* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Molecules Power Point
Protein (nutrient) wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Protein moonlighting wikipedia , lookup
Genetic code wikipedia , lookup
Cell-penetrating peptide wikipedia , lookup
Western blot wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Deoxyribozyme wikipedia , lookup
Expanded genetic code wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Protein structure prediction wikipedia , lookup
Endomembrane system wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Protein adsorption wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
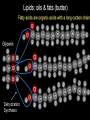
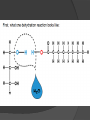
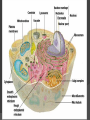
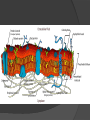
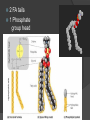
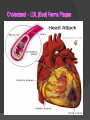
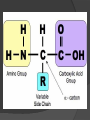
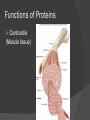
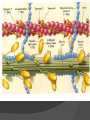
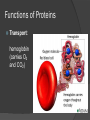
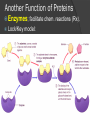
Organic Molecules The Chemistry of Life What makes it ORGANIC? • Molecules made up of mainly CARBONs and HYDROGENs – (w/other elements at times like O, N, P) – Carbon’s special trait: Making 4 bonds How many bonds? 4 Major Categories 1st: Carbohydrates 4 Major Categories 2nd: Proteins 4 Major Categories 3rd: Lipids 4 Major Categories 4th: Nucleic Acids RNA and DNA Carbohydrates • Monosaccharides (Simple sugars): – The smallest of sugars; taste sweet – Single carbon ring – Ex: Fructose, Glucose (pictured) Carbohydrates • Disaccharides – Two monosaccharides chemically combined – Ex: Sucrose (table sugar) , Lactose (in milk) How they’re made: Dehydration Synthesis (Condensation Reaction) How they’re made: Dehydration Synthesis (Condensation Reaction) How they’re made: Dehydration Synthesis (Condensation Reaction) Carbohydrates • Polysaccharides – Large, long molecules – Made up of many monosaccharides – Found in plants: • Starch • Energy storage • Cellulose • Structural support Carbohydrates • Polysaccharides – Large, long molecules – Made up of many monosaccharides – Found in animals: • Glycogen • Energy Storage in muscle tissue • Chitin • Exoskeleton of insects, arthropods • Other glycogen: A note about some big molecules: Some very large molecules are actually built out of small, repeating units. Ex: This (**************************) is made up of a bunch of these: (*) The repeating units are referred to as MONOMERS. The large molecule is a POLYMER So, starch is a polymer made of monomers called _______________ . Lipids Fatty, greasy, oily, or waxy Made of C,H, & some O Insoluble in water Lipids: 4 categories Triglicerides/fats Phospholipids Waxes Steroids Importance of LIPIDS to humans: Energy source Cushion vital organs Insulation Majority of cell membranes In some vitamins and hormones Fats: Made from Fatty Acids Carbon chains; 12-28 C long Three types of Fatty Acids: saturated, unsaturated, polyunsaturated stearic acid oleic acid linolenic acid Saturated Monounsaturated Polyunsaturated Dehydration Synthesis Triglyceride is formed Phospholipids Main component of membranes of cells and their organelles 2 FA tails 1 Phosphate group head Steroids (Sterols) 4 fused Carbon rings Some hormones (testosterone) Cholesterol – most familiar ○ Produced in liver & eaten (meat, dairy products, etc.) ○ Found in cell membranes (reduces fluidity of membrane) & in vertebrate brains Cholesterol – LDL (Bad) Forms Plaque Waxes Firm consistency Important in water-proofing Review! What is the difference between a saturated fat and an unsaturated fat? Why does the body need cholesterol? Where do you find phospholipids? PROTEINS Building blocks for tissues (more than a source for energy) Made of C, H, O, N, sometimes S Is a POLYMER Made of monomers called amino acids… Types of amino acids Here are the 20 amino acids that we have. Notice the tops are the same, but the colored areas are the different R groups. Protein Uses… Functions of Proteins Structural: connective tissue, hair, nails You are mostly protein. Functions of Proteins Contractile (Muscle tissue) Functions of Proteins Transport: hemoglobin (carries O2 and CO2) Functions of Proteins Some Hormones like insulin, growth hormone Another Function of Proteins Enzymes; facilitate chem. reactions (Rx). Lock/Key model: More about Enzymes Allows chem. Rx to occur with less “Activation energy” More about Enzymes 1. 2. Many end in “-ase” Are catalysts: substance that initiates or accel. a chem. Rx w/o itself being affected 3. Important Characteristics: 1. Each unique: Used for Only 1 type Rx 2. Are reusable 3. Aren’t consumed in Rx 4. Can run Rx in reverse What can mess up enzymes? 1. 2. HEAT Think of a frying egg… pH Very high/low pH’s sabotage active sites on enzyme This is called DENATURING Is irreversible Nucleic acids • DNA (Deoxyribonucleic acid) – Genetic material; carries heredity – codes for all proteins • RNA (Ribonucleic acid) – Similar in structure to DNA; used for building of proteins • Nucleic acids’ monomers: Nucleotides (5 C sugar, P grp, nitrogenous base) What’s a calorie? Unit used to measure energy The amount of energy it takes to raise 1 g of water 1ºC = 1 calorie One Kilocalorie (C) = 1000 calories (c) Kcal is what’s on food labels. What are your calorie needs per day? What is your BMR (Base Metabolic Rate)? Michael Phelps, at the height of his training, requires 12,000 Calories/day. Caloric values… • Carbohydrates and Proteins have about the same caloric values per unit vol. – One gram of carbs = 4 calories – One gram of protein = 4 calories – One gram of lipids = 9 calories • Lipids (fats), made for energy storage, have a higher caloric value. – One pound of fat = 3500 Calories.