* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Families of SMA - Children with Spinal Muscular Atrophy
No-SCAR (Scarless Cas9 Assisted Recombineering) Genome Editing wikipedia , lookup
Genealogical DNA test wikipedia , lookup
Neocentromere wikipedia , lookup
Oncogenomics wikipedia , lookup
Epigenetics of diabetes Type 2 wikipedia , lookup
Gene desert wikipedia , lookup
Public health genomics wikipedia , lookup
Epigenetics of human development wikipedia , lookup
Gene nomenclature wikipedia , lookup
Copy-number variation wikipedia , lookup
Genetic engineering wikipedia , lookup
Gene therapy wikipedia , lookup
Neuronal ceroid lipofuscinosis wikipedia , lookup
Cell-free fetal DNA wikipedia , lookup
Gene therapy of the human retina wikipedia , lookup
Genome evolution wikipedia , lookup
Gene expression profiling wikipedia , lookup
X-inactivation wikipedia , lookup
History of genetic engineering wikipedia , lookup
Gene expression programming wikipedia , lookup
Saethre–Chotzen syndrome wikipedia , lookup
Site-specific recombinase technology wikipedia , lookup
Frameshift mutation wikipedia , lookup
Nutriepigenomics wikipedia , lookup
Vectors in gene therapy wikipedia , lookup
Helitron (biology) wikipedia , lookup
Therapeutic gene modulation wikipedia , lookup
Genome (book) wikipedia , lookup
Designer baby wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Point mutation wikipedia , lookup
Microevolution wikipedia , lookup
Epigenetics of neurodegenerative diseases wikipedia , lookup
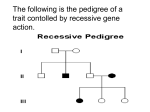
Genetics: The Whole Picture SMA Takes the Hill 2003 Debra G.B. Leonard, M.D., Ph.D. Director, Molecular Pathology Laboratory University of Pennsylvania Health System Philadelphia, PA Objectives Explain the genetic testing options for SMA Leave no one behind What We Will Talk About Basic Clinical Features of SMA Basics of Genetics SMN Gene Structure SMA Diagnostic Test SMA Carrier Test Questions and Discussion Spinal Muscular Atrophy The brain makes the body move by sending nerve signals from the brain to nerve cells in the spinal cord called the anterior horn motor neurons These motor neurons relay signals to the muscles which cause the muscles to contract Movement occurs when muscles contract The anterior horn motor neurons no longer function in individuals with SMA Since muscles are not signaled to contract and are not used, the muscles atrophy or get smaller Clinical Types of SMA SMA Type I: Werdnig-Hoffmann SMA Type II: Intermediate SMA Type III: Kugelberg-Welander Most severe form of SMA Onset at birth to 3 months, death by ~2 yrs Symptoms begin at infancy to toddler age Survive beyond 4 yrs of age Onset after age 2 yrs to adult Basics of Genetics Genetic Information DNA and Chromosomes Genes Messenger RNA (mRNA) Proteins Inheritance Pedigrees Genetic Information Genetic information directs growth and development, and determines physical characteristics Every cell in the human body has the same genetic information Each cell uses a different part of the genetic information to perform that cell’s function, e.g. skin, blood, muscle, nerve, etc. DNA and Chromosomes Genetic information is encoded by DNA Pieces of DNA in cells are called chromosomes There are 24 kinds of human chromosomes: Each normal cell has 46 chromosomes: 1 through 22 (1 is the longest; 22 is the shortest) X and Y are the “sex” chromosomes 2 copies of 1 through 22, plus 2 sex chromosomes XX is female, XY is male Chromosomes Chromosomes consist of DNA plus proteins The proteins help to organize the DNA pieces Each chromosome has a centromere The centromere divides the DNA into two parts Each part has a centromeric end and a free end The free end is called the telomeric end Chromosome Structure One Chromosome p q Arm Arm Centromeric Telomeric Centromeric Telomeric Centromere If arms are of unequal length: short arm is called p (petite) long arm is called q What Makes Each Person Unique? Each egg or sperm contains 23 chromosomes One egg and one sperm combine to make a fetus One of each pair of chromosomes 1 to 22, at random One of the two sex chromosomes, at random Each person gets half their chromosomes from their mother and half from their father Siblings are similar because they share some of the same chromosomes, but different because they have some different chromosomes DNA Encodes Genetic Information DNA is a chain of four different building blocks (or bases) called A, C, G and T A, C, G and T are the letters of the genetic alphabet Some parts of each DNA chain encode instructions which the cell uses to make proteins, that do work in cells Protein-coding parts of DNA are called genes Other parts of each DNA chain are nonsense Genes - 1 One gene encodes one protein, more or less Each gene has regulatory regions, protein coding regions and nonsense regions Coding parts of genes are called exons Noncoding, nonsense parts of genes are called introns Gene Structure Gene Promoter Intron Exon Intron Exon Exon Promoter region regulates gene expression, i.e., controls when a gene will be used to make the protein it encodes Genes - 2 Genes are located on the arms of the chromosomes Each kind of chromosome contains a different set of genes Because each cell contains two of each kind of chromosome, each cell contains two copies of all the human genes, except the genes on the X and Y chromosomes in males There are ~25,000 human genes Gene Expression: How Are Proteins Made from Genes? When the protein encoded by a gene is needed by the cell, RNA copies of the gene are made DNA and RNA are both called nucleic acids RNA uses bases A, C, G and U, that correspond to the A, C, G and T bases of DNA The RNA copy is processed to remove the introns and is then called messenger RNA or mRNA mRNA is the blueprint used to make the protein The Genetic Code A protein is a chain of amino acids 3 mRNA bases code for one amino acid Therefore, the mRNA is used as the blueprint to make a protein by the protein-making or translation machinery of a cell While DNA is very stable, the mRNA is short-lived (minutes to hours), so the cell can change its gene expression, and therefore what it is doing, as needed Gene Expression DNA TRANSCRIPTION RNA RNA PROCESSING mRNA TRANSLATION Protein Cell Work TRANSCRIPTION Nucleic acid Nucleic acid (DNA) (RNA) Same Language TRANSLATION Nucleic acid Protein (RNA) (Amino Acid) Different Language Genetic Diseases A genetic disease is due to a change in the DNA sequence of a gene Because DNA in chromosomes is passed from parent to child, genetic diseases are also passed from parent to child A change in the DNA sequence of a gene is called a mutation Examples of Gene Mutations A change of one base of a gene can change an amino acid in the protein or can shorten (or truncate) the protein, affecting the function of the protein Deletion of part or all of the gene sequence, so the protein is not made Change sequences that direct intron removal, so the mRNA is not correctly made, so the protein is not made Types of Inheritance Single gene diseases are caused by mutation of one gene, e.g. cystic fibrosis, SMA, Huntington disease Multi-gene diseases are caused by a combination of mutations in several genes, e.g. heart disease, asthma, arthritis Types of Inheritance Single gene diseases are caused by mutation of one gene, e.g. cystic fibrosis, SMA, Huntington disease Dominant inheritance: Mutation of one gene copy causes disease Recessive inheritance: Mutation of both gene copies causes disease Genetic Terminology Affected: Someone who has a genetic disease Can be either a dominant or recessive disease Carrier: Someone who has a gene mutation for a recessive disease in only one gene copy Person does not have disease symptoms, but may pass on mutation to their children Pedigrees = Male = Female = Carrier = Affected = Fetus = Deceased = Marriage = Children Used to Describe Family Relationships and Diseases Dominant Disease Risk Family 1 Family 2 Family 3 A/A A/a A/A A/A A/A A/a A/A A/A A/A A/A A/a A/A A/A Mother A/A A/A A = Normal copy a = Mutant copy Father A a A A/A A/a A A/A A/a 2 of 4 or 50% risk of having an affected child Recessive Disease Risk B/b B/B B/b Family 2 B/B B/b b/b B/B B/B Family 3 B/b B/B B/B B/b B/B B/B B/b Father Family 1 B = Normal gene b = Mutant gene Mother B b B B/B B/b b B/b b/b 1 of 4 or 25% risk of having an affected child 2 of 4 or 50% risk of having a child who is a carrier Spinal Muscular Atrophy Single gene recessive disease Second most common lethal recessive disease after cystic fibrosis Carrier frequency of ~1 in 50 Incidence of ~1 in 10,000 births 1995: Identification of Gene for SMA Lefebvre, S, et al., Cell 80: 155, 1995. SMN gene (Survival of Motor Neurons) located on long arm of chromosome 5 (5q) SMN gene has 9 exons & encodes a 294 aa protein In addition to the SMN gene, a copy of the SMN gene is present on 5q, located centromeric to the SMN gene SMNt for telomeric or SMN1 is mutated to cause SMA SMNc for centromeric or SMN2 may alter severity of SMA SMN1 and SMN2 have only two base differences located in exons (one in exon 7 & one in exon 8) Structure of SMN Gene Region SMNc or 2 SMNt or 1 RNA 1 2a mRNA Protein 2b 3 4 5 6 7 8 2 base differences in exons between SMN1 and SMN2 SMN Gene Mutation Causes SMA Lefebvre, S, et al., Cell 80: 155, 1995. Deletion of exon 7 or 7 & 8 associated with SMA 229 Patients: 103 Type I, 91 Type II, 35 Type III 213/229 (93%): exon 7 & 8 deleted on both SMN1 copies 13/229 (5.6%): only exon 7 deleted on both SMN1 copies 2/229 (0.9%): exon 7 deletion on one SMN1 gene copy and a smaller mutation on the other SMN1 gene copy 1/229 (0.4%) had point mutation on one gene only 246 Controls: None with deletion of exon 7 + 8 on both SMN1 genes Mutation Types in SMA ~94% of SMA patients have deletion of exon 7 from both of their SMN1 genes ~6% of SMA patients have an exon 7 deletion on one SMN1 gene copy and a small mutation on the second SMN1 copy Rarely, SMA patients may have non-deletion mutations on both SMN1 gene copies (estimated to be ~1 in 1,000 people with SMA) SMA Diagnostic Test Diagnosis of SMA is by absence of SMN1 exon 7 Testing complicated by presence of SMN2 gene which has an exon 7 with only 1 base difference from SMN1 Diagnostic test uses PCR method to make millions of copies of exon 7 from both the SMN1 and SMN2 genes The 1 base difference allows the SMN2 PCR copies to be cut into 2 pieces, but not the SMN1 PCR copies The PCR copies are examined and an absence of the intact SMN1 PCR copies is diagnostic of SMA for 94% of individuals with SMA SMA Diagnostic Test Gel electrophoresis to examine intact SMN1 and cut SMN2 PCR copies Normal (95%) SMN1 (200 bp) SMN2 (176 bp) SMN2 (24 bp) Normal (5%) SMA Specimens for SMA Diagnostic Test All cells of the body have the same DNA Therefore, SMA testing can be performed on any cells from a person who needs to be tested Generally, a tube of blood is used Prenatal specimens can also be used Method for SMA Diagnostic Test DNA is purified from the cells of the specimen DNA is used for PCR of SMN1 and SMN2 exon 7 The SMN2 PCR copies are cut The PCR products are examined on a gel Absence of SMN1 exon 7 copies confirms SMA diagnosis SMA Diagnostic Test: The Limitations SMA (94%) SMA (6%) NonCarrier Carrier SMN1 (200 bp) SMN2 (176 bp) SMN2 (24 bp) Only positive for ~94% of individuals with SMA. Cannot distinguish SMA carrier from non-carrier. Family 1: Requesting Prenatal Counseling SMA Type II Diagnosed 1995 10 weeks What choices does this family have? Family 1: The Options Can use direct amniotic fluid, cultured amniocytes or CVS to test the fetus Does the affected son have an exon 7 SMN1 deletion on both his SMN1 gene copies? If not known, testing the son will increase the predictive value of fetal testing Can do tests for son and fetus at the same time or sequentially Family 1: The Decision The family chooses to: Use an amniotic fluid specimen so do not have to wait for culturing the amniocytes Have the son and the fetus tested at the same time SMA Diagnostic Test Results Son Fetus SMN1 (200 bp) SMN2 (176 bp) SMN2 (24 bp) SMA Will Not Be Diagnosis Affected Confirmed (2 Deletions) Family 1: Extended Family Wife’s brother and his wife want to know their risk of having a child affected with SMA What can be done? Family 1: Extended Family The SMA Diagnostic Test can only be used to diagnose people with SMA symptoms The brother and his wife are not affected, but may be carriers Need a test that can detect SMA carriers SMA Carrier Test Drs. Tom Prior and Arthur Burghes from Ohio State University first reported SMA Carrier Test method in 1997 Non-radioactive adaptation of the their method developed at UPenn McAndrew et al., Am J Hum Genet 60: 1411, 1997 SMA Carrier Test: Theory The goal is to determine the number of SMN1 exon 7 copies a person has The number of PCR copies made depends on the number of gene copies in the DNA used for PCR More SMN1 gene copies produce more SMN1 PCR copies Fewer SMN1 gene copies produce fewer SMN1 PCR copies The number of SMN1 PCR copies made is compared to the number of PCR copies made from a gene “always” present in 2 copies (CFTR gene) SMA Carrier Test: Method Two PCRs done in one test: Cut SMN2 PCR copies Quantify SMN1 and CFTR PCR copies Calculate SMN1 gene copies: Exon 7 of SMN1 and SMN2 genes Part of the CFTR gene Number of SMN1 copies SMN1 Gene Copy # = X2 Number of CFTR copies SMA Carrier Test: Gel Analysis Normal Carrier Affected Normal Normal (2 SMN1) (1 SMN1) (0 SMN1) (3 SMN1) (0 SMN2) CFTR SMN1 SMN2 SMN2 SMN Gene Region Possibilities NORMAL CHROMOSOMES SMN2 SMN1 SMN1 SMN1 SMN1 SMA Carrier Test: Limitations Carrier test will not detect 3% of SMN1 gene mutations that are not SMN1 exon 7 deletions 6% of SMA patients have one non-deletion mutation This equals 3% of the SMN1 gene copies Carrier test cannot differentiate: One SMN1 gene copy on each of 2 chromosomes (not a carrier), from 2 SMN1 gene copies on one chromosome and no SMN1 gene copies on the second chromosome (carrier) Two SMN1 Copies by Carrier Test 1 Copy on Each Chromosome 5 (Not a Carrier) SMN2 SMN1 SMN2 SMN1 2 Copies on One Chromosome 5 with a Deletion (Carrier) SMN2 SMN1 SMN1 Family 1: Extended Family Wife’s brother and his wife want to know their risk of having a child affected with SMA What can be done? Family 1: The Choices The wife can be tested by the SMA Carrier Test to determine her SMN1 gene copy # The brother can be tested by the SMA Carrier Test, but his carrier risk would be reduced if his sister is shown to have an exon 7 SMN1 deletion Most likely sister is a carrier since her son has two deletion mutations, although new mutation frequency is high New Mutations in SMA Approximately 2% of SMA patients have a new mutation on one of their SMN1 genes This means that one parent was not a carrier The majority of new mutations occur in the SMN1 gene copy inherited from the father Family 1: The Choices The wife can be tested by the SMA Carrier Test to determine her SMN1 gene copy # The brother can be tested by the SMA Carrier Test, but his carrier risk would be reduced if his sister is shown to have an exon 7 SMN1 deletion. The sister and her husband could be tested to rule out a new mutation in their son. Family 1: The Decision The family chooses to: Test both the brother and his wife Test both the sister and her husband to: Improve the interpretation of testing for the brother Check for a possible new mutation in their son Family 1: SMA Carrier Test Results CFTR SMN1 SMN2 SMN2 Family 1: SMA Carrier Test Results 2 copies 0 copies 1 copy Not tested 1 copy 2 copies Brother and sister are both carriers. Brother’s risk before testing was 1 in 2, and now is 1 Family 1: SMA Carrier Test Results 2 copies 1 copy 1 copy 2 copies What do carrier results mean for brother’s wife? 0 copies Not tested Family 1: Married into SMA Family Before testing, the wife had ~1 in 50 chance of being a carrier (carrier frequency in general population) She has 2 copies, but still has a small risk of carrying a non-deletion mutation or having 2 SMN1 copies on one chromosome and a deletion on the other chromosome (2+0 Carrier) Carrier with 2 SMN1 Gene Copies 2 + 0 Carrier SMN2 SMN1 SMN1 Non-deletion Mutation Carrier SMN2 SMN1 SMN2 SMN1 Family 1: Married into SMA Family Before testing, the wife had ~1 in 50 chance of being a carrier (carrier frequency in general population) She has 2 copies, but still has a small risk of carrying a non-deletion mutation or having 2 SMN1 copies on one chromosome and a deletion on the other chromosome (2 + 0 Carrier) By Bayesian analysis, wife’s carrier risk is reduced from ~1 in 50 to ~1 in 800 Family 1: SMA Carrier Test Results 2 copies 1 copy 1 copy 2 copies What is this couple’s risk of having a child with SMA? 0 copies Not tested Family 1: Couple’s Combined Risk Before testing, the couple’s risk of having a child with SMA was ~1 in 400 (1/2 X 1/50 X 1/4) After testing know: Brother is a carrier (risk of 1) Wife’s risk of being a carrier is ~1 in 800 without including risk of a new mutation since she is female Therefore, the risk of having an affected child is reduced to ~1 in 3200 (1 X 1/800 X 1/4) Family 1: SMA Carrier Test Results 2 copies 1 copy 1 copy 2 copies Why was fetus not tested? 0 copies Not tested Family 1: Prenatal SMA Testing In general, the SMA Carrier Test is not used for prenatal diagnosis Use SMA Diagnostic Test to test if fetus has deletion of SMN1 Individual can choose to have Carrier testing in the future as an adult May use SMA Carrier Test for testing of a fetus in a family with a non-deletion mutation Family 1: SMA Carrier Test Results ? 2 copies 1 copy 1 copy 2 copies Why does “obligate carrier” have 2 copies? 0 copies Not tested Family 1: “Carrier” with 2 Copies New Mutation SMN1 new mutation rate estimated at ~2% (7 in 340 SMA families) 11 of 15 cases had new mutation on father’s chromosome revealing a high incidence of rearrangement during spermatogenesis Son may have a new mutation, reducing couple’s future risk 2+0 Carrier Gonadal Mosaicism: Some but not all sperm have deletion Two SMN1 copies on one chromosome and none on other Frequency ~8% of people not affected with SMA We have seen 1 case with 2 copies in blood and <2 copies in sperm Resolve new mutation from 2+0 by linkage analysis If new mutation, test father’s sperm for mosaicism Linkage Analysis Method for tracking chromosomes in a family For SMA, track chromosome 5q Must include affected family member to define which 5q’s have mutated SMN1 genes In combination with Carrier Test, can distinguish 2+0 from new mutation, but requires extended family members Can be used to identify carriers in families with non-deletion mutations Uses of SMA Carrier Test Family member of person with SMA (parents, sibling, aunt, uncle, cousin, grandparent, etc.) Married into family with SMA Married to someone affected with SMA Symptomatic with negative SMA Direct Test Parents of one child with SMA to potentially identify a new mutation and decrease future risk Sperm donors and/or recipients Prenatal diagnosis for non-deletion mutation Non-Deletion Mutation Testing Most non-deletion mutations occur in exon 6 of the SMN1 gene Sequence analysis of the SMN1 gene can sometimes identify the mutation Can use the known mutation to track the mutated gene through a family and for prenatal diagnosis Not currently available except for research (Dr. Gonzalez, Dupont Children’s Hospital, DE) SMA Genetic Testing Summary SMA Diagnostic Test SMA Carrier Test Use for diagnosis of SMA Only positive for ~94% of people with SMA Cannot distinguish SMA carrier from non-carrier Determines SMN1 gene copy number Cannot detect non-deletion or 2:0 carriers Further clarification by linkage analysis by tracking chromosome 5 in a family Questions ?