* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Magnesium transporter wikipedia , lookup
Protein (nutrient) wikipedia , lookup
Gene expression wikipedia , lookup
History of molecular evolution wikipedia , lookup
Molecular evolution wikipedia , lookup
Biochemistry wikipedia , lookup
List of types of proteins wikipedia , lookup
Protein moonlighting wikipedia , lookup
Interactome wikipedia , lookup
Immunoprecipitation wikipedia , lookup
Metalloprotein wikipedia , lookup
Intrinsically disordered proteins wikipedia , lookup
Circular dichroism wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Proteolysis wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein adsorption wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Protein purification wikipedia , lookup
Capillary electrophoresis wikipedia , lookup
Community fingerprinting wikipedia , lookup
Gel electrophoresis of nucleic acids wikipedia , lookup
Western blot wikipedia , lookup
Agarose gel electrophoresis wikipedia , lookup
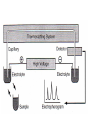
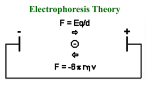
Electrophoresis Overview Electrophoresis • Definition – refers to the migration of charged solutes or particles in a liquid medium under the influence of an electrical field – Iontophoresis • limited to the migration of small ions • free solution or moving boundary method a typical electrophoresis apparatus • Zone Electrophoresis – Migration of charged molecules – usually in a porous supporting medium • cellulose acetate sheets or agarose gel film – generates an electrophoretogram • Support medium • Protein zones are visualized by – staining with a protein-specific stain • Support medium can be dry and record • Quantitation – Densitometer • Solutes of interest – Proteins in serum, urine, cerebrospinal fluid • Background electrolyte (buffer) isotachophoresis • applies specifically to the migration of small ions • no background electrolyte (buffer) is mixed with the sample • leading electrolyte solution – contains ions that are faster than any in the sample • Trailing solution – slower than any in the sample • Sample between leading and Trailing electrolyte isotachophoresis • Applications – separation of small anions and cations, organic and amino acids, peptides, nucleotides, nucleosides, and proteins. • The rate of migration is dependent on – Net electrical charge – Size and shape of the molecule – Electric field strength – Properties of the supporting medium – Temperature of operation • resisting force (counter-force) – Ionic radius of the solute – the viscosity of the buffer solution • electrophoretic mobility – the rate of migration (cm/s) per unit field strength – expressed by the symbol µ – directly proportional to the net charge – inversely proportional to the size of the molecule – inversely proportional to the viscosity of the electrophoresis medium • Factors that can affect mobility – Temperature – Ionic strength – Rate of endosmotic flow – Average pore size of support medium – Point of sample application – wick flow • Drying effect – flow of buffer from both directions Instrumentation and Reagents • Power Supplies – operation at either constant current – constant voltage – constant power • flow of current through a medium – Resistance, production of heat – Heat • thermal agitation of all dissolved ions – increase in both the migration rate – and the rate of evaporation of water • The water loss – increase in ion concentration • decrease in resistance – increases the conductance of the system • To minimize these effects on the migration rate – it is best to use a constant-current power supply Pulsed-power or pulsed-field • periodically change the orientation of the applied field • molecules must reorient • new field direction to fit through the pores in gel • reorientation time depends on molecular size • net migration becomes a function of the frequency of field alteration • separation of very large molecules – Such as DNA fragments greater than 50 kilobases Buffers • they carry the applied current • • • • fix the pH at which electrophoresis is carried out determine the kind of electrical charge on the solute the extent of ionization of the solute they determine the electrode toward which the solute will migrate • The buffer’s ionic strength – concentration of ions • With increasing – molecule becomes more hindered in its movement Buffers • sharpness of the electrophoretic zones • High ionic strength buffer – reduction in resistance – leads to increased current • excessive heat – leads to denaturation of heat-labile proteins – degradation of other components – sharper band separations • Ionic strength – ion concentration – the charge on the ion • most widely used buffers – barbital buffers – Tris-boric acid-EDTA buffers Protein Stains • visualize and locate the separated protein fractions • Dyes commonly used in electrophoresis table – Amido Black (Naphthol Blue Black) – Ponceau S – reactivity toward carrier ampholytes • not suitable for polyacrylamide gel-isoelectric focusing • The amount of dye taken up by the sample – type of protein – degree of denaturation of the proteins Protein Stains Support Media • solutions – such as a sucrose gradient • insoluble gels – sheets, slabs, or columns of starch, agarose, or polyacrylamide,membranes of cellulose acetate Automated Systems • prepackaged gels, sample application through electrophoresis, staining, scanning of gels, and computation of results. • partially automated – ability to process multiple gels of different compositions – simultaneous processing of seven samples by using multiple capillaries General Procedures • a hydrated support material – freshly prepared agarose gel – wetted cellulose acetate • Excess buffer removed from the support surface by blotting • bubbles not be present • support is placed in contact with buffer • Sample is applied to the support • and electrophoresis is conducted using either constant voltage or constant current General Procedures • • • • rapidly dried or placed in a fixative treated with a dye-fixative reagent (staining) washing out excess dye the support is dried – agarose • placed in a clearing agent • cellulose acetate membranes Detection and Quantitation • Densitornetry – moved past a measuring optical system • the area under each peak • report the results – percentage of each fraction present – in terms of absolute concentration • Reliable quantitation of stained zones – requires light of an appropriate wavelength – a linear response from the instrument – a transparent background Detection and Quantitation • agarose gels – satisfies the requirement for a clear background • problems associated with densitometry – differences in quantity of stain taken up by individual proteins – differences in protein zone sizes TYPES OF ELECTROPHORESIS • Starch Gel Electrophoresis – Separates macromolecular ions on the basis • surface charge • molecular size – may be used in a horizontal or vertical direction – proper preparation of gels is relatively difficult – rarely used in the clinical laboratory TYPES OF ELECTROPHORESIS • Agarose Gel Electrophoresis (AGE) – purified, essentially neutral fraction of agar – a convenient method – applied to the analysis of serum proteins, hemoglobin variants, lactate dehydrogenase and creatine kinase isoenzymes, lipoprotein fractions – free of ionizable groups • exhibits little endosmosis • exhibits little background staining – native clarity TYPES OF ELECTROPHORESIS • the agarose surface remain undisturbed – This avoids the surface artifact • Requires sample volume of 0.6 to 3 µL • Electrophoresis time of 30 to 90 min TYPES OF ELECTROPHORESIS • Cellulose Acetate (CAE) – come as dry, opaque, brittle films – the film is soaked in buffer – Characteristics vary with • • • • • extent of acetylation prewashing procedure used by the manufacturer the additives used the pore size thickness of the membrane TYPES OF ELECTROPHORESIS • Serum samples (0.3-2.0 µL) are generally applied • may be made transparent – soaking in a solvent mixture • 95 parts methanol and 5 parts glacial acetic acid • Advantage – speed of separation 20 min- 1 h – ability to store for long periods • disadvantage – need for presoaking before use – need clearing the strips prior to densitometry • largely been replaced by agarose gel TYPES OF ELECTROPHORESIS • Disc Electrophoresis – discontinuities in the electrophoretic matrix – layers of gel that differ in composition and pore size – several proteins with the same electrophoretic mobility • to overcome these deficiencies – Diffusion,broading – Polyacrylamide and Starch gel • pore size is controlled by the percent composition • much smaller than that found in agarose gel TYPES OF ELECTROPHORESIS • Proteins are separated – on the basis of charge and molecular size • molecular sieving – may yield 20 or more fractions – to study individual proteins in serum • genetic variants and isoenzymes TYPES OF ELECTROPHORESIS • Polyacrylamide Gel Electrophoresis (PAGE) – three different layers of gel • small-pore separation gel • large-pore gel (spacer gel ) • a large-pore monomer solution contaming a small amount of serum – Separation then takes place in the bottom separation gel • Advantage – – – – – Thermostable Transparent Strong relatively chemically inert can be made in a wide range of pore sizes • potential carcinogenicity • larger pores; less resistance to the passage of large molecules • ideally suited to the separation of DNA fragments up to 20 kilobases • In homogeneous (non-pulsed) electric field Isoelectric Focusing • The protein migration in a medium possessing a stable pH gradient • moves to a zone in the medium where the pH is equal to the isoelectric point (pI) of the protein. • the charge becomes zero • migration ceases • the protein zones are very sharp – diffusion is also counteracted • acquisition of charge • only 0.02 pH unit • carrier ampholytes – create buffered zones – high concentrations • a high-voltage power source – matrix must be cooled • matrix – polyacrylamide gel • optically clear and supple • large enough pore size – IgM impeded • the anode is surrounded by a dilute acid solution • the cathode by a dilute alkaline solution • Matrix characteristics – Agarose, cellulose acetate • Electroendosmosis-free materials • operating conditions are simple • large pore sizes Two-Dimensional (2D) Electrophoresis • charge-dependent IEF electrophoresis in the first dimension • molecular weight-dependent electrophoresis in the second dimension • first dimensional in a large-pore medium – Ampholytes are added to yield a pH gradient • The second dimension is often polyacrylamide in a line ar or gradient format • Additives – SDS is used in the second dimension – β-mercaptoethanol in the first – SDS in both dimensions and in sample preparation • Electrophoresis under – Native condition – Denaturing conditions • Detection method – – – – Autoradiography Using Coomassie dyes Radiography fluorographic analysis • method limitation – From 7000 polypeptide spots – Autoradiography • 1100 spots are detected – Using Coomassie dyes • about 400 polypeptides are detected • Coomassie dyes – three times more sensitive than Amido Black • silver staining – 100 times more sensitive than Coomassie dye • Radiography , fluorographic analysis – greatest analytical sensitivity High-Resolution Electrophoresis • high-ionic-strength buffer – µ = 0.075 • pH 8.6 • admixture of calcium lactate • Support – agarose gel • temperature control is necessary • Serum proteins resolved into as many as 13 zones High-Resolution Electrophoresis • Discontinuous buffer system – different buffer is used in the electrode chambers from that in the gel • two different pH – alter relative mobility Capillary Electrophoresis • electrophoresis are carried out in capillary tube – 10-100 µm diameter – 20 to 200 cm in length – detector at its terminal end – high-voltage power up to 30 kV – mostly made of fused silica (i.e., pure glass), polyethylene • Advantage – Improved heat dissipation – Sample volumes • in the picoliter to nanoliter range Capillary Electrophoresis • Reduced separation time • Can be fully automated • applications – low- molecular-weight ions to proteins and other macromolecules – Even uncharged molecules • minimizing band spreading – Improved resolution • Due to narrow bore • a variety of detector types can be used Capillary Electrophoresis • Detectors – Optical methods • Ultraviolet- visible photometers • Fluorescence – laser-induced fluorescence • Refractive index • Chemiluminescence – Mass spectrometers – Electrochemical detection methods • Efficient and high-resolution separations – High electric fields • Produce high Joule heat – non uniform temperature gradients – local changes in viscosity » subsequent zone broadening • Control of heat production – reducing the diameter of the capillary tube – reducing the ionic strength of the running electrolyte – reducing the applied voltage • Charged solutes migrate through a polymer network • Larger solutes hindered more than small ones • Molecular sieve • Separation using either – Free solution electrophoresis • Macromolecules, such as DNA and SDS-saturated proteins, cannot be separated without a gel – or a precast gel Blotting Techniques • Southern Blotting – DNA or fragments of DNA are first separated by AGE – fragments are transferred or ‘‘blotted” onto nitrocellulose or a nylon membrane – detected and identified by hybridization with a labeled, complementary nucleic acid probe – determining the presence, position, and number of copies of a gene in a genome Blotting Techniques • Northern Blotting – to separate and detect RNAs and RNA fragments – first separated by AGE (agarose gel electrophoresis ) – blotted to an overlying strip of nitrocellulose – detected and identified by hybridization to a labeled RNA probe Blotting Techniques • Western Blotting – to separate, detect, and identify one or more proteins – first separating the individual proteins by SDS PAGE – transferred or “blotted” onto an overlying strip of nitrocellulose – reacted with a reagent that contains an antibody raised against the protein of interest TECHNICAL CONSIDERATIONS • Electroendosmosis or Endosmosis effect – Support medium • in contact with water takes on a negative charge – fixed ions • Formation an ionic cloud – associated cloud of ions » free to move • the ions in solution are highly hydrated – current is applied • Movement to the electrode of opposite polarity • movement of solvent and its solutes relative to the fixed support – preferential movement of water in one direction TECHNICAL CONSIDERATIONS • media in which endosmosis is strong – conventional cellulose acetate – conventional agarose gel • Sulfate or carboxylic acid groups • surface charges are minimal – starch gel – polyacrylamide gel TECHNICAL CONSIDERATIONS • Buffers – Good culture media – should be refrigerated – discarded after each run • pH changes resulting from the electrolysis – For four electrophoretic runs • Switched the polarity after each run • Mix both buffer boxes TECHNICAL CONSIDERATIONS • Stain Solution – 100 mL for a combined total of 387 cm2 – Considered faulty • if leaching of stained protein zones occurs in the 5% acetic acid • Whenever protein zones appear too lightly stained – stored tightly covered to avoid evaporation TECHNICAL CONSIDERATIONS • Sampling – Typical amounts • to cellulose acetate are 0.3 to 1 .6 µL • In PAGE 3 µL (about 210 pg of total protein) • Agarose – 0.6 to 2.0 µL • depending on the test requirements – For isoenzyme analysis » as much as 25 µL of a normal serum – Discontinuities in Sample Application • due to dirty applicators TECHNICAL CONSIDERATIONS • Unequal Migration Rates – dirty electrodes • uneven application of the electric field – Uneven wetting of the gel TECHNICAL CONSIDERATIONS • Distorted, Unusual, or Atypical Bands – Zone Distortion • bent applicators • incorporation of an air bubble • Over-application of sample – overfill the sample well • excessive drying of the electrophoretic support • excessively wet – cellulose acetate films or agarose gels • Unusual Bands – Artifacts – Hemolyzed samples • an increased β-globulin • unusual band between the α2- and β2-globulins – A band at the starting point • may be fibrinogen – The sample should be verified as being serum – Split zone • α1-, α2-, and β-globulins • split albumin zone – Widened zone • Albumin, in certain medications Atypical Bands • the result of binding – In an isoenzyme pattern • by an immunoglobulin • Irregular but sharp protein zone at the starting point – artifact • lacks the regular, somewhat diffuse appearance – Fibrinogen – Denatured protein • deteriorated serum – Damage done to the cellulose acetate • Paraprotein Evaluation of electrophoretic quality • Include a control serum