* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Motor Proteins and The Cytoskeleton
Survey
Document related concepts
Biochemical switches in the cell cycle wikipedia , lookup
Cell nucleus wikipedia , lookup
Cell culture wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell growth wikipedia , lookup
Microtubule wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell membrane wikipedia , lookup
Extracellular matrix wikipedia , lookup
Signal transduction wikipedia , lookup
Endomembrane system wikipedia , lookup
Cytoplasmic streaming wikipedia , lookup
Transcript
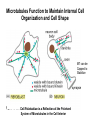
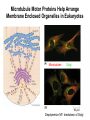
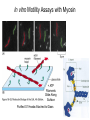
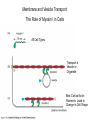
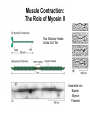
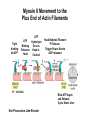
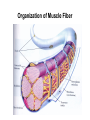
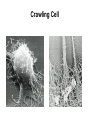
Cell Motility Lecture 17 Cell Motility • Includes: – Changes in Cell Location – Limited Movements of Parts of Cells • Occurs at the Subcellular, Cellular, and Tissue Level • Is Dependent Upon: – The Cytoskeletal Scaffold – Motor Proteins – ATP Motor Proteins and The Cytoskeleton Microtubule-based Movements: Kinesins and Dyneins (MAPS); Fast Axonal Transport Movement of Cell Appendages Movement of Internal Membranes Movement of Chromosomes during Mitosis Actin-Based Movements: Myosins Muscle Contraction Cell Contraction Whole Cell Movement All Motor Proteins use ATP as an energy source. Microtubule Motor Proteins Two Main Families Kinesins- Primarily Plus End Directed -Away from Centrosome Dyneins- Minus End Directed -Toward Centrosome -Largest Motor Proteins 2 Globular Heads -MT Binding -ATP Binding/Hydrolyzing Middle Flexible Domain -Mediates Dimerization NeckTail For Binding Cargo/Adaptors Microtubules Function to Maintain Internal Cell Organization and Cell Shape MT can be Capped to Stabilize Cell Polarization is a Reflection of the Polarized System of Microtubules in the Cell Interior Microtubule Motor Proteins Help Arrange Membrane Enclosed Organelles in Eukaryotes Microtubules Golgi Depolymerize MT- breakdown of Golgi The Tail of the MT Motor Proteins Determines What Cargo Is Transported Cargo Binding: Often Involves Adaptor Proteins Kinesin- Processive -Fast Axonal Transport -Arrange ER Cytoplasmic Dynein- Processive -Localize Golgi Axonemal Dynein Motion of Cilia and Flagella Kinesin Walking to the Plus End of MTs (2) (1) (1) (2) (1) (2) (2) Leading Head (1) Attaches to a b Tubulin (No ATP) Head 1 Binds ATP Head 2 Propelled Forward Past Leading Head ADP Release From Head 2 and Hydrolysis Of ATP by Head 1 Cycle Can Begin Again Moved One Step Processive: At Least One Head is Always Attached Kinesin in vitro Motility Assays Axonemal Dynein: The Motor that Moves Cilia and Flagella Cilia of Rabbit Trachea Cilia from Rabbit Trachea Flagellum Of Sperm ++++ Stationary CellsAllows Movement over Tissue Shorter than Flagella 100+ per cell Generate force by bending at base Flagella Drive Movement of Individual Cell Longer than Cilia 1-2 per cell S shaped wave MT are StabilizedNo Longer Demonstrate Dynamic Instability Organization of the Axoneme “9 + 2” Pattern Nexin Connects Adjacent Doublets Dynein -Head and Tail Bound to Different MT Model for Dynein-Mediated Sliding of MTs (No Nexin) Actin Based Movements: The Myosin Superfamily 18 Classes of Myosins Move toward Plus(Barbed) End of Actin (except one Class) 4 Broad Groups Based on Function: 1)Power Muscle and Cellular Contraction Striated Muscle Contractile Ring-Will cover later lecture 2) Power Membrane and Vesicle Transport Short Range Endocytic and Phagocytic Vesicles 3)Cell Shape and Polarity Head- binds/hydrolyzes ATP and Actin Regulatory Sites Tail- 4) Signal Transduction and Sensory Photoreceptors and Hearing In vitro Motility Assays with Myosin + ATP Filaments Glide Along Surface Purified S1 Heads Attached to Glass Membrane and Vesicle Transport: The Role of Myosin I in Cells All Cell Types Transport a Vesicle or Organelle Bind Cortical Actin Filaments- Lead to Change in Cell Shape Muscle Contraction: The Role of Myosin II Two Globular Heads Coiled Coil Tail Assemble into Bipolar Myosin Filament Myosin II Movement to the Plus End of Actin Filaments Tight binding No ATP ATP ATP Hydrolysis Binding Occurs Releases Head is Head Cocked Head Rebinds Filament Pi Release Trigger Power Stroke ADP released Bind ATP Again and Release Cycle Starts Over Not Processive Like Kinesin Organization of Muscle Fiber Organization of the Skeletal Muscle Fiber Skeletal Muscle Cell (Muscle Fiber) Myofilaments make up Myofibrils Thick- Myosin Thin- Actin Hexagonal Arrangement of Actin Filaments Around the Myosin Skeletal Muscle Banding Pattern Light Band= I Band Thin Actin Fibers Dark Band=A Band Thin Actin and Thick Myosin Fibers Overlap Structural Proteins of the Sarcomere Actin Filament Stability Requires: Cap Z- Cap Plus End of Actin Filament Tropomodulin- Cap Minus Ends of Actin Filament Nebulin –Stabilize Filament/Role in Length a Actinin- Component of Z Disc - Actin Bundling Sarcomeres Shorten During Skeletal Muscle Contraction No Shortening of Thin or Thick Filaments Sliding over one another Myosin Walks Along Actin Filament Attached Released Cocked Nucleotide Free Form ATP Binding ATP Hydrolysis Conformational Change Pi release-Initiates Power Stroke Force Generating Attached Strong BindingPower Stroke (Conformational change to Original Shape ) and ADP Release The Role of Ca2+ and Regulatory Proteins in Skeletal Muscle Contraction The Cytoskeleton in Moving Fibroblast Meshwork Thin, Sheetlike Thin,Stiff Protrusions Crawling Cell Actin-Rich Cortex Moves a Cell Forward Myosin II functions in Cell Contraction • Extension of lamellipodium • Lamellipodium anchors to substrate • Retraction of the opposite end of the cell Formation of Leading Edge of a Lamellipodia Nucleation of Actin at the Plasma Membrane Size of Lamellipodia Remains Constant As Assembly at Leading Edge is Balanced By Disassembly At Rear Extracellular Signals Activate Small G proteins That Regulate ARP Complex Lamellipodia Filopodia