* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Organic Chemistry
Marcus theory wikipedia , lookup
Metal carbonyl wikipedia , lookup
Kinetic resolution wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Bottromycin wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Elias James Corey wikipedia , lookup
Stille reaction wikipedia , lookup
George S. Hammond wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Diels–Alder reaction wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Hofmann–Löffler reaction wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
1,3-Dipolar cycloaddition wikipedia , lookup
Hydroformylation wikipedia , lookup
Ene reaction wikipedia , lookup
Discodermolide wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Asymmetric induction wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
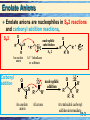
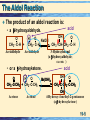
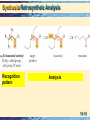
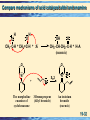
Chapter 19 Enolates and Enamines 19-1 Formation of an Enolate Anion Enolate anions are formed by treating an aldehyde, ketone, or ester, which has at least one a-hydrogen, with base, O CH3 -C- H + N aOH O Na + O H C C-H + H2 O H C C-H H H An enolate anion • Most of the negative charge in an enolate anion is on oxygen. oxygen Reactive carbon 19-2 Enolate Anions Enolate anions are nucleophiles in SN2 reactions and carbonyl addition reactions, O SN2 – R R R An enolate anion Carbonyl addition + R' Br R' + Br R SN 2 R R A 1° haloalkane or sulfonate O R O nucleophilic subs titution – R + R An enolate anion R' nucleophilic addition O R' A ketone O O R R' R' R R A tetrahedral carbonyl addition intermediate 19-3 The Aldol Reaction The most important reaction of enolate anions is nucleophilic addition to the carbonyl group of another molecule of the same or different compound. • Catalysis: Base catalysis is most common although acid also works. Enolate anions only exist in base. 19-4 The Aldol Reaction The product of an aldol reaction is: acid • a -hydroxyaldehyde. OH O O H O a N aOH + CH3 - C-H CH2 - C-H CH3 - CH- CH 2 -C- H Acetaldehyde Acetaldehyde 3-Hydroxybutanal (a -hydroxyaldehyde; racemic ) • or a -hydroxyketone. O H O CH3 -C-CH3 + CH2 -C-CH3 Acetone Acetone acid OH O a CH3 -C-CH2 -C-CH3 CH3 4-Hydroxy-4-meth yl-2-p entanone (a -hydroxyketone) Ba(OH) 2 19-5 Mechanism: the Aldol Reaction, Base Base-catalyzed aldol reaction (good nucleophile) Step 1: Formation of a resonance-stabilized enolate anion. H- O O + H- CH2 - C-H p Ka 20 (weaker acid) H- O-H + O CH2 - C-H pK a 15.7 (stronger acid) O CH2 = C- H An enolate anion Step 2: Carbonyl addition gives a TCAI. O CH3 -C-H + O CH2 -C-H O OCH3 -CH-CH2 -C-H A tetrahed al carbon yl addition intermediate Step 3: Proton transfer to O- completes the aldol reaction. 19-6 Mechanism: the Aldol Reaction: Acid catalysis Before showing the mechanism think about what is needed. • On one molecule the beta carbon must have nucleophilic capabilities to supply an electron pair. • On the second molecule the carbonyl group must function as an electrophile. • One or the other molecules must be sufficiently reactive. 19-7 Mechanism: the Aldol Reaction: Acid catalysis Acid-catalyzed aldol reaction (good electrophile) • Step 1: Acid-catalyzed equilibration of keto and enol forms. O OH Nucleophilic HA carbon CH3 - C-H CH2 = C- H • Step 2: Proton transfer from HA to the carbonyl group of a second molecule of aldehyde or ketone. O CH3 -C-H + H A H O CH3 -C-H + A Reactive carbonyl 19-8 Mechanism: the Aldol Reaction: Acid catalysis • Step 3: Attack of the enol of one molecule on the protonated carbonyl group of the other molecule. • Step 4: Proton transfer to A- completes the reaction. H H O O CH3 -C-H + CH2 =C-H + :A OH O CH3 -CH-CH2 -C-H + H-A (racemic) This may look a bit strange but compare to 19-9 The Aldol Products: Dehydration to alkene • Aldol products are very easily dehydrated to a,unsaturated aldehydes or ketones. OH O CH3 CHCH 2 CH warm in either acid or base O a CH3 CH= CHCH + H2 O An a -unsaturated aldehyde • Aldol reactions are reversible and often little aldol is present at equilibrium. • Keq for dehydration is generally large. • If reaction conditions bring about dehydration, good yields of product can be obtained. 19-10 Crossed Aldol Reactions In a crossed aldol reaction, one kind of molecule provides the enolate anion and another kind provides the carbonyl group. O O NaOH CH3 CCH3 + HCH acid O CH3 CCH2 CH2 OH 4-Hydroxy-2-butanone Nonacidic, no alpha hydrogens 19-11 Crossed Aldol Reactions Crossed aldol reactions are most successful if • one of the reactants has no a-hydrogen and, therefore, cannot form an enolate anion, O • CHO CHO HCH Formald ehyde Benzaldehyde CHO O Furfural 2,2-D imethylprop anal • One reactant has a more acidic hydrogen than the other (next slide) • One reactant is an aldehyde which has a more reactive carbonyl group. 19-12 Crossed Aldol Reactions, Nitro activation Nitro groups can be introduced by way of an aldol reaction using a nitroalkane. O O + H-CH2 -N HO O H-O-H + CH2 -N O N itromethane pK a 10.2 (stronger acid) CH2 =N O Water p Ka 15.7 (weaker acid) O Resonance-stabilized an ion • Nitro groups can be reduced to 1° amines. O HO + CH3 NO2 Cyclohexan on e N itrometh ane NaOH HO CH2 NO2 CH2 NH2 H2 , Ni ( aldol) 1-(N itromethyl)cyclohexanol 1-(A min omethyl)cycloh exanol 19-13 Intramolecular Aldol Reactions • Intramolecular aldol reactions are most successful for formation of five- and six-membered rings. • Consider 2,7-octadione, which has two a-carbons. a O a O O a -H2 O KOH O 2,7-Octanedione O HO (formed) O a O -H2 O KOH OH O (n ot formed) 19-14 Synthesis: Retrosyntheic Analysis Two Patterns to look for 19-15 Synthesis: Retrosyntheic Analysis Recognition pattern Analysis 19-16 Synthesis: Retrosyntheic Analysis Example Mixed aldol Benzaldehyde No alpha hydrogens 19-17 Claisen Condensation, Ester Substitution Esters also form enolate anions which participate in nucleophilic acyl substitution. O 2 CH3 COEt Ethyl ethan oate (Ethyl acetate) - 1 . EtO Na + O O + EtOH CH3 CCH2 COEt 2 . H2 O, HCl Eth yl 3-oxobutanoate Ethanol (Ethyl acetoacetate) • The product of a Claisen condensation is a ketoester. O O a Recognition C C C C OR Element A -ketoest er 19-18 Claisen Condensation • Claisen condensation of ethyl propanoate O O OEt Ethyl propan oate + - OEt Ethyl propan oate 1 . Et O Na + 2 . H2 O, HCl O O OEt + EtOH Eth yl 2-methyl-3oxopen tan oate (racemic) Here the enolate part of one ester molecule has replaced the alkoxy group of the other ester molecule. 19-19 Mechanism: Claisen Condensation Step 1: Formation of an enolate anion. Et O - O + H CH -COEt 2 pK a = 22 (w eaker acid) OO EtOH + CH2 -COEt CH2 =COEt pK a 15.9 Res on ance-s tab ilized enolate anion (stronger acid ) Step 2: Attack of the enolate anion on a carbonyl carbon gives a TCAI. O CH3 -C-OEt + O CH2 -COEt O - O CH3 -C-CH2 -C-OEt OEt A tetrahedral carbonyl add ition in termediate 19-20 Mechanism: Claisen Condensation Step 3: Collapse of the TCAI gives a -ketoester and an alkoxide ion. O O CH3 -C-CH2 -C-OEt O O CH3 -C-CH2 -C-OEt + Et O OEt Step 4: An acid-base reaction drives the reaction to completion. This consumption of base must be anticipated. O O Et O + CH3 -C-CH-C-OEt H pK a 10.7 (stron ger acid) O O CH3 -C-CH-C-OEt + Et OH p Ka 15.9 (w eaker acid) 19-21 Intramolecular Claisen condensation: Dieckman Condensation O Et O - 1 . Et O Na OEt O Diethyl hexanedioate (Diethyl adipate) + 2 . H2 O, HCl O O OEt Acidic + Et OH Ethyl 2-oxocyclopentanecarboxylate 19-22 Crossed Claisen Condsns Crossed Claisen condensations between two different esters, each with a-hydrogens, give mixtures of products and are usually not useful. But if one ester has no a-hydrogens crossed Claisen is useful. OO O HCOEt O EtOCOEt EtOC-COEt Eth yl formate D ieth yl carbonate D iethyl ethaned ioate (D iethyl oxalate) O COEt Ethyl ben zoate No a-hydrogens 19-23 Crossed Claisen Condsns • The ester with no a-hydrogens is generally used in excess. O Ph O OCH3 Meth yl benzoate + - 1 . CH3 O Na OCH3 2 . H2 O, HCl Meth yl p ropan oate O + Ph O OCH3 Methyl 2-meth yl-3-oxo3-ph enylprop anoate (racemic) Used in excess 19-24 Synthesis: Claisen Condensation Claisen condensations are a route to ketones via decarboxylation Reactions 1 & 2: Claisen condensation followed by acidification. O O O + 1 . Et O Na OEt 2 . H O, HCl OEt + Et OH 2 Reactions 3 & 4: Saponification and acidification O O O OEt 3 . Na OH, H2 O, he a t O OH + Et OH 4 . H2 O, HCl Reaction 5: Thermal decarboxylation. O O 5 . he at OH O + CO 2 19-25 Synthesis: Claisen Condensation The result of Claisen condensation, saponification, acidification, and decarboxylation is a ketone. from the ester furnish ing the carbonyl group s everal O O steps + CH -C-OR' R-CH2 -C 2 OR' R from the ester furnish ing the enolate anion O R-CH2 -C-CH2 -R + 2 HOR' + CO2 Note that in this Claisen (not crossed) the ketone is symmetric. Crossed Claisen can yield non symmetric ketones. 19-26 Synthesis: Retrosynthetic Analysis Site of acidic Site of hydrogen, substitution, nucleophile electrophile New bond 19-27 Enamines (and imines, Schiff bases) Recall primary amines react with carbonyl compounds to give Schiff bases (imines), RN=CR2. Primary amine But secondary amines react to give enamines Secondary Amine 19-28 Formation of Enamines Again, enamines are formed by the reaction of a 2° amine with the carbonyl group of an aldehyde or ketone. • The 2° amines most commonly used to prepare enamines are pyrrolidine and morpholine. O N H Pyrrolidine N H Morpholine 19-29 Formation of Enamines • Examples: O H + + + N N H -H2 O OH H An enamine O O O + N H N H O + + N OH H -H2 O N An en amin e 19-30 Enamines – Alkylation at a position. value of enamines is that the -carbon is nucleophilic. The • Enamines undergo SN2 reactions with methyl and 1° haloalkanes, a-haloketones, and a-haloesters. • Treatment of the enamine with one equivalent of an alkylating agent gives an iminium halide. O O •• N + The morph olin e en amin e of cyclohexan on e Br SN 2 3-Bromopropene (Allyl bromide) N Br An iminiu m bromid e (racemic) 19-31 Compare mechanisms of acid catalyzed aldol and enamine H H OH O O CH3 -C-H + CH2 =C-H + :A O CH3 -CH-CH2 -C-H + H-A (racemic) O O •• N + The morph olin e en amin e of cyclohexan on e Br SN 2 3-Bromopropene (Allyl bromide) N Br An iminiu m bromid e (racemic) 19-32 Enamines - Alkylation • Hydrolysis of the iminium halide gives an alkylated aldehyde or ketone. O + N Br- O O + HCl/ H 2 O 2-Allylcyclohexanone + N ClH H Morpholinium chloride Overall process is to render the alpha carbonss of ketone nucleophilic enough so that substitution reactions can occur. 19-33 Enamines – Acylation at a position • Enamines undergo acylation when treated with acid chlorides and acid anhydrides. N Could this be made via a crossed Claisen followed by decarboxylation. O + CH3 CCl Acetyl ch loride + Cl N - O O HCl O + H2 O A n iminium chlorid e (racemic) + N ClH H 2-Acetylcyclohexan on e (racemic) 19-34 Overall, Acetoacetic Ester Synthesis The acetoacetic ester (AAE) synthesis is useful for the preparation of mono- and disubstituted acetones of the following types: O O RX CH3 CCH2 COEt Ethyl acetoacetate (Acetoacetic ester) O CH3 CCH2 R A mon os ubs tituted acetone O CH3 CCHR R' A dis ubs tituted acetone Main points 1. Acidic hydrogen providing a nucleophilic center. 2. Carboxyl to be removed thermally 3. Derived from a halide 19-35 Overall, Malonic Ester Synthesis The strategy of a malonic ester (ME) synthesis is identical to that of an acetoacetic ester synthesis, except that the starting material is a diester rather than a -ketoester. O O RX EtOCCH2 COEt D iethyl malonate (Malonic ester) O RCH2 COH A mon os ubs tituted acetic acid R O RCHCOH A dis ubs tituted acetic acid Main points 1. Acidic hydrogen providing a nucleophilic center 2. Carboxyl group removed by decarboxylation 3. Introduced from alkyl halide 19-36 Malonic Ester Synthesis Consider the synthesis of this target molecule: O These tw o carbons are from diethyl malon ate MeO OH 5-Methoxyp entanoic acid Recognize as substituted acetic acid. Malonic Ester Synthesis 19-37 Malonic Ester Synthesis Steps 1. Treat malonic ester with an alkali metal alkoxide. Na+ COOEt - + EtO Na + COOEt + COOEt Sodiu m s alt of dieth yl malonate COOEt D ieth yl malon ate Sodiu m ethoxide pK a 13.3 (s tronger acid) EtOH Eth anol p Ka 15.9 (w eaker acid) 2. Alkylate with an alkyl halide. Na+ MeO Br COOEt + COOEt SN2 MeO COOEt + Na+ Br- COOEt 19-38 Malonic Ester Synthesis 3. Saponify and acidify. MeO COOEt COOEt 3 . NaOH, H2 O 4 . HCl, H2 O MeO COOH + 2 EtOH COOH 4. Decarboxylation. MeO COOH heat COOH COOH + CO2 MeO 5-Methoxypentanoic acid 19-39 Michael Reaction, addition to a,-unsaturated carbonyl Michael reaction: the nucleophilic addition of an enolate anion to an a,-unsaturated carbonyl compound. • Example: EtOOC O + COOEt Dieth yl 3-Buten-2-one prop anedioate (Methyl vinyl (D iethyl malonate) k eton e) - Et O Na EtOH O + EtOOC COOEt Recognition Pattern: Nucleophile – C – C – CO (nitrile or nitro) 19-40 Michael Reaction These Types of a -Unsaturated Compounds are Nucleophile Acceptors in Michael Reactions O CH2 = CHCH O CH2 = CHCCH 3 O CH2 = CHCOEt O CH2 = CHCNR 2 Aldehyde Ketone Ester Amide CH2 = CHC N Nitrile CH2 = CHN O2 Nitro compound These Types of Compounds Provide Effective Nucleophiles for Michael Reactions O O CH3 CCH2 CCH 3 O O CH3 CCH2 COEt O CH3 CCH2 CN O O Et OCCH2 COEt N CH3 C= CH2 -Diketone -Ketoester -Ketonitrile -Diester Enamine N H3 , RNH2 , R2 NH Amine 19-41 Michael Reaction in base Example:O O - EtO Na + O + O EtOH COOEt Eth yl 3-oxobutanoate (Ethyl acetoacetate) 2-Cyclohexen on e COOEt • The double bond of an a,-unsaturated carbonyl compound is activated for attack by nucleophile. O O O + + More positive carbon 19-42 Mechanism: Michael Reaction Mechanism 1: Set up of nucleophile; Proton transfer to the base. Nu-H + :BBas e Nu:- + H- B 2: Addition of Nu:- to the carbon of the a,-unsaturated carbonyl compound. O Nu + C C C O Nu C C C O Nu C C C Resonance-stabilized enolate anion 19-43 Michael Reaction Step 3: Proton transfer to HB gives an enol. 1 O Nu C C C O-H + 4 H-B 3 2 Nu C C C + B A n enol (a p rodu ct of 1,4-ad dition) Step 4: Tautomerism of the less stable enol form to the more stable keto form. H O O-H Nu C C C Nu C C C Less stable en ol form More stab le keto form 19-44 Michael Reaction, Cautions 1,4 vs 1,2 • Resonance-stabilized enolate anions and enamines are weak bases, react slowly with a,-unsaturated carbonyl compounds, and give 1,4-addition products. • Organolithium and Grignard reagents, on the other hand, are strong bases, add rapidly to carbonyl groups, and given primarily 1,2addition. O PhLi + Phenyl4-Methyl-3lithiu m pen ten-2-one - + Ph O Li Ph OH H2 O HCl 4-Methyl-2-phen yl3-penten -2-ol 19-45 Michael Reaction: Thermodynamic vs Kinetic O C C C fast - ROH - + RO C C C Nu 1,2-Add ition (les s stab le prod uct) Nu O Nu: + C C C s low OH - O Nu C C C ROH H O Nu C C C + RO- 1,4-Add ition (more stable p rodu ct) Addition of the nucleophile is irrevesible for strongly basic carbon nucleophiles (kinetic product) 19-46 Micheal-Aldol Combination a unsaturated Carbanion site O O 1 . Na OEt , Et OH + (Michael reaction) COOEt Ethyl 2-oxocyclohexanecarboxylate 3-Buten-2-one (Methyl vinyl ketone) O COOEt O O 2 . Na OEt , Et OH (Aldol reaction) COOEt Dieckman 19-47 Retrosynthesis of 2,6-Heptadione thes e three carbons from acetoacetic ester O O this b on d formed in a Mich ael reaction O O O O + this carb on los t by decarboxylation COOH COOEt Eth yl acetoacetate Meth yl vinyl k eton e Recognize as substituted acetone, aae synthesis Recognize as Nucleophile – C – C – CO Michael 19-48 Michael Reactions • Enamines also participate in Michael reactions. O N 1 . CH2 =CHCN CN 2 . H2 O, HCl Pyrrolidin e enamine of cycloh exanone + N + H ClH (racemic) 19-49 Gilman Reagents vs other organometallics Gilman reagents undergo conjugate addition to a,-unsaturated aldehydes and ketones in a reaction closely related to the Michael reaction. O O 1 . ( CH3 ) 2 CuLi, eth er, -78°C CH3 3-Methyl-2cyclohexenone 2 . H2 O, HCl CH3 CH3 3,3-D imethylcyclohexanone • Gilman reagents are unique among organometallic compounds in that they give almost exclusively 1,4addition. • Other organometallic compounds, including Grignard reagents, add to the carbonyl carbon by 1,2-addition. 19-50 Crossed Enolate Reactions using LDA With a strong enough base, enolate anion formation can be driven to completion. The base most commonly used for this purpose is lithium diisopropylamide , LDA. LDA is prepared by dissolving diisopropylamine in THF and treating the solution with butyl lithium. [ ( CH3 ) 2 CH] 2 N H + CH3 ( CH2 ) 3 Li Diisopropylamine (pK a 40 (stronger acid) Butyllithium (stronger base) [ ( CH3 ) 2 CH] 2 N - Li + + CH3 ( CH2 ) 2 CH 3 Butane Lithium diis opropylamde pK a 50 (weaker base) (weaker acid) LDA 19-51 Crossed Enolate Reactions using LDA The crossed aldol reaction between acetone and an aldehyde can be carried out successfully by adding acetone to one equivalent of LDA to completely preform its enolate anion, which is then treated with the aldehyde. O Acetone LDA O O Li 1.C6 H5 CH2 CH - OH O + -78°C Lithium enolate C6 H5 2. H 2O 4-Hydroxy-5-phenyl-2-pentanone (racemic) 19-52 Examples using LDA Crossed aldol Michael Alkylation Acylation 19-53 Crossed Enolate Reactions using LDA Question: For ketones with nonequivalent ahydrogens, can we selectively utilize the nonequivalent sites? Answer: A high degree of regioselectivity exists and it depends on experimental conditions. 19-54 Crossed Enolate Reactions using LDA • When 2-methylcyclohexanone is treated with a slight excess of LDA, the enolate is almost entirely the less substituted enolate anion. O slight excess of base + LDA O - Li + O - Li + 0°C + + (racemic) 99% [ ( CH3 ) 2 CH] 2 N H 1% • When 2-methylcyclohexanone is treated with LDA where the ketone is in slight excess, the product is richer in the more substituted enolate. slight excess of the ketone O - Li + O - Li + O + LDA 0°C + + (racemic) 10% 90% [ ( CH3 ) 2 CH] 2 N H 19-55 Crossed Enolate Reactions using LDA The most important factor determining the composition of the enolate anion mixture is whether the reaction is under kinetic (rate) or thermodynamic (equilibrium) control. Thermodynamic Control: Experimental conditions that permit establishment of equilibrium between two or more products of a reaction.The composition of the mixture is determined by the relative stabilities of the products. 19-56 Crossed Enolate Reactions using LDA • Equilibrium among enolate anions is established when the ketone is in slight excess, a condition under which it is possible for proton-transfer reactions to occur between an enolate and an a-hydrogen of an unreacted ketone. Thus, equilibrium is established between alternative enolate anions. O Li+ O O - Li + H CH3 + (racemic) O + Less stable enolate anion (racemic) More stable enolate anion (racemic) 19-57 Crossed Enolate Reactions using LDA Kinetic control: Experimental conditions under which the composition of the product mixture is determined by the relative rates of formation of each product. First formed dominates. • In the case of enolate anion formation, kinetic control refers to the relative rate of removal of alternative a-hydrogens. • With the use of a bulky base, the less hindered hydrogen is removed more rapidly, and the major product is the less substituted enolate anion. • No equilibrium among alternative structures is set up. 19-58 Example 1. 1.01 mol LDA, kinetic control 1. 0.99 mol LDA, thermodynamic control 19-59