* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download DF-Bio714-2
Signal transduction wikipedia , lookup
Extracellular matrix wikipedia , lookup
Cytokinesis wikipedia , lookup
Tissue engineering wikipedia , lookup
Cell growth wikipedia , lookup
Biochemical switches in the cell cycle wikipedia , lookup
Organ-on-a-chip wikipedia , lookup
Cell encapsulation wikipedia , lookup
Cellular differentiation wikipedia , lookup
Cell culture wikipedia , lookup
List of types of proteins wikipedia , lookup
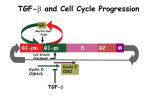
Defective TGF- signaling creates a synthetic lethality for suppression of mTOR Regulation of Cell Cycle Progression G0 (Quiescence) Growth Factor Signals Restriction Point G1-pm G1-ps Cell Growth Checkpoint (mTOR) Tyrosine kinases Ras/Raf/MEK/MAPK S Gatekeepers G2 Myc SV40 Early Region (Suppression of p53, Rb and PP2A) M Survival Signals Generated by Phospholipase D History: •Phospholipase D activity is elevated in cells transformed by v-Src (Song et al., MCB 11:4903, 1991) •Phospholipase D cooperates with elevated tyrosine kinase expression to transform rat fibroblasts (Lu et al., MCB 20:462, 2000) •Phospholipase D suppresses apoptosis induced by over-expressed Raf (Joseph et al., Oncogene 21:3651, 2002) •Phospholipase D suppresses both p53 expression and PP2A activity (Hui et al., MCB 24:5677, 2004; Hui et al., JBC 280, 35829, 2005) •Phospholipase D is required for the phosphorylation (suppression) of Rb (Gadir et al., Cell Cycle 62840,2007) •Phospholipase D stimulates Myc stabilization in breast cancer cells (Rodrik et al., MCB 25, 7917, 2005; FEBS Lett 580:5647, 2006) Regulation of Cell Cycle Progression G0 (Quiescence) Restriction Point Growth Factor Signals G1-pm G1-ps Cell Growth Checkpoint (mTOR) Hypothesis: Tyrosine kinases Ras/Raf/MEK/MAPK S G2 M Gatekeepers Myc SV40 Early Region (Suppression of p53, Rb and PP2A) Elevated PLD activity provides gatekeeper overrides for progression through G1-ps; and cooperates with growth factor signals that promote passage through the Restriction Point PLD Phospholipase D Hydrolysis Ch H O O O=P O- O=P OO CH2 CH2 C=O C=O OH CH2 + H PLD O CH2 CH2 C=O C=O CH2 (CH2)n (CH2)n (CH2)n (CH2)n CH3 CH3 Phosphatidylcholine CH3 CH3 Phosphatidic acid + Ch OH Ch Phospholipase D CH3 Transphosphatidylation CH2 O O O=P O- O=P O- O O CH2 CH2 C=O C=O OH PLD CH2 + CH 2 CH2 CH2 CH3 C=O C=O CH2 (CH2)n (CH2)n (CH2)n (CH2)n CH3 CH3 Phosphatidylcholine CH3 CH3 Phosphatidyl-ethanol + Ch OH Regulators of PLD1 Rho family GTPases (Rho, Rac, Cdc42) Ral GTPase Arf family GTPases Rheb GTPase PKC Phosphatidylinositol-4,5-bis-phosphate (PIP2) Vps34 (PI-3-P) Regulators of PLD2 Constitutively active in vitro Fatty acids PIP2 Vps34? PLD1? Phospholipase D activity is elevated in human cancer and cancer cell lines Breast (PLD1) (Noh et al. Cancer Lett.161:207, 2000) Kidney (PLD2) (Zhao et al. BBRC 278:140, 2000) Gastric (?) (Uchida et al. Anticancer Res. 19:671, 1999) Colorectal (PLD2) (Yamada et al. J. Mol. Med. 81:126, 2003) Lung (?) (Zheng et al., JBC 281:15862, 2006) Bladder (?) (Zheng et al., JBC 281:15862, 2006) Pancreatic (?) (Our unpublished results) Blocking PLD-mediated PA Synthesis Induces Apoptosis In MDA-MB-231 Cells Deprived of Serum 45% % Apoptotic Cells 40% 35% PARP 30% 25% 20% 15% 10% 5% 0% Controll 1-BtOH iso- 2-BtOH t-BtOH BtOH (Zhong et al.. BBRC 302, 615, 2003) PLD provides a survival signal in 786-O renal cancer cells FBS + - + - - - + + + Parp Cl. Parp PLD1 PLD2 Actin Toschi et al. Oncogene. 2008 Conclusion • Elevated PLD activity in human cancer cells provides a survival signal that prevents apoptosis induced by the stress of serum withdrawal Question • How does elevated PLD activity generate survival signals in these cells? Targets of Phosphatidic Acid PLD PA Ras-GAP PI(4,5)P2 Rho/Arf-GAP PI(4)P5-kinase PI(4)P NADPH oxidase Raf mTOR Vesicle formation MEK MAP Kinase Survival Endocytosis Exocytosis mTOR (Mammalian Target of Rapamycin) •Regulator of cell proliferation and cell growth •Responds to nutrients (amino acids, glucose, lipids?) •Regulates initiation of protein synthesis - including Myc •Inhibited by rapamycin •There are two mTOR complexes - mTORC1 and mTORC2 •Phosphatidic acid (PA), the product of PLD, interacts with mTOR competitively with rapamycin •How does PA impact on mTOR? •How does rapamycin work? mTOR and PLD are part of a signaling network that responds to nutrients, energy, and insulin/IGF1 PLD and mTOR are required for the survival of cancer cells - especially when deprived of serum G0 (Quiescence) PLD and mTOR are required for progression through G1-ps - at what we are calling a Cell Growth Checkpoint Restriction Point G1-pm G1-ps Growth Factor Signals Tyrosine kinases Ras/Raf/MEK/MAPK S Cell Growth Checkpoint (mTOR) G2 Gatekeepers Myc SV40 Early Region (Suppression of p53, Rb and PP2A) M Points: Suppression of either PLD or mTOR in the absence of serum results in apoptosis Importantly, suppression of PLD or mTOR does not induce apoptosis in the presence of serum Conclusion There is a factor(s) in serum that prevents apoptosis in cells in response to the suppression of PLD or mTOR Point: Danielpour and colleagues showed that mTOR suppresses TGF- signaling (Song et al., EMBO J, 25:58, 2006). Question: Is TGF- the factor in serum that prevents rapamycin-induced apoptosis in MDA-MB-231 cells? TGF- and Cell Cycle Progression G0 Restriction Point G1-pm G1-ps S Cell Growth Checkpoint Cyclin D CDK4/6 Cyclin E CDK2 TGF- G2 M Effect of rapamycin on cell cycle progression in MDA-MB-231 cells G1 S G2/M G1 S G2/M G1 S G2/M Sub genomic Rapamycin induces primarily G1 arrest in the presence of serum - and apoptosis in the absence of serum Can TGF- suppress rapamycin-induced apoptosis? TGF- is sufficient to suppress rapamycin-induced apoptosis Is TGF- necessary for serum to suppress rapamycininduced apoptosis? Is TGF- in serum necessary for serum to suppress rapamycin-induced apoptosis % Non-Viable Cells Figure 3A 100 % Non-Viable 50 Cells Series1 0 1 - 2 - + 3 + Rap - + + + TGF- -Ab - - - + Serum 4 Cl PARP actin TGF- is necessary for serum to suppress rapamycin-induced apoptosis Summary: •Rapamycin induces apoptosis in MDA-MB-231 cells in the absence of serum •In the presence of serum, rapamycin induces G1 arrest •TGF- is sufficient to suppress rapamycin-induced apoptosis in the absence of serum •TGF- present in serum is necessary for serum to suppress rapamycin-induced apoptosis Question: Why does rapamycin induce apoptosis when TGF- is absent? G0 Restriction Point G1-pm G1-ps S G2 Cell Growth Checkpoint Cyclin D CDK4/6 Cyclin E CDK2 TGF- TGF- suppresses G1 Cell Cycle Progression M G0 Restriction Point G1-pm G1-ps S G2 M Cell Growth Checkpoint Cyclin E CDK2 Cyclin D CDK4/6 TGF- mTOR mTOR suppresses TGF--induced G1 Cell Cycle Arrest Nutrients G0 Restriction Point G1-pm G1-ps S G2 M Cell Growth Checkpoint Cyclin E CDK2 Cyclin D CDK4/6 TGF- mTOR Rapamycin Rapamycin reverses the mTOR suppression of TGF- signaling and cells arrest in G1 in a TGF--dependent mechanism G0 Restriction Point G1-pm G1-ps S G2 M Cell Growth Checkpoint Cyclin D CDK4/6 Cyclin E CDK2 X TGF- mTOR Rapamycin If TGF- signaling is suppressed or defective, there is no G1 arrest with rapamycin treatment - and now the cells die in the presence of rapamycin - Why? G0 Restriction Point G1-pm G1-ps S G2 M Cell Growth Checkpoint Cyclin E CDK2 Cyclin D CDK4/6 TGF- mTOR Rapamycin Hypothesis: There is a critical requirement for mTOR in Sphase. Therefore, allowing cells into S-phase in the presence of rapamycin (ie w/o mTOR) could result in apoptosis G0 Aphidicolin Restriction Point G1-pm Synchronizes Cells in early S G1-ps S G2 M Cell Growth Checkpoint Cyclin E CDK2 Cyclin D CDK4/6 TGF- mTOR Rapamycin If hypothesis is correct, then blocking cells in S-phase - in the presence of serum/TGF- - should result in apoptosis. This is because cells have passed the putative “Cell Growth Checkpoint” and need mTOR signals to facilitate cell cycle progression through S Blocking cells in S-phase with aphidicolin sensitizes cells to rapamycin In the presence of serum/TGF- - if cells are allowed to enter S-phase, then the lack of mTORC1 signals to 4E-BP1 tells the cell that nutrients are in short supply and that replicating the genome is probably a bad career move! % Non-Viable Cells Figure 6A 100 % Non-Viable 50 Cells Series1 0 Rap Aph Cl PARP actin 1 - 2 + 3 - 4 - + + + The cells then do the honorable thing – and commit suicide IMPLICATION: Cancer cells with defective TGF- signaling could be selectively killed by rapamycin in the presence of either serum or TGF- Importantly: Many cancers have defects in TGF- signaling – especially Smad4 - that is critical for suppression of G1 cell cycle progression Cancer cells with defective TGF- signaling are Selectively killed by rapamycin in the presence of serum Breast (Smad4) Breast (No TGF- defect) MDA-MB-231 Breast (PKCδ) 100 SW480 50 Series1 % Non Viable Cells % Non Viable Cells Colon (Smad4) 100 BT-549 50 Series1 0 0 Serum + - - + - Serum + - - + - Rap - - + + + Rap - - + + + TGF- - - - - + TGF- - - - - + Cl PARP actin Cl PARP actin Summary: 1) If TGF- is present, rapamycin induces cell cycle arrest in G1 - by increasing TGF- signaling 2) In the absence of TGF- signaling, rapamycin does not arrest cells in late G1 and they progress through the remainder of G1 into S-phase 3) However, if cells progress into S-phase in the presence of rapamycin, they undergo apoptosis rather than arrest - because of an apparent stringent requirement for mTOR during S-phase Cell Growth Checkpoint Rapamycin induces arrest Rapamycin induces apoptosis G1 S Nutrients Cyclin D-CDK4/6 Cyclin E-CDK2 p27 Rapamycin PLD mTOR TGF- Survival Signals PI3K Growth Factors Implication: Defects in G1 cell cycle progression can create a “Synthetic Lethality” by allowing cells into S-phase where they are more susceptible to apoptotic insult