers two H+ to FAD to form FADH2. It is inhibited by oxaloacetate. FUMARATE HYDRATASE (fumarase) reversebly hydrates fumarate to form malate. MALATE DEHYDRGENASE forms oxaloacetate and one more FADH from malate to complete the cycle Hence, one cycle produces 1 GTP (step 5), 3 NADH’s (steps 3 ,4, and 8) and 1FADH2 (step 6). SUMMARY OF THE REACTIONS The sum of all reactions in the citric acid cycle is: Acetyl-CoA + 3 NAD+ + FAD + GDP + Pi + 3 H2O → CoA-SH + 3 NADH + H+ + FADH2 + GTP + 2 CO2 + 3 H+ Two carbons are oxidized to CO2, and the energy from these reactions is stored in GTP , NADH and FADH2. NADH and FADH2 are coenzymes (molecules that enable or enhance enzymes) that store energy and are utilized in oxidative phosphorylation. SIMPLIFIED VIEW OF THE PROCESS The process begins with the oxidation of pyruvate, producing one CO2, and one acetyl-CoA. Acetyl-CoA reacts with the four carbon carboxylic acid, oxaloacetate-to form the six carbon carboxylic acid, citrate. Through a series of reactions citrate is converted back to oxaloacetate. This cycle produces 2 CO2 and consumes 3 NAD+, producing 3 NADH and 3 H+. It consumes 3 H2O and consumes one FAD, producing one FADH+. 1st turn end= 1 ATP, 3 NADH, 1 FADH2, 2 CO2 Since there are two molecules of Pyruvic acid to deal with, the cycle turns once more. The complete end result= 2 ATP, 6 NADH, 2 FADH2, 4 CO2 REGULATION Many of the enzymes in the TCA cycle are regulated by negative feedback from ATP when the energy charge of the cell is high. Such enzymes include the pyruvate dehydrogenase complex that synthesises the acetyl-CoA needed for the first reaction of the TCA cycle. Also the enzymes citrate synthase, isocitrate dehydrogenase and alpha-ketoglutarate dehydrogenase, that regulate the first three steps of the TCA cycle, are inhibited by high concentrations of ATP. This regulation ensures that the TCA cycle will not oxidise excessive amounts of pyruvate and acetyl-CoA when ATP in the cell is plentiful. This type of negative regulation by ATP is by an allosteric mechanism REGULATION contd. Several enzymes are also negatively regulated when the level of reducing equivalents in a cell are high (high ratio of NADH/NAD+). This mechanism for regulation is due to substrate inhibition by NADH of the enzymes that use NAD+ as a substrate. This includes both the entry point enzymes pyruvate dehydrogenase and citrate synthase. ENERGETICS OF TCA The overall consumption of one molecule of acetylCoA in the TCA is a spontaneous, exergonic process (having overall negative free energy change) ; ΔGo’ = -60 kJ mol-1. The rate of utilization of the ACoA in the cycle depends on the energy status within the mitochondria. Under conditions of high energy, the concentrations of NADH and ATP are high, and those of NAD+ and FADH2 are low. The reoxidation of NADH and FADH2 occurs in the electron transport chain and is necessary for he cycle to continue. IMPORTANT FACTS Many of the intermediates of the citric acid cycle are used in in the synthesis of other biomolecules Many biomolecules feed into the citric acid cycle Thus, the TCA is considered to be amphibolic OVERALL CHEMICAL REACTIONS DURING ONE TURN OF TCA The overall reactions are the complete oxidation of one molecule of ACoA, the release of two molecules of CO2, the reduction of three molecules of NAD+ and one FAD, and the phosphorylation of one molecule of GDP.