* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Enantioselective synthesis wikipedia , lookup
Marcus theory wikipedia , lookup
Cracking (chemistry) wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Discodermolide wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Ene reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Stille reaction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Petasis reaction wikipedia , lookup
Elias James Corey wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Hydroformylation wikipedia , lookup
Kinetic resolution wikipedia , lookup
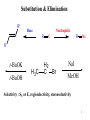
Substitution & Elimination R" Base Nucleophile R R X Nu R' t-BuOK t-BuOH H3C H2 C Br NaI MeOH Selectivity : SN or E, regioselectivity, stereoselectivity 1 Chapter 9 Alcohols, Ethers and Epoxides 2 Alcohols, Ethers and Epoxides Introduction—Structure and Bonding • Alcohols contain a hydroxy group (OH) bonded to an sp3 hybridized carbon. 3 Alcohols, Ethers and Epoxides Introduction—Structure and Bonding • enols and phenols Compounds having a hydroxy group on a sp2 hybridized carbon undergo different reactions than alcohols. • Will be discussed in chapter 11, 19 4 Alcohols, Ethers and Epoxides Introduction—Structure and Bonding • Ethers have two alkyl groups bonded to an oxygen atom. 5 Alcohols, Ethers and Epoxides Introduction—Structure and Bonding • Epoxides are ethers having the oxygen atom in a three-membered ring. Epoxides are also called oxiranes. • The C—O—C bond angle for an epoxide must be 60°, a considerable deviation from the tetrahedral bond angle of 109.5°. Thus, epoxides have angle strain, making them much more reactive than other ethers. 6 Alcohols, Ethers and Epoxides Introduction—Structure and Bonding • The oxygen atom in alcohols, ethers and epoxides is sp3 hybridized. Alcohols and ethers have a bent shape like that in H2O. • The bond angle around the O atom in an alcohol or ether is similar to the tetrahedral bond angle of 109.5°. • Because the O atom is much more electronegative than carbon or hydrogen, the C—O and O—H bonds are all polar. 7 Alcohols, Ethers and Epoxides Nomenclature of Alcohols : -- ol 8 Alcohols, Ethers and Epoxides For cyclic compounds • When an OH group is bonded to a ring, the ring is numbered beginning with the OH group. • Because the functional group is at C1, the 1 is usually omitted from the name. • The ring is then numbered in a clockwise or counterclockwise fashion to give the next substituent the lowest number. 9 Alcohols, Ethers and Epoxides Nomenclature of Alcohols • Common names are often used for simple alcohols. Name all the carbon atoms of the molecule as a single alkyl group. Add the word alcohol, separating the words with a space. 10 Alcohols, Ethers and Epoxides Nomenclature of Alcohols • Compounds with two hydroxy groups : diols or glycols. • Compounds with three hydroxy groups : triols and so forth. 11 Alcohols, Ethers and Epoxides Nomenclature of Ethers • Simple ethers are usually assigned common names. To do so: Name both alkyl groups bonded to the oxygen, arrange these names alphabetically, and add the word ether. For symmetrical ethers, name the alkyl group and add the prefix “di-”. 12 Alcohols, Ethers and Epoxides • More complex ethers are named using the IUPAC system. One alkyl group is named as a hydrocarbon chain, and the other is named as part of a substituent bonded to that chain: Name the simpler alkyl group as an alkoxy substituent by changing the –yl ending of the alkyl group to –oxy. Name the remaining alkyl group as an alkane, with the alkoxy group as a substituent bonded to this chain. 13 Alcohols, Ethers and Epoxides • Cyclic ethers have an O atom in the ring. A common example is tetrahydrofuran (THF). This name has a different origin 14 Nomenclature of Epoxides • Epoxides can be named in three different ways—As epoxyalkanes, oxiranes, or alkene oxides. • To name an epoxide as an epoxyalkane, first name the alkane chain or ring to which the O atom is attached, • and use the prefix “epoxy” to name the epoxide as a substituent. • Use two numbers to designate the location of the atoms to which the O’s are bonded. 15 •Epoxides can be named in three different ways—As epoxyalkanes, oxiranes, or alkene oxides. • Epoxides bonded to a chain of carbon atoms can also be named as derivatives of oxirane, the simplest epoxide having two carbons and one oxygen atom in a ring. • The oxirane ring is numbered to put the O atom at position one, and the first substituent at position two. • No number is used for a substituent in a monosubstituted oxirane. 16 •Epoxides can be named in three different ways—As epoxyalkanes, oxiranes, or alkene oxides. • Epoxides are also named as alkene oxides, since they are often prepared by adding an O atom to an alkene. To name an epoxide in this way: Mentally replace the epoxide oxygen with a double bond. Name the alkene. Add the word oxide. 17 Alcohols, Ethers and Epoxides Physical Properties • Alcohols, ethers and epoxides exhibit dipole-dipole interactions because they have a bent structure with two polar bonds. • Alcohols are capable of intermolecular hydrogen bonding. Thus, alcohols are more polar than ethers and epoxides. • Steric factors affect hydrogen bonding. 18 Alcohols, Ethers and Epoxides 19 Alcohols, Ethers and Epoxides Interesting Alcohols 20 Alcohols, Ethers and Epoxides Interesting Alcohols glycerol Sugars, cellulose, starch etc. 21 Alcohols, Ethers and Epoxides Interesting Ethers Diethyl ether is the best-known ether, first used as a general anesthetic in the 1830s. Because of its high flammability and side-effects on some patients, its place as an anesthetic has been taken by other substances. Cyclic polyether : crown ether 22 Alcohols, Ethers and Epoxides Interesting Ethers • The ability of crown ethers to complex cations can be exploited in nucleophilic substitution reactions, as shown in Figure 9.5. 23 Alcohols, Ethers and Epoxides Interesting Ethers : nature’s crown ether • Brevetoxin B is a naturally occurring polyether that interferes with Na+ ion transport across cell membranes. “Red Tide” 24 마산 앞바다 적조 25 26 San Diego Interesting Epoxides : epoxides are reactive! • Ethylene oxide : precursor of ethylene glycol, polyethylene glycol OH O O * O HO n • Natural epoxides 27 Preparation of Alcohols, Ethers, and Epoxides • Alcohols and ethers are both common products of nucleophilic substitution of alkyl halides. Williamson ether synthesis. 28 Preparation of Alcohols, Ethers, and Epoxides 29 Williamson ether synthesis. • SN2 reaction Strong Nucleophile (An alkoxide salt) is needed to make an ether. • Cyclic ethers are formed through intramolecular reaction. Br NaH HO n When n= n O -2, epoxides 30 Preparation of Epoxides • Organic compounds that contain both a hydroxy group and a halogen atom on adjacent carbons are called halohydrins. 31 Reactions of Alcohols Substitution, elimination, oxidation • The OH group in alcohols is a very poor leaving group. • For an alcohol to undergo nucleophilic substitution, OH must be converted into a better leaving group (activated). • By using acid, ¯OH can be converted into H2O, a good leaving group. • By converting to sulfonates, ¯OH can be converted into 32 ¯OSO2R, a good leaving group. Reactions of Alcohols Br OH HBr (for SN) or H2SO4 (for E) + OH2 SN Halide E Alkene 33 Reactions of Ethers and Epoxides Ethers do not have a good leaving group and are difficult to activate. Epoxides are highly reactive due to high angle strain. Ring-opening results in relief of strain O C C O C _ C Nu _ Nu: 34 Reactions of Alcohols—Dehydration • Dehydration, like dehydrohalogenation, is a elimination reaction in which the elements of OH and H are removed from the and carbon atoms respectively. • Dehydration is typically carried out using H2SO4 and other strong acids, or phosphorus oxychloride (POCl3) in the presence of an amine base. 35 Reactions of Alcohols—Dehydration • Typical acids used for alcohol dehydration : H2SO4 or ptoluenesulfonic acid (TsOH). strong organic acid • More substituted alcohols dehydrate more easily, giving rise to the following order of reactivity. 36 Alcohols, Ethers and Epoxides Reactions of Alcohols—Dehydration •Dehydration follows the Zaitsev rule. The more substituted alkene is the major product when a mixture of constitutional isomers is possible. 37 Alcohols, Ethers and Epoxides Reactions of Alcohols—Dehydration • Secondary and 3° alcohols react by an E1 mechanism 38 Alcohols, Ethers and Epoxides Reactions of Alcohols—Dehydration • The E1 dehydration of 20 and 30 alcohols with acid gives clean elimination products without any by-products formed from an SN1 reaction. • Clean elimination takes place because the reaction mixture contains no good nucleophile to react with the intermediate carbocation, so no competing SN1 reaction occurs. • This makes the E1 dehydration of alcohols much more synthetically useful than the E1 dehydrohalogenation of alkyl halides. 39 Alcohols, Ethers and Epoxides Reactions of Alcohols—Dehydration • 1° alcohols undergo dehydration following an E2 mechanism. • 1° carbocations are highly unstable, their dehydration cannot occur by an E1 mechanism involving a carbocation intermediate. 40 Reactions of Alcohols—Dehydration While dehydrations have favorable entropy changes, the position of equilibrium favors reactants. Operation of the Principle of Le Châtelier in Alcohol Dehydrations • removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alkene, which usually has a lower boiling point than the starting alcohol, can be removed by distillation as it is formed, thus driving the 41 equilibrium to the right to favor production of more product. Carbocation Rearrangements CH3 H CH3 H2SO4 H3C CH3 CH3 OH CH3 H H3C H3C CH3 CH2 CH3 CH3 42 Alcohols, Ethers and Epoxides Carbocation Rearrangements • Often, when carbocations are intermediates, a less stable carbocation will be converted into a more stable carbocation by a shift of a hydrogen or an alkyl group. This is called a rearrangement. 43 Carbocation Rearrangements • A 1,2-shift can convert a less stable carbocation into a more stable carbocation. • Rearrangements can occur whenever a carbocation is formed as a reactive intermediate. 1,2-hydride shift 44 45 Dehydration of Alcohols Using POCl3 and Pyridine • Dehydration under basic condition • A common method uses phosphorus oxychloride (POCl3) and pyridine (an amine base) in place of H2SO4 or TsOH. Dehydration proceeds by an E2 mechanism. 46 Dehydration of Alcohols Using POCl3 and Pyridine Mechanism : E2 mechanism 47 Dehydration of Alcohols in the natural product synthesis 48 Conversion of Alcohols to Alkyl Halides with HX • Substitution reactions do not occur with alcohols unless ¯OH is converted into a good leaving group. • The reaction of alcohols with HX (X = Cl, Br, I) is a general method to prepare 1°, 2°, and 3° alkyl halides. 49 Conversion of Alcohols to Alkyl Halides with HX • More substituted alcohols usually react more rapidly with HX: • The mechanism depends on the structure of the R group. 50 Conversion of Alcohols to Alkyl Halides with HX 1o alcohols and methanol react via an SN2 mechanism 1. Protonation CH3 H HX C OH D (S) CH3 H C + OH2 D Primary alcohol 2. SN2 with inversion of configuration _ X CH3 + H H C D + OH2 CH3 X C + (R) D OH2 51 2o and 3o alcohols react via an SN1 mechanism 1. Protonation CH3 CH3CH2 C Ph (R) OH CH3 CH3CH2 C Ph HX + OH2 _ + X Tertiary alcohol 2. SN1 reaction with racemization (a) Dissociation CH3 CH3CH2 + C OH2 Ph CH3CH2 CH3 C+ + OH2 Ph Carbocation (planar) (b) Attack of nucleophile CH3 CH3CH2 C Ph (R) X _ CH3CH2 X Attack from right CH3 X C+ Ph _ X Attack from left Racemate CH3 CH2CH3 C (S) Ph 52 Alcohols, Ethers and Epoxides Conversion of Alcohols to Alkyl Halides with HX • The reactivity of hydrogen halides increases with increasing acidity. HI RCH2 OH RCH2 I HCl RCH2 OH too slow to detect products 53 Conversion of Alcohols to Alkyl Halides with HX • 2o alcohols 54 Conversion of Alcohols to Alkyl Halides HI RCH2 OH RCH2 I 55 Conversion of Alcohols to Alkyl Halides with SOCl2 and PBr3 • For primary and 2° alcohols • SOCl2 (thionyl chloride) converts alcohols into alkyl chlorides. • PBr3 (phosphorus tribromide) converts alcohols into alkyl bromides. via an SN2 reaction 56 57 Conversion of Alcohols to Alkyl Halides with SOCl2 and PBr3 • For primary and 2° alcohols • SOCl2 (thionyl chloride) converts alcohols into alkyl chlorides. • PBr3 (phosphorus tribromide) converts alcohols into alkyl bromides. via an SN2 reaction 58 Alcohols, Ethers and Epoxides Conversion of Alcohols to Alkyl Halides with SOCl2 and PBr3 59 60 Alcohols, Ethers and Epoxides 61 Tosylate—Another Good Leaving Group • An alkyl tosylate is composed of two parts: the alkyl group R, derived from an alcohol; and the tosylate (short for ptoluenesulfonate), which is a good leaving group. • A tosyl group, CH3C6H4SO2¯, is abbreviated Ts, so an alkyl tosylate becomes ROTs. 62 Tosylate—Another Good Leaving Group • This process converts a poor leaving group (¯OH) into a good one (¯OTs). • Tosylate is a good leaving group because its conjugate acid, ptoluenesulfonic acid (CH3C6H4SO3H, TsOH) is a strong acid (pKa = -7). 63 Tosylate—Another Good Leaving Group • Because alkyl tosylates have good leaving groups, they undergo both nucleophilic substitution and elimination, exactly as alkyl halides do. 64 Tosylate—Another Good Leaving Group • Substitution occurs via an SN2 mechanism. • Tosylates are equivalent to halides. 65 Example: 66 Alcohols, Ethers and Epoxides Reaction of Ethers with Strong Acid cleavage occurs either side of the ether oxygen atom to give two alkyl halide products, so ethers are not generally as useful as alcohols in synthesis. 67 Mechanism of 68 Mechanism of 69 Reactions of Epoxides • Nucleophilic attack opens the strained three-membered ring, making it a favorable process even with a poor leaving group. Reactions occur with strong nucleophiles (such as OH-, CH3S-, CH3O- or CN-), in the absence of acid and with weaker nucleophiles (such as Cl-) in the presence 70 of strong acid (such as HCl). Reactions of Epoxides • Common nucleophiles that open the epoxide ring include ¯OH, ¯OR, ¯CN, ¯SR and NH3. With these strong nucleophiles. • The reaction occurs by an SN2 mechanism. 71 Reactions of Epoxides : stereochemical consequences Nucleophilic attack of ¯OCH3 occurs from the backside at either C—O bond, because both ends are similarly substituted. Since attack at either side occurs with equal probability, an equal amount of the two enantiomers (i.e., a 72 racemic mixture) is formed. Alcohols, Ethers and Epoxides Reactions of Epoxides Optically inactive starting materials give optically inactive products! 73 Alcohols, Ethers and Epoxides Reactions of Epoxides • Acids HZ that contain a nucleophile Z also open epoxide rings by a two-step sequence. • HCl, HBr and HI, as well as H2O and ROH in the presence of acid, all open an epoxide ring in this manner. 74 Reactions of Epoxides 75 Alcohols, Ethers and Epoxides Reactions of Epoxides 76 Reactions of Epoxides 77 Leukotriene Synthesis and Asthma Drugs OOH CO2H C5H11 CO2H C5H11 C5H11 Lipoxygenase Zileuton blocks this step Arachidonic acid OH CO2H 5-HPETE Sulfur .. nucleophile, RSH .. (glutathione) O CO2H C5H11 SR Leukotriene C4 Leukotriene A4 HO N CONH2 S Zileuton 78 Alcohols, Ethers and Epoxides Important Epoxide Ring Opening Reactions 79 Homework 9.48, 9.49, 9.50, 9.52, 9.56, 9.58, 9.66, 9.67, 9.70, 9.71, 9.76 Preview of Chapter 10 Alkenes Preparation of alkenes Dehydration, dehydrohalogenation Reactions of alkenes Addition reaction --- Markovnikov’s rule -Hydrohalogenation, hydration, halogenantion, hydroboration - stereochemistry of the reaction products Write mechanisms for following reactions. HI RCH2 OH RCH2 I 82